Optimized Solid-Phase Extraction (SPE) Cleanup for Metoprolol Tartrate Tablet Analysis: A Comprehensive Guide from Method Development to Validation
This article provides a systematic guide for researchers and drug development professionals on implementing solid-phase extraction (SPE) for the analysis of metoprolol tartrate in tablet formulations and complex biological matrices.
Optimized Solid-Phase Extraction (SPE) Cleanup for Metoprolol Tartrate Tablet Analysis: A Comprehensive Guide from Method Development to Validation
Abstract
This article provides a systematic guide for researchers and drug development professionals on implementing solid-phase extraction (SPE) for the analysis of metoprolol tartrate in tablet formulations and complex biological matrices. Covering the full analytical workflow, it details the foundational principles of SPE, method development tailored to metoprolol's properties, and advanced strategies for troubleshooting common issues like low recovery and poor reproducibility. The content also explores modern validation techniques and comparative analyses with other sample preparation methods, incorporating the latest advancements such as automated online SPE and computational modeling to enhance sensitivity, selectivity, and throughput in pharmaceutical and clinical testing.
Understanding Metoprolol Tartrate and SPE Fundamentals: Principles, Challenges, and Sorbent Selection
Metoprolol tartrate (MPT) is a selective β₁-adrenergic receptor blocking agent extensively used in clinical practice for managing cardiovascular disorders such as hypertension, angina pectoris, and myocardial infarction [1]. Its molecular structure consists of two metoprolol molecules combined with a tartaric acid molecule, giving it the chemical formula 2C₁₅H₂₅NO₃·C₄H₆O₆ and a molecular weight of 684.82 g/mol [1]. This application note details the essential physicochemical properties of MPT and addresses the significant analytical challenges encountered during its quantification in both pharmaceutical dosage forms and complex biological matrices, with particular emphasis on solid-phase extraction (SPE) cleanup methodologies within a comprehensive research framework.
Physicochemical Profile of Metoprolol Tartrate
Understanding the fundamental properties of MPT is crucial for developing robust analytical methods and stable pharmaceutical formulations.
Table 1: Key Physicochemical Properties of Metoprolol Tartrate
| Property | Description / Value | Analytical Significance |
|---|---|---|
| CAS Number | 56392-17-7 [1] | Unique compound identifier for regulatory and literature searches. |
| Melting Point | 120°C [1] | Purity indicator; affects processing in hot-melt methods. |
| Solubility | Highly soluble in water (>1000 mg/mL) [1]; freely soluble in methanol, chloroform; soluble in ethanol and DMSO [1]. | High aqueous solubility poses a challenge for developing extended-release formulations and necessitates rapid polymer hydration to prevent dose dumping [2]. |
| Optical Activity | λₘₐₓ at 223 nm in H₂O [1] | Enables UV-based detection in HPLC. The compound is chiral, requiring specialized methods for enantiomer resolution [3]. |
| BCS Class | Class 1 (High Solubility, High Permeability) [1] | Predicts good absorption and in vivo-in vitro correlation. |
| pKa | Basic compound | Impacts ionization state, which is critical for extraction efficiency and chromatographic retention. |
Solid-State and Stability Characteristics: MPT demonstrates high resistance to γ-irradiation in the solid phase, maintaining its crystallinity at absorbed doses between 20 and 40 kGy, suggesting suitability for radiation-based sterilization processes [4]. However, stability is closely tied to moisture content. Repackaging into unit-dose blister packs requires careful consideration, as studies have shown significant moisture uptake (from 3.5% to 10.5%) when repackaged tablets are stored under accelerated conditions of 40°C/75% relative humidity, which can subsequently affect tablet hardness and dissolution [5].
Analytical Techniques for Quantification
The analysis of MPT spans from simple spectrophotometric methods for bulk drug and tablets to highly sophisticated techniques for biological matrices.
Analysis in Pharmaceutical Dosage Forms
For quality control of tablets, techniques like UV-Vis spectrophotometry offer a simple and cost-effective solution. One established method is based on complex formation with Copper(II) ions [6].
Protocol 3.1.1: Spectrophotometric Determination via Cu(II) Complexation
- Principle: MPT forms a blue-colored, binuclear complex with Cu(II) ions (Cu₂MPT₂Cl₂) in a 1:1 metal-to-ligand ratio, which can be quantified at 675 nm [6].
- Reagents: MPT standard, CuCl₂·2H₂O, Britton-Robinson buffer (pH 6.0).
- Procedure:
- Prepare a stock solution of MPT in water at 0.2 mg/mL.
- Transfer aliquots containing 8.5-70 μg of MPT into a series of 10 mL volumetric flasks.
- Add 1 mL of Britton-Robinson buffer (pH 6.0) and 1 mL of 0.5% (w/v) CuCl₂ solution to each flask.
- Mix well and heat for 20 minutes in a water bath at 35°C.
- Cool the solutions rapidly and dilute to the mark with distilled water.
- Measure the absorbance at 675 nm against a reagent blank.
- Calibration: A linear calibration curve (r = 0.998) is constructed over the range of 8.5-70 μg/mL, with a limit of detection (LOD) of 5.56 μg/mL [6].
For more specific and stability-indicating methods, High-Performance Liquid Chromatography (HPLC) is the standard. A typical protocol for analyzing MPT in tablets, especially after repackaging or stability studies, is outlined below.
Protocol 3.1.2: HPLC-UV Analysis of Tablet Content and Dissolution
- Chromatographic Conditions:
- Column: Reverse-phase C18 (e.g., 150 x 4.6 mm, 5 μm).
- Mobile Phase: Phosphate buffer (pH 7.0) and Acetonitrile (e.g., 70:30 v/v).
- Flow Rate: 1.0 mL/min.
- Detection: UV at 223 nm.
- Internal Standard: Oxprenolol HCl [5].
- Sample Preparation:
- Crush and powder not less than 20 tablets.
- Accurately weigh a portion of the powder equivalent to 50 mg of MPT.
- Dissolve and dilute to an appropriate volume with solvent (e.g., mobile phase or water).
- Filter the solution through a 0.45 μm membrane filter before injection.
- System Suitability: The method should be validated for specificity, accuracy, precision, and linearity. The tailing factor for MPT should be ≤ 2.0, and the RSD of peak responses from replicate injections should be ≤ 2.0% [5].
Analysis in Biological Matrices
The determination of MPT and its metabolites in plasma, urine, or other biological fluids is essential for pharmacokinetic, bioequivalence, and therapeutic drug monitoring (TDM) studies. This requires sophisticated sample cleanup and sensitive detection due to the complex matrix and low concentration levels (ng/mL to μg/mL).
Solid-Phase Extraction (SPE) Cleanup Protocol
SPE is a fundamental sample preparation technique for purifying and concentrating MPT from biological samples, reducing matrix effects, and improving assay sensitivity and reliability [3] [7].
Protocol 3.2.1: SPE for Plasma Sample Cleanup (Exemplary Workflow)
- Sorbent: C18 (Octadecylsilane) or mixed-mode cation exchange (MCX) sorbents are commonly used [3] [7].
- Procedure:
- Conditioning: Pass 1 mL of methanol followed by 1 mL of water (or a mild buffer) through the SPE cartridge. Do not let the sorbent bed run dry.
- Sample Loading: Acidify the plasma sample (e.g., 1 mL) slightly. Load the sample onto the cartridge at a slow, drop-wise flow rate (∼1 mL/min) to maximize analyte retention.
- Washing: Rinse the cartridge with 1-2 mL of a mild aqueous solution (e.g., 5% methanol in water or a weak buffer) to remove polar proteins and salts. For MCX sorbents, this step can include a wash with a mild acid.
- Drying: Centrifuge the cartridge or apply a brief vacuum (∼5 min) to remove residual water.
- Elution: Elute the retained MPT with 2 x 1 mL of a strong organic solvent. For C18, a mixture like Dichloromethane:Isopropanol:Ammonium Hydroxide (80:20:2, v/v/v) is highly effective, achieving recoveries >95% [3]. For MCX, elution is typically performed with a basic organic solvent (e.g., 5% ammonium hydroxide in methanol).
- Reconstitution: Evaporate the eluate to dryness under a gentle stream of nitrogen at 40°C. Reconstitute the residue in 100-200 μL of the initial mobile phase used for LC-MS/MS analysis, vortex mix, and centrifuge before injection.
The following diagram illustrates the logical workflow for selecting and executing an appropriate analytical method based on the sample type and research objective.
Analytical Method Selection Workflow
Advanced Techniques and Research Applications
Enantioselective Separation
MPT is a chiral drug administered as a racemate. The (R)- and (S)-enantiomers may exhibit different pharmacological activities and metabolic rates, driving the need for enantioselective methods [3].
Protocol 4.1.1: Chiral HPLC for Enantiomer Resolution in Plasma
- Principle: Direct separation on a chiral stationary phase.
- Chromatographic Conditions:
- Column: Cellulose tris(3,5-dimethylphenylcarbamate) (e.g., Chiralcel OD) or a Chirobiotic T column [3].
- Mobile Phase: Hexane-Ethanol-Diethylamine (for normal phase) or Methanol-Acetic Acid-Ammonia (100:0.15:0.15, v/v/v) for polar organic mode [3].
- Detection: Fluorescence detection (Ex: 225 nm, Em: 310 nm) or MS/MS.
- Sample Prep: Plasma samples require SPE cleanup (as in Protocol 3.2.1) before injection. This method can accurately quantify enantiomer concentrations as low as 0.5 ng/mL [3].
LC-MS/MS for High-Sensitivity Quantification
Liquid Chromatography coupled with Tandem Mass Spectrometry (LC-MS/MS) represents the gold standard for bioanalysis due to its superior specificity and sensitivity.
Protocol 4.2.1: TurboFlow LC-MS/MS for Direct Plasma Analysis
- Principle: Online SPE coupled with MS/MS detection for high-throughput analysis.
- System Configuration:
- TurboFlow Column: Cyclone P (50 x 0.5 mm) for initial trapping and cleanup.
- Analytical Column: C18 (e.g., 50 x 2.1 mm, 1.9 μm).
- Mobile Phase: 0.1% Formic acid in Water (A) and Acetonitrile (B).
- MS Detection: Positive electrospray ionization (ESI+). Monitor the transition m/z 268.1 → 130.96 for MPT [8].
- Performance: This automated method can achieve a lower limit of quantification (LLOQ) as low as 0.042 ng/L, with a linear range of 5-1000 ng/L, making it ideal for detailed pharmacokinetic studies [8].
The intricate process of analyzing MPT in complex biological samples, integrating advanced sample cleanup and detection, is visualized below.
Advanced Bioanalysis Workflow
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Key Reagents and Materials for MPT Analysis
| Item | Function / Application | Exemplary Details |
|---|---|---|
| Metoprolol Tartrate Reference Standard | Primary standard for calibration and quantification; essential for method validation and ensuring analytical accuracy. | Available from pharmacopoeial sources (USP, BP, EP) [1]. |
| C18 SPE Sorbents | Reversed-phase extraction of MPT from biological fluids; relies on hydrophobic interactions. | Provides high recovery (>95%) with optimized elution solvents like DCM:IPA:NH₄OH [3] [7]. |
| Mixed-Mode Cation Exchange (MCX) Sorbents | SPE for basic compounds like MPT; combines reversed-phase and cation-exchange mechanisms for superior cleanup. | Ideal for complex matrices; elution with basic organic solvent [7]. |
| Chiral HPLC Columns | Resolution of (R)- and (S)-metoprolol enantiomers for stereospecific pharmacokinetic studies. | e.g., Cellulose- or Amylose-based columns (Chiralcel OD, Chiralpak AD) or Chirobiotic T [3]. |
| LC-MS/MS System with TurboFlow | Automated online SPE and high-sensitivity quantification; minimizes manual sample prep. | Uses turbulent flow chromatography for direct injection of plasma; LLOQ in ng/L range [8]. |
| Copper(II) Chloride | Complexing agent for spectrophotometric determination of MPT in pharmaceutical formulations. | Forms a blue 1:1 Cu:MPT complex measurable at 675 nm [6]. |
Solid-phase extraction (SPE) serves as a fundamental sample preparation technique extensively employed in pharmaceutical analysis to isolate and concentrate analytes from complex matrices. This process is crucial for enabling accurate and reproducible quantification in subsequent chromatographic systems. Within the context of research on metoprolol tartrate tablet analysis, SPE provides a robust mechanism for cleaning up samples, thereby mitigating matrix effects and enhancing overall assay sensitivity and specificity. The core principle of SPE revolves around the selective retention and elution of target compounds based on their physicochemical interactions with a solid sorbent material. This article delineates the fundamental mechanisms underpinning SPE technology and provides a detailed workflow for its application in the analysis of metoprolol in pharmaceutical formulations and biological specimens, supporting a broader thesis on analytical method development.
Fundamental Mechanisms of Solid-Phase Extraction
The efficacy of SPE is governed by the selective interactions between the analyte, the solid sorbent, and the liquid matrix. The primary retention mechanisms are categorized as follows:
- Reversed-Phase SPE: This mechanism is predominantly used for isolating non-polar to moderately polar analytes, such as metoprolol, from polar matrices like water or plasma. The sorbent, typically C18 (octadecyl) or C8 (octyl) silica, features hydrophobic alkyl chains. Retention occurs via van der Waals forces and hydrophobic interactions, which are favored in aqueous environments. Analytes are retained while polar interferences are washed away, and elution is achieved using strong organic solvents like methanol or acetonitrile [3] [9].
- Ion-Exchange SPE: This mechanism targets charged molecules. Cation-exchange sorbents (e.g., SCX) retain positively charged bases, while anion-exchange sorbents (e.g., SAX) retain acids. Retention is based on electrostatic attraction between the charged functional groups of the analyte and the oppositely charged sorbent. For metoprolol, which possesses a secondary amine group, a cation-exchange mechanism can be leveraged at a pH where the analyte is protonated. Elution is facilitated by altering the pH or increasing the ionic strength of the solvent to disrupt ionic bonding [9].
- Mixed-Mode SPE: Mixed-mode sorbents, such as Hydrophilic-Lipophilic Balanced (HLB) polymers, combine multiple interaction mechanisms—typically reversed-phase and ion-exchange—within a single cartridge. This offers a broader selectivity and enhanced capacity for cleaning up complex samples, allowing for the simultaneous retention of analytes based on hydrophobicity and ionic interactions [9].
The selection of an appropriate sorbent and mechanism is paramount for developing a successful SPE method, directly influencing the recovery, purity, and reproducibility of the analytical results.
Standard SPE Workflow: A Step-by-Step Protocol
A standardized SPE procedure consists of several critical stages designed to condition the sorbent, retain the target analyte, remove impurities, and finally recover the purified analyte. The following protocol is adapted for the extraction of metoprolol from plasma, a common requirement in pharmacokinetic studies [3] [9] [10].
Protocol for Metoprolol Extraction from Plasma
- Sorbent Selection: A C18 reversed-phase or a mixed-mode cation-exchange sorbent is recommended for metoprolol.
- Conditioning: Pre-wet the sorbent bed with 1-2 column volumes of methanol to solvate the functional groups, followed by 1-2 column volumes of water or a buffer to create an optimal environment for sample loading. It is critical to avoid letting the sorbent bed run dry between conditioning and sample loading [9].
- Sample Loading: Adjust the plasma sample pH to ensure metoprolol is in its neutral form (for reversed-phase) or protonated form (for ion-exchange). Load the sample onto the cartridge at a controlled, moderate flow rate (e.g., 0.5–1 mL/min) to ensure efficient analyte retention without overloading the sorbent's capacity [3] [9].
- Washing: Remove weakly retained matrix interferences by applying 1-3 mL of a solvent with intermediate strength. A common wash solution is a water-organic mixture (e.g., 5% methanol in water) that is strong enough to elute impurities but not the target metoprolol [9].
- Elution: Recover the purified metoprolol by disrupting the analyte-sorbent interactions using a strong solvent. For reversed-phase SPE, methanol or acetonitrile is typically effective. For ion-exchange, an organic solvent modified with a volatile acid or base (e.g., 2% ammonium hydroxide in methanol) is used. A volume of 1-2 mL is standard, and multiple small aliquots may improve recovery [3] [9].
- Post-Elution Processing: Evaporate the eluent to dryness under a gentle stream of nitrogen gas or via vacuum centrifugation. Reconstitute the dry residue in a solvent compatible with the subsequent analytical instrument, such as the initial mobile phase for HPLC analysis [9].
The workflow for this standardized procedure is summarized in the diagram below.
Sorbent Selection and Method Development
Selecting the correct sorbent is the cornerstone of SPE method development. The choice depends on the analyte's chemical structure and the sample matrix composition.
Table 1: Guide to Sorbent Selection for Solid-Phase Extraction
| Sorbent Type | Retention Mechanism | Typical Applications | Considerations for Metoprolol |
|---|---|---|---|
| C18 / C8 | Reversed-Phase (Hydrophobic) | Non-polar to moderately polar organics from aqueous matrices | High retention for metoprolol; elution with methanol or acetonitrile [9]. |
| Mixed-Mode (e.g., HLB, MCX) | Hydrophilic-Lipophilic & Ion Exchange | Broad-spectrum analytes, particularly bases and acids from complex matrices | Offers dual retention; excellent cleanup for plasma samples; allows for stringent washing [9]. |
| Silica | Normal Phase (Polar) | Polar analytes from non-polar solvents | Less common for aqueous biological samples. |
| Ion Exchange (e.g., SCX, SAX) | Ionic Interaction | Charged compounds (Cationic or Anionic) | Effective when metoprolol is protonated; requires careful pH control during loading and elution [9]. |
Method development involves iterative optimization of several parameters:
- Sample Pretreatment: Procedures such as protein precipitation, centrifugation, or pH adjustment are often necessary before loading a biological sample like plasma onto the SPE cartridge [9].
- Wash and Elution Optimization: The solvent strength and volume for the wash and elution steps must be calibrated to maximize the removal of interferences while achieving quantitative recovery of the analyte. This is typically done through stepwise experiments and monitoring recovery via HPLC [9].
Application in Metoprolol Analysis: Exemplary Data
SPE has been successfully integrated into numerous validated analytical methods for metoprolol, demonstrating its reliability for sensitive and precise quantification.
Table 2: Summary of SPE-based HPLC Methods for Metoprolol Quantification
| Application Context | SPE Sorbent / Technique | HPLC & Detection Details | Performance Metrics | Reference |
|---|---|---|---|---|
| Enantiomer Separation in Plasma | Solid-phase extraction (unspecified sorbent) | Chiral stationary phase; Fluorescence detection (λex 225 nm, λem 310 nm) | LOQ: 10 ng/mL; Precise and accurate for pharmacokinetic study [3]. | |
| Pediatric Drug Monitoring | Solid-phase extraction columns | C6 column; Fluorescence detection (λex 225 nm, λem 310 nm) | LOQ: 2.4 ng/mL; Recovery: 73.0 ± 20.5%; Precision (RSD) < 15.5% [10]. | |
| Enantiomer & Metabolite Assay | C2 (ethyl) silica solid-phase extraction | Chirobiotic T column; Fluorescence detection | LOQ: 0.5 ng/mL for enantiomers; Absolute recovery ≥ 95% [3]. |
The Scientist's Toolkit: Essential Research Reagents and Materials
A successful SPE-based analysis requires a suite of specialized reagents and materials. The following table itemizes the key components for an experiment focused on extracting metoprolol from a sample.
Table 3: Essential Materials and Reagents for SPE of Metoprolol
| Item | Function / Purpose | Typical Examples |
|---|---|---|
| SPE Cartridges | The core medium for selective analyte retention. | C18, Mixed-Mode Cation Exchange (MCX), HLB [9] [10]. |
| Organic Solvents | Conditioning, washing, and eluting the sorbent. | Methanol, Acetonitrile, Ethyl Acetate [3] [9]. |
| Aqueous Buffers | pH adjustment for sample and wash steps to control ionization. | Potassium acetate buffer, Ammonia/Acetic acid solutions [3] [10]. |
| Internal Standard | Correction for variability during sample preparation and analysis. | A structurally similar analog, e.g., Atenolol [3]. |
| Evaporation System | Concentrating the eluted sample post-SPE. | Nitrogen evaporator, Vacuum centrifuge [9]. |
Advanced SPE Techniques and Automation
The field of SPE continues to evolve with advancements aimed at improving efficiency, reducing solvent consumption, and facilitating high-throughput analysis.
- Solid-Phase Microextraction (SPME): SPME is a solvent-free technique that integrates sampling, extraction, and concentration into a single step. A fiber coated with a stationary phase is exposed to the sample, and the adsorbed analytes are then desorbed directly into an analytical instrument. This technique is noted for its convenience and compatibility with chromatographic methods [11].
- Automation and High-Throughput SPE: Modern laboratories increasingly employ automated SPE systems to process large numbers of samples, such as those generated in clinical trials or quality control. Formats like 96-well plate SPE are handled by robotic liquid handlers, which enhance precision, improve reproducibility, and significantly reduce manual labor and potential for human error [9] [12].
Solid-phase extraction remains an indispensable tool in the analytical scientist's arsenal, particularly for the precise analysis of pharmaceuticals like metoprolol in complex matrices. A deep understanding of its core principles—the interaction mechanisms between analyte, sorbent, and solvent—is fundamental. The structured workflow of conditioning, loading, washing, and elution, when properly optimized, yields clean extracts that are vital for the accuracy and longevity of sophisticated detection systems like HPLC. As demonstrated through various applications, a well-developed SPE method provides robust, reproducible, and sensitive quantification, forming a critical component of rigorous pharmaceutical research and development.
The analysis of active pharmaceutical ingredients (APIs) in complex matrices, such as tablet formulations, requires sophisticated sample preparation to ensure accuracy, sensitivity, and reproducibility. This application note details the strategic selection and use of solid-phase extraction (SPE) sorbents for the cleanup and analysis of metoprolol tartrate, a widely prescribed β-adrenergic blocker, from tablet formulations. The context is framed within a broader thesis research on SPE cleanup for metoprolol tartrate tablet analysis. Metoprolol is a basic compound (pKa ~9.7) possessing both hydrophobic aromatic rings and a polar, ionizable secondary amine functional group, making it an ideal candidate for evaluating multiple SPE retention mechanisms. We provide a comparative evaluation of reversed-phase, ion-exchange, and mixed-mode SPE, including structured protocols, quantitative recovery data, and a detailed reagent toolkit to guide researchers and drug development professionals in optimizing their analytical methods.
Sorbent Selection: Mechanisms and Rationale
The selection of an appropriate SPE sorbent is predicated on the physicochemical properties of the analyte and the composition of the sample matrix. For basic compounds like metoprolol tartrate, three primary sorbent chemistries are relevant.
2.1 Reversed-Phase Sorbents Reversed-phase (RP) sorbents (e.g., C8, C18) retain analytes via non-polar, van der Waals interactions between the analyte's hydrophobic moieties and the alkyl chains bonded to the sorbent surface [13]. For metoprolol, which contains a hydrophobic aryl-oxypropyl chain, these sorbents can provide adequate retention from polar aqueous samples. However, in purely RP mode, the polar, ionizable amine can lead to poor retention and peak tailing, especially if the sample matrix has a high ionic strength or if the analysis is conducted without careful pH control [14]. RP sorbents are most effective when the analyte is in its uncharged, neutral form.
2.2 Ion-Exchange Sorbents Ion-exchange (IEX) sorbents retain analytes through electrostatic interactions between the charged functional groups on the analyte and the oppositely charged functional groups on the sorbent surface [15]. For a basic compound like metoprolol, which carries a positive charge on its amine group at a pH below its pKa (~9.7), a cation-exchange sorbent is required.
- Strong Cation Exchange (SCX): Contains a sulfonic acid group (SO3-) that is permanently negatively charged across the entire operational pH range [15] [13]. It is ideally paired with weak bases, like metoprolol, because the analyte's charge can be "turned off" for elution by raising the pH above its pKa [16].
- Weak Cation Exchange (WCX): Typically contains a carboxylic acid group (COOH) that is charged only at higher pH (typically >5) [15]. WCX is best suited for retaining strong bases that possess a permanent positive charge, as the sorbent's charge can be neutralized for elution by lowering the pH [16].
2.3 Mixed-Mode Sorbents Mixed-mode sorbents incorporate two or more orthogonal retention mechanisms, typically reversed-phase (e.g., C8) and ion-exchange (e.g., SCX), on the same sorbent particle [13]. Products like ISOLUTE HCX (C8/SCX) are specifically designed for basic compounds [17]. This dual mechanism allows for superior selectivity and cleaner extracts. Analytes are initially retained by both mechanisms. Stringent washes (e.g., with organic solvents or buffers at high ionic strength) can then be used to remove non-polar and polar matrix interferences without prematurely eluting the analyte, which remains locked via the ion-exchange mechanism. Elution is achieved with a solvent that disrupts both mechanisms, typically a volatile organic solvent like methanol modified with a base (e.g., ammonium hydroxide) to neutralize the analyte's charge [17] [18]. This combination provides a robust platform for extracting basic drugs like metoprolol from complex biological and formulation matrices.
Table 1: Comparison of SPE Sorbents for Basic Compounds like Metoprolol
| Sorbent Type | Retention Mechanism | Optimal Sample pH | Elution Condition | Key Advantages | Key Limitations |
|---|---|---|---|---|---|
| Reversed-Phase (e.g., C8) | Van der Waals forces (non-polar) | ≥ 2 pH units above pKa (Neutral form) | Organic solvent (e.g., Methanol, Acetonitrile) | Simple protocol; wide applicability | Poor retention of polar bases; susceptible to matrix effects |
| Strong Cation Exchange (SCX) | Electrostatic attraction to SO3- group | ≤ 2 pH units below pKa (Charged form) | High ionic strength buffer OR pH ≥ pKa + 2 | Excellent retention of charged bases; clean extracts | Requires careful pH control; may need high-salt elution |
| Mixed-Mode (C8/SCX) | Van der Waals + Electrostatic | ≤ 2 pH units below pKa (Charged form) | Organic solvent + Base (e.g., 5% NH4OH in MeOH) | Superior cleanup; very high selectivity; flexible washing | More complex protocol than single-mode sorbents |
Experimental Protocols
The following protocols are adapted from generic methods for basic pharmaceuticals and can be applied to the extraction of metoprolol tartrate from powdered tablet samples [18].
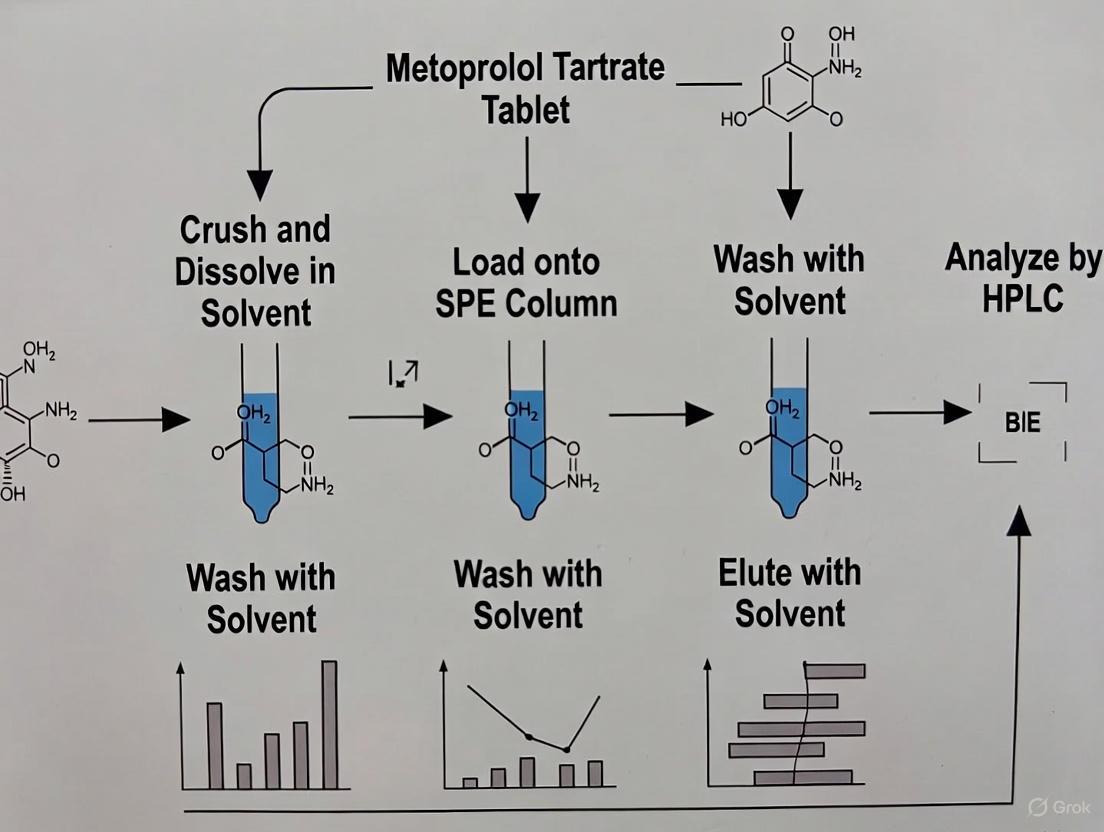
3.1 Protocol for Mixed-Mode SPE (ISOLUTE HCX or DSC-MCAX) This protocol is recommended for obtaining the cleanest extracts of metoprolol from tablet formulations.
Table 2: Reagents and Solutions for Mixed-Mode SPE
| Reagent/Solution | Function in the Protocol |
|---|---|
| Methanol | Conditions the sorbent bed and removes non-polar impurities. |
| 50 mM Ammonium Acetate Buffer (pH 6) | Equilibrates the sorbent at a pH where both the sorbent's SCX group and metoprolol (pKa ~9.7) are charged. |
| 1% Acetic Acid in Water | Acidic wash to remove very polar interferences and residual proteins/peptides. |
| Methanol | Organic wash to remove non-polar and neutral interferences while metoprolol is retained by ion-exchange. |
| 5% Ammonium Hydroxide in Methanol | Elution solvent. The base neutralizes metoprolol, breaking the ion-exchange bond, while methanol disrupts the reversed-phase interaction. |
Steps:
- Sample Preparation: Weigh and finely powder ten tablets. Transfer an amount equivalent to 40 mg of metoprolol tartrate to a conical flask. Extract with 4 x 20 mL of deionized water, filter into a 100 mL volumetric flask, and make up to the mark with water [19].
- Conditioning: Load a 100 mg/3 mL mixed-mode HCX or MCAX SPE cartridge with 1 mL of methanol and allow it to drip to waste.
- Equilibration: Load 1 mL of 50 mM ammonium acetate buffer (pH 6) to the cartridge. Do not let the sorbent bed run dry.
- Sample Loading: Dilute the tablet extract 1:1 with 50 mM ammonium acetate buffer (pH 6). Load the diluted sample onto the cartridge at a controlled flow rate of approximately 1 mL/min [18].
- Washing: Pass the following solutions through the cartridge to waste:
- 1 mL of 50 mM ammonium acetate buffer (pH 6).
- 1 mL of 1% acetic acid in water.
- 1 mL of methanol. Ensure the cartridge is drained completely after the methanol wash.
- Elution: Elute the purified metoprolol into a clean collection tube using 2 x 1 mL of 5% ammonium hydroxide in methanol. The eluate can be evaporated to dryness under a gentle stream of nitrogen and reconstituted in a mobile phase compatible with the subsequent HPLC analysis.
3.2 Protocol for Strong Cation Exchange (SCX) SPE This protocol utilizes a purely ion-exchange mechanism.
Steps:
- Conditioning & Equilibration: Condition the SCX cartridge with methanol followed by water. Equilibrate with 1 mL of a buffer at pH ~7.7 (e.g., 10 mM potassium phosphate), ensuring the sorbent is charged and the metoprolol is fully protonated [15] [16].
- Sample Loading: Load the prepared tablet extract (in a weak aqueous buffer at pH ~7.7).
- Washing: Wash with the equilibration buffer, followed by a wash with 100% methanol to remove uncharged interferences [15].
- Elution: Elute metoprolol using a buffer with high ionic strength (e.g., 1 M ammonium acetate) or a buffer at pH ≥ 11.7 to neutralize the analyte [15].
3.3 Spectrophotometric Determination of Metoprolol The extracted metoprolol can be quantified using a simple and sensitive spectrophotometric method based on complexation with Cu(II) ions [19].
- Procedure: Transfer an aliquot of the reconstituted SPE eluate containing 8.5-70 μg of metoprolol to a 10 mL volumetric flask. Add 1 mL of Britton-Robinson buffer (pH 6.0) and 1 mL of 0.5% (w/v) CuCl₂·2H₂O solution. Mix well and heat for 20 minutes in a water bath at 35°C. Cool rapidly, make up to volume with distilled water, and measure the absorbance at 675 nm against a reagent blank [19].
- Calibration: A calibration curve plotted within the 8.5-70 μg/mL range typically shows a good correlation coefficient (r = 0.998) [19].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagent Solutions for SPE of Metoprolol
| Item | Function / Explanation |
|---|---|
| ISOLUTE HCX SPE Cartridges (100 mg/3 mL) | Mixed-mode sorbent (C8/SCX) providing dual retention for superior cleanup of basic drugs from complex matrices [17]. |
| Discovery DSC-MCAX SPE Cartridges | An alternative mixed-mode sorbent also containing C8 and benzenesulfonic acid (SCX) groups [18]. |
| Ammonium Acetate | Used to prepare volatile buffers for sample dilution and SPE washing; compatible with downstream LC-MS analysis. |
| Ammonium Hydroxide (e.g., 5% in MeOH) | A volatile base used in the elution solvent for mixed-mode and SCX SPE to neutralize basic analytes [18]. |
| Copper(II) Chloride Dihydrate | Reacts with metoprolol to form a colored complex for spectrophotometric detection at 675 nm [19]. |
| Britton-Robinson Buffer (pH 6.0) | A universal buffer used to maintain the optimal pH for the formation of the metoprolol-Cu(II) complex [19]. |
| High-pH Stable C18 HPLC Column | Recommended for the final chromatographic separation, as it provides superior peak shape for basic compounds like metoprolol in their neutral form [14]. |
Workflow and Sorbent Selection Logic
The following diagrams illustrate the experimental workflow and the logical process for selecting the appropriate sorbent.
Diagram 1: SPE Workflow for Mixed-Mode Extraction
Diagram 2: Sorbent Selection Logic
Solid-phase extraction (SPE) is a fundamental sample preparation technique critical for purifying and concentrating analytes from complex matrices. For the accurate quantification of active pharmaceutical ingredients such as metoprolol tartrate from tablet formulations or biological samples, a meticulously optimized SPE protocol is indispensable. This application note details a standardized SPE procedure, framed within broader thesis research on metoprolol analysis, providing drug development professionals with a validated method to achieve high recovery and minimal matrix interference. The protocols outlined leverage mixed-mode cationic exchange sorbents, which are particularly effective for basic compounds like metoprolol (pKa ~9.7), ensuring selective extraction and clean chromatographic results [20].
Experimental Protocols and Workflows
Reagents and Materials
The following reagents and solutions are required for the SPE procedure:
- SPE Cartridges: Mixed-mode Weak Cation Exchange (WCX) cartridges (e.g., 60 mg/3 mL bed mass) [20] [21].
- Conditioning Solvent: HPLC-grade methanol.
- Equilibration Solvent: Deionized water with 1% (v/v) formic acid [21].
- Wash Solvent 1: Deionized water with 1% (v/v) formic acid [21].
- Wash Solvent 2: HPLC-grade methanol [21].
- Elution Solvent: Methanol with 5% (v/v) ammonium hydroxide [20] [21].
- Sample Diluent: Aqueous solution with 1% formic acid to ensure metoprolol is in its ionized, protonated form for optimal retention on the WCX sorbent [21].
Sample Preparation
For the analysis of metoprolol tartrate tablets, prepare a sample solution by dissolving a powdered tablet in an appropriate aqueous solvent. Prior to SPE loading, the sample must be acidified with formic acid to a final concentration of approximately 1% to ensure the metoprolol (a basic compound) is fully protonated and positively charged [21].
Optimized SPE Procedure for Metoprolol
The sequential SPE protocol is designed to maximize analyte recovery while removing interfering contaminants. The workflow and key parameters for each stage are summarized in the following diagram and table.
Table 1: Detailed Steps for the Optimized SPE Protocol for Metoprolol.
| Step | Objective | Recommended Solvent & Volume | Critical Parameters & Rationale |
|---|---|---|---|
| 1. Conditioning | Activate sorbent and solvate functional groups. | 500 μL HPLC-grade Methanol [21]. | Ensures the sorbent is prepared for optimal interaction with the analyte. Do not let the sorbent dry out after this step [21]. |
| 2. Equilibration | Create a compatible environment for sample loading. | 500 μL Deionized Water with 1% Formic Acid [21]. | The low pH maintains the protonated state of metoprolol, facilitating immediate retention upon loading. |
| 3. Sample Loading | Retain the analyte on the sorbent. | Load the acidified sample solution at a flow rate of ~1 mL/min [21]. | Sample pH is critical; acidification ensures metoprolol is charged for strong ionic interaction with the WCX sorbent [20] [21]. |
| 4. Washing | Remove weakly retained interferences and matrix components. | Wash 1: 500 μL Water with 1% Formic Acid [21].Wash 2: 500 μL Methanol [21]. | The first wash (aqueous, acidic) removes polar impurities. The second wash (organic) removes less polar interferences without eluting the ionically bound metoprolol. |
| 5. Drying | Remove residual water to enable efficient elution. | Apply full vacuum for several minutes [21]. | Eliminates water that can dilute the elution solvent and reduce recovery, especially for hydrophobic interactions. Check dryness by touching the cartridge; it should not feel cool [21]. |
| 6. Elution | Disrupt analyte-sorbent interactions and recover the analyte. | 500 μL Methanol with 5% Ammonium Hydroxide [20] [21]. | The high pH deprotonates metoprolol, neutralizing its charge and breaking the ionic bond. The organic solvent disrupts hydrophobic interactions. |
Optimization of Critical SPE Parameters
Successful SPE method development requires systematic optimization of key parameters to achieve maximum recovery and cleanliness. The following table outlines the primary variables to investigate for a metoprolol-specific method.
Table 2: Key Parameters for Optimization of SPE for Metoprolol Analysis.
| Parameter | Influence on SPE Performance | Recommended Optimization Range for Metoprolol |
|---|---|---|
| Sorbent Chemistry | Determines the primary retention mechanism (reversed-phase, ion-exchange). | Mixed-mode Weak Cation Exchange (WCX) is optimal for basic drugs [20] [21]. |
| Sample Load pH | Controls the ionization state of the analyte and its retention on ion-exchange sorbents. | Acidify to pH ~2-3 (at least 2 units below pKa ~9.7) to ensure metoprolol is protonated [22] [21]. |
| Elution Solvent Composition | Disrupts specific analyte-sorbent interactions. | 80-100% Methanol with 2-5% Ammonium Hydroxide [22] [20]. |
| Elution Volume | Must be sufficient to completely displace the analyte from the sorbent bed. | 2-5 Bed Volumes (e.g., 4-10 mL for a 3 mL cartridge); test with 2-3 consecutive fractions [22]. |
| Flow Rate | Affects the contact time between analyte and sorbent, influencing retention and elution efficiency. | Maintain at 1-2 mL/min during loading and elution for consistent performance [21]. |
Troubleshooting Common Issues
- Low Recovery: Ensure the sorbent does not dry out between conditioning and sample loading steps. Verify the elution solvent strength and pH are sufficient to completely neutralize the analyte's charge and disrupt hydrophobic interactions. Increasing the elution volume or adding a small percentage of a stronger solvent like isopropanol can help elute highly hydrophobic analytes [22].
- Excessive Matrix Co-elution: Introduce an intermediate wash step with a solvent of moderate elution strength to remove impurities without displacing the target analyte. For example, a wash with 5% methanol in water can remove very polar contaminants, while a methanol wash can remove less polar interferences before the final elution [22] [21].
- Poor Chromatographic Peak Shape/Ion Suppression in LC-MS: Replace non-volatile salts in wash or elution buffers with volatile additives (e.g., ammonium formate, formic acid, ammonium hydroxide) to prevent source contamination and signal suppression [22] [20].
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Materials and Reagents for SPE Clean-up of Metoprolol.
| Item | Function / Rationale |
|---|---|
| Mixed-Mode WCX SPE Cartridges | The sorbent provides dual retention mechanisms (reversed-phase and ion-exchange) for highly selective clean-up of basic metoprolol from complex matrices [20]. |
| Formic Acid (ACS Grade) | Used to acidify the sample and wash buffers. Ensures metoprolol remains protonated and positively charged for strong retention on the WCX sorbent [20] [21]. |
| Ammonium Hydroxide (LC-MS Grade) | A volatile base used in the elution solvent to deprotonate metoprolol, neutralizing its charge and enabling efficient elution. Its volatility prevents interference in downstream LC-MS analysis [20]. |
| HPLC-Grade Methanol & Acetonitrile | High-purity organic solvents are used for conditioning, washing, and elution. Methanol is often preferred for its ability to elute a wide range of compounds, while acetonitrile can provide sharper peaks in LC-MS [22]. |
This application note provides a robust and optimized SPE protocol for the clean-up and pre-concentration of metoprolol tartrate, a critical step in pharmaceutical analysis. By adhering to the detailed procedures for conditioning, loading, washing, and elution—and by understanding the underlying principles of parameter optimization—researchers can achieve highly reproducible results with excellent recovery and minimal matrix effects. The use of mixed-mode WCX SPE, coupled with careful pH control, offers a superior strategy for isolating metoprolol, ensuring the reliability of subsequent chromatographic quantification in both formulation and bioanalytical studies.
Developing a Robust SPE Method for Metoprolol: A Step-by-Step Protocol from Sample Prep to Analysis
Sample Pre-treatment Strategies for Tablet Formulations and Biological Fluids
Sample pre-treatment is a critical and often rate-limiting step in pharmaceutical analysis, particularly for complex biological matrices and solid dosage forms. For researchers investigating metoprolol tartrate tablet analysis, effective sample preparation is essential to achieve accurate, reproducible, and reliable results. This application note provides detailed protocols and strategies for sample pre-treatment of tablet formulations and biological fluids, specifically contextualized within solid-phase extraction (SPE) cleanup for metoprolol tartrate research. The methodologies outlined enable researchers to overcome significant analytical challenges posed by complex matrices, including contamination, analyte loss, and degradation, which are particularly emphasized in biological samples and can lead to substantial errors if not properly addressed [23].
Sample Preparation Strategies for Tablet Formulations
Fundamental Challenges in Tablet Analysis
Tablet formulations present unique challenges for analytical scientists, including the need to dissolve the solid matrix, ensure complete analyte extraction, and eliminate interference from excipients, binders, fillers, and other inactive ingredients. For metoprolol tartrate tablets, sample preparation must achieve complete dissolution while maintaining analyte stability and preventing degradation during processing.
Standard Protocol for Tablet Dissolution and Preparation
Materials: Metoprolol tartrate tablets, analytical grade methanol, acetonitrile, and water, ultrasonic bath, volumetric flasks, syringe filters (0.45 μm or 0.2 μm).
Procedure:
- Weighing: Accurately weigh 5 intact metoprolol tartrate tablets and calculate average weight.
- Crushing: Gently crush tablets into fine powder using mortar and pestle.
- Homogenization: Mix powder thoroughly to ensure uniform distribution.
- Aliquot Transfer: Transfer powder equivalent to one tablet to 100 mL volumetric flask.
- Solvent Addition: Add approximately 70 mL of methanol:water (50:50, v/v) solution.
- Sonication: Sonicate mixture for 30 minutes with occasional shaking.
- Dilution: Bring to volume with same solvent mixture and mix well.
- Centrifugation: Centrifuge at 4000 rpm for 10 minutes if particulate matter persists.
- Filtration: Filter supernatant through 0.45 μm syringe filter.
- Dilution: Further dilute with mobile phase as needed for analysis.
Critical Considerations:
- Method validation should establish complete extraction efficiency.
- Solvent composition may require optimization based on tablet excipients.
- Stability of metoprolol in solution should be verified over analysis period.
Sample Preparation Strategies for Biological Fluids
Complexity of Biological Matrices
Biological samples present significantly greater challenges than tablet formulations due to their complex composition. Plasma, serum, and other biofluids contain numerous interfering components including proteins, phospholipids, salts, and endogenous compounds that can obstruct analyte detection and quantification [23]. For metoprolol analysis, these matrix effects are particularly problematic because the drug typically circulates at nanogram to microgram per milliliter concentrations, requiring highly sensitive and selective sample preparation techniques [8].
Comprehensive Comparison of Biological Sample Preparation Techniques
Table 1: Comparison of Biological Sample Preparation Techniques for Metoprolol Analysis
| Technique | Principle | Best For | Recovery | Advantages | Limitations |
|---|---|---|---|---|---|
| Protein Precipitation (PPT) | Organic solvent denatures and precipitates proteins | High-throughput analysis, minimal method development | Moderate to high | Rapid, simple, low cost | Does not remove phospholipids, matrix effects possible [24] |
| Phospholipid Depletion (PLD) | Removes phospholipids using specialized sorbents | LC-MS/MS applications where phospholipids cause ion suppression | High for analytes | Specifically targets phospholipids, reduces ion suppression | Additional step after PPT, increased cost [24] |
| Liquid-Liquid Extraction (LLE) | Partitioning between immiscible solvents | Broad range of analytes, traditional approach | High | Effective clean-up, high recovery | Emulsion formation, difficult to automate [25] [24] |
| Solid-Phase Extraction (SPE) | Selective adsorption/desorption from solid phase | High purity extracts, complex matrices | High | Excellent clean-up, customizable, concentrative | Requires method development, higher cost [25] |
| Supported Liquid Extraction (SLE) | LLE on solid support | Replacement for traditional LLE | Very high | No emulsions, easier automation, high recovery | Limited by partitioning coefficients [24] |
| Turbulent Flow Chromatography | Online extraction using high flow rates | Automated analysis, high throughput | High | Full automation, high throughput | Specialized equipment required [8] [25] |
Detailed Protocol: Solid-Phase Extraction for Metoprolol in Plasma
Materials: C18 SPE cartridges (100 mg/3 mL), vacuum manifold, metoprolol standard, internal standard (e.g., bisoprolol fumarate), methanol, acetonitrile, water, formic acid, ammonium acetate, human plasma samples.
SPE Procedure:
- Conditioning: Condition SPE cartridge with 2 mL methanol followed by 2 mL deionized water at flow rate of 1-2 mL/min.
- Sample Preparation: Thaw frozen plasma samples at room temperature and vortex mix. Transfer 1 mL aliquot to clean tube. Add 50 μL internal standard working solution (1 μg/mL bisoprolol fumarate). Add 2 mL 0.1 M ammonium acetate buffer (pH 6.0) and vortex mix.
- Loading: Load prepared sample onto conditioned SPE cartridge at controlled flow rate (1-2 mL/min).
- Washing: Wash with 2 mL deionized water followed by 2 mL 5% methanol in water.
- Drying: Apply full vacuum for 10 minutes to dry sorbent bed completely.
- Elution: Elute metoprolol with 2 × 1 mL aliquots of methanol:acetonitrile (80:20, v/v) with 0.1% formic acid into clean collection tubes.
- Evaporation: Evaporate eluent to dryness under gentle nitrogen stream at 40°C.
- Reconstitution: Reconstitute dry residue in 200 μL mobile phase (0.1% formic acid in water:acetonitrile, 70:30, v/v) and vortex mix thoroughly.
- Analysis: Transfer to autosampler vials for LC-MS/MS analysis.
Method Optimization Tips:
- Condition and wash steps are critical for removing interfering compounds.
- Complete drying prevents residual water from diluting elution solvent.
- Quantitative elution may require optimization of solvent composition and volume.
Advanced Protocol: Online SPE-LC-MS/MS for High-Throughput Metoprolol Analysis
For laboratories requiring high-throughput analysis, online SPE coupled directly with LC-MS/MS provides an efficient solution. This approach has been successfully applied to metoprolol analysis in plasma, demonstrating excellent performance characteristics [8].
Materials and Instrumentation: Transcend TLX HPLC system with TurboFlow Cyclone-P column (50 × 0.5 mm), Thermo Gold C18 analytical column (50 × 2.1 mm, 1.9 μm), TSQ Quantum Access Max Mass Spectrometer, mobile phases: 0.1% formic acid in water (FMA) and acetonitrile with 0.1% formic acid (FMB).
Online SPE Procedure:
- Sample Preparation: Dilute 100 μL plasma with 300 μL internal standard solution in 0.1% formic acid. Centrifuge at 13,000 rpm for 5 minutes.
- Loading: Inject 100 μL supernatant onto TurboFlow Cyclone-P column using loading pump with 0.1% formic acid in water at 1.5 mL/min for 60 seconds.
- Transfer: Reduce elution flow rate to 0.1 mL/min over 30 seconds. Switch mobile phase to 40% FMA(C2) and 60% FMB(C2) to transfer analytes from TurboFlow column to analytical column within 30 seconds.
- Separation: Elute analytes on UHPLC column using isocratic method with 50% FMA(C1) and 50% FMB(C1).
- Reconditioning: Simultaneously recondition TurboFlow column with 100% FMB(C2) for 60 seconds.
- Detection: Quantify metoprolol using triple quadrupole mass spectrometry with transition m/z 268.1 → m/z 130.96, and internal standard bisoprolol fumarate at m/z 326.3 → m/z 116.2 [8].
Validation Parameters: This method has demonstrated linearity over 5-1000 ng/L range, with LLOQ of 0.042 ng/L. Precision and accuracy show maximum CV% of 10.28 and maximum relative error of 5.38%, suitable for bioequivalence studies [8].
Quantitative Data for Method Selection
Table 2: Performance Characteristics of Sample Preparation Methods for Metoprolol
| Parameter | Protein Precipitation | Liquid-Liquid Extraction | Offline SPE | Online SPE |
|---|---|---|---|---|
| Sample Volume | 100-200 μL | 500-1000 μL | 500-1000 μL | 50-100 μL |
| Extraction Time | 30 min | 45-60 min | 60-90 min | 4.5 min (runtime) |
| Recovery (%) | 70-85 | 85-95 | 90-98 | 85-95 |
| Matrix Effect (%) | Significant (ion suppression) | Moderate | Minimal | Controlled (89% reported) [8] |
| Cost per Sample | Low | Low to moderate | Moderate | High (equipment) |
| Throughput | High | Moderate | Moderate | Very high |
| Automation Potential | Moderate | Low | Moderate | Full |
| LLOQ | 1-5 ng/mL | 0.5-1 ng/mL | 0.1-0.5 ng/mL | 0.042 ng/L [8] |
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Reagents and Materials for Metoprolol Sample Preparation
| Item | Specification | Application/Function |
|---|---|---|
| Solid-Phase Extraction Cartridges | C18, 100 mg/3 mL | Primary extraction and clean-up of metoprolol from biological samples |
| Internal Standard | Bisoprolol fumarate | Quantification standard for LC-MS/MS analysis [8] |
| Mass Spectrometry Solvents | HPLC grade methanol, acetonitrile with 0.1% formic acid | Mobile phase components for chromatographic separation [8] |
| TurboFlow Cyclone-P Column | 50 × 0.5 mm | Online sample clean-up and concentration [8] |
| Analytical Column | C18, 50 × 2.1 mm, 1.9 μm | Chromatographic separation of metoprolol [8] |
| Ammonium Acetate | Analytical grade, for buffer preparation | pH adjustment and buffer for SPE procedures |
| Formic Acid | LC-MS grade, ≥99% | Mobile phase modifier to enhance ionization [8] |
| Beta-Glucuronidase Enzyme | Helix pomatia or recombinant forms | Urine hydrolysis for metabolite analysis [24] |
| Phospholipid Depletion Plates | 96-well format | Specific removal of phospholipids from plasma samples [24] |
| Protein Precipitation Plates | 96-well format with filters | High-throughput protein removal from biological samples [24] |
Workflow Visualization
Sample Preparation Workflows for Pharmaceutical Analysis
Effective sample pre-treatment is fundamental to successful metoprolol tartrate analysis in both tablet formulations and biological fluids. The selection of appropriate sample preparation methodology should be guided by analytical requirements, including required sensitivity, throughput, available equipment, and matrix complexity. For tablet analysis, straightforward dissolution and filtration typically suffice, while biological samples demand more sophisticated clean-up approaches to address matrix effects. Solid-phase extraction, particularly in its online format, provides an optimal balance of clean-up efficiency, recovery, and throughput for metoprolol analysis in plasma, making it especially suitable for pharmacokinetic studies and bioequivalence assessments where accuracy, precision, and sensitivity are paramount.
Metoprolol tartrate is a cardioselective β1-adrenergic receptor blocker widely used for treating cardiovascular conditions such as hypertension, angina pectoris, and myocardial infarction [26]. The analysis of metoprolol in pharmaceutical formulations and biological samples requires precise and efficient sample preparation to isolate the analyte from complex matrices. Solid-phase extraction (SPE) has emerged as a powerful technique for this purpose, offering superior clean-up capabilities compared to traditional methods like liquid-liquid extraction or protein precipitation [27].
The complexity of variables in solid-phase extraction presents significant challenges in analytical development, making the optimization of extraction conditions essential for obtaining reliable results [28]. This application note details an optimized SPE protocol specifically developed for metoprolol analysis, with particular emphasis on sorbent mass, solvent volumes, and flow rates. The protocol is framed within broader thesis research on SPE cleanup for metoprolol tartrate tablet analysis, providing researchers and drug development professionals with a validated method that ensures high recovery and reproducibility while minimizing matrix effects in subsequent chromatographic analysis [20].
Theoretical Background
Metoprolol Physicochemical Properties
Metoprolol, chemically known as 1-(isopropylamino)-3-[4-(2-methoxyethyl)phenoxy]-2-propanol, is a moderately lipophilic basic compound with a molecular weight of 267.36 g/mol [29]. Its partition coefficient (log P) is approximately 0.6, and it features a secondary amine functional group with a pKa of ~9.7 [20] [29]. These properties significantly influence its behavior in SPE, particularly its retention characteristics on different sorbent materials. The alkaline nature (pKa ∼9.7) and presence of a secondary amine make metoprolol particularly suitable for retention with mixed-mode cationic sorbents, which exploit both reversed-phase and ion-exchange mechanisms for selective extraction [20].
SPE Principles for Basic Drugs
The effective extraction of metoprolol requires understanding of multiple interaction mechanisms. Reversed-phase retention relies on hydrophobic interactions between the drug's aromatic rings and alkyl chains of sorbents, while ion-exchange mechanisms utilize the protonated secondary amine under acidic conditions [9]. The optimal pH for sample loading is typically 2-3 units below the pKa of the analyte to ensure positive charge on the amine group, enhancing retention on mixed-mode cationic exchangers [20]. The selectivity achieved through these complementary mechanisms enables effective separation of metoprolol from complex matrix components in pharmaceutical formulations and biological samples [27].
Optimized SPE Protocol
Materials and Equipment
- SPE Cartridges: Mixed-mode cationic exchange (MCX) cartridges (30 mg, 1 mL bed volume) [20]
- Solvents: Methanol (MeOH), acetonitrile (ACN), deionized water, ammonium hydroxide (NH₄OH), formic acid (HCOOH) - all HPLC grade [20] [8]
- Equipment: Vacuum manifold system, calibrated pH meter, positive displacement pipettes, evaporation system (nitrogen evaporator or centrifugal vacuum concentrator) [9]
- Solutions:
- Conditioning solvent: Methanol
- Equilibration solvent: Deionized water acidified to pH 2-3 with formic acid
- Wash solvent: 2% (v/v) formic acid in water
- Elution solvent: 5% ammonium hydroxide in methanol [20]
Sample Preparation
For metoprolol tartrate tablet analysis, accurately weigh and pulverize ten tablets. Transfer a powder quantity equivalent to 40 mg metoprolol to a conical flask and extract with 4 × 20 mL of water. Filter into a 100 mL volumetric flask and dilute to volume with water [19]. For plasma samples, prior to SPE, precipitate proteins by adding 300 μL of acetonitrile to 100 μL of plasma, vortex for 30 seconds, and centrifuge at 10,000 × g for 5 minutes [20].
SPE Procedure
The optimized protocol employs a 4-step SPE procedure on mixed-mode cationic exchange cartridges:
Conditioning: Activate the sorbent with 1 mL of methanol, followed by 1 mL of acidified water (pH 2-3 with formic acid). Apply gentle vacuum to maintain a flow rate of approximately 1 mL/min. Do not allow the sorbent to dry between steps [20] [9].
Sample Loading: Adjust the sample pH to 2-3 with formic acid. Load the prepared sample at a controlled flow rate of 0.5-1 mL/min to ensure optimal retention [20] [9].
Washing: Remove matrix interferences with 1 mL of 2% formic acid in water, followed by 1 mL of methanol. Maintain a flow rate of 1 mL/min [20].
Elution: Elute metoprolol with 1 mL of 5% ammonium hydroxide in methanol. Collect the eluate in a clean tube at a flow rate of 0.5-1 mL/min [20].
Post-Extraction Processing
Evaporate the eluate to dryness under a gentle stream of nitrogen at 40°C. Reconstitute the residue in 100-200 μL of mobile phase compatible with your chromatographic system (e.g., water:acetonitrile, 90:10, v/v). Vortex for 30 seconds and centrifuge at 10,000 × g for 2 minutes before analysis [20] [9].
Critical Parameters and Method Optimization
Optimized SPE Conditions
Table 1 summarizes the optimized parameters for SPE of metoprolol.
Table 1: Optimized SPE Parameters for Metoprolol Extraction
| Parameter | Optimized Condition | Alternative Options | Impact on Performance |
|---|---|---|---|
| Sorbent Type | Mixed-mode cationic exchange (MCX) | Oasis PRiME HLB, C18 | MCX provides superior cleanup for basic compounds [20] |
| Sorbent Mass | 30 mg | 60 mg for complex matrices | 30 mg provides adequate capacity with minimal solvent [20] |
| Conditioning Solvent | 1 mL methanol, then 1 mL acidified water (pH 2-3) | Acetonitrile as alternative organic solvent | Ensures proper sorbent activation and compatibility [9] |
| Sample Loading pH | 2-3 | --- | Ensures protonation of amine for strong cation exchange [20] |
| Loading Flow Rate | 0.5-1 mL/min | Up to 2 mL/min with recovery verification | Slower rates improve retention efficiency [28] [9] |
| Wash Solvent | 1 mL 2% formic acid, then 1 mL methanol | Water with 1-5% organic modifier | Removes interferences while retaining analyte [20] |
| Elution Solvent | 1 mL 5% NH₄OH in methanol | 2-10% NH₄OH in methanol, or acetonitrile | Alkaline organic solvent disrupts ionic and hydrophobic interactions [20] |
| Elution Flow Rate | 0.5-1 mL/min | Up to 1 mL/min with collection verification | Controlled flow ensures complete elution [9] |
Method Optimization Data
Table 2 presents quantitative optimization data for metoprolol SPE.
Table 2: Method Optimization and Performance Data
| Optimization Variable | Tested Range | Optimal Value | Recovery at Optimal Value |
|---|---|---|---|
| Sorbent Mass (MCX) | 10-60 mg | 30 mg | 98.2% [20] |
| Sample Loading pH | 1-10 | 2-3 | >95% [20] |
| Loading Flow Rate | 0.5-2 mL/min | 0.5-1 mL/min | >90% [28] |
| Eluent Volume | 0.5-2 mL | 1 mL | >95% [20] |
| Ion Strength (NaCl) | 0-5% | 0% | Prevents disruption of interactions [28] |
| Wash Solvent Strength | 0-50% MeOH in water | 100% MeOH after aqueous wash | <5% analyte loss in wash [20] |
Experimental Workflow
The following workflow diagram illustrates the complete SPE procedure for metoprolol extraction:
Research Reagent Solutions
Table 3 lists essential research reagents and materials for implementing this SPE protocol.
Table 3: Essential Research Reagents and Materials
| Reagent/Material | Function/Purpose | Specifications/Alternatives |
|---|---|---|
| Mixed-mode Cationic Exchange (MCX) Cartridges | Primary sorbent for simultaneous reversed-phase and cation-exchange retention | 30 mg/1 mL bed volume; Oasis MCX recommended [20] |
| Methanol (HPLC Grade) | Conditioning solvent and elution solvent component | High purity, low UV absorbance; acetonitrile as alternative [9] |
| Formic Acid (≥95%) | Sample acidification and wash solvent component | Maintains low pH for amine protonation; acetic acid as milder alternative [20] |
| Ammonium Hydroxide (28-30% NH₃) | Elution solvent alkalizer | Disrupts ionic interactions; triethylamine as alternative [20] |
| Metoprolol Tartrate Standard | Reference standard for quantification and method validation | Pharmaceutical secondary standard; ≥99% purity [19] |
| Internal Standard (e.g., Bisoprolol) | Normalization of extraction variability | Structurally similar β-blocker; isotopically labeled metoprolol ideal for MS [8] |
Analytical Method Integration
For analysis following SPE, liquid chromatography coupled to tandem mass spectrometry (LC-MS/MS) provides optimal sensitivity and specificity. The recommended chromatographic conditions include:
- Column: C18 column (50 × 2.1 mm, 1.9 µm)
- Mobile Phase: Water and acetonitrile, both containing 0.1% formic acid
- Ionization: Positive electrospray ionization (ESI+)
- Detection: Multiple reaction monitoring (MRM) with transition m/z 268.1 → 130.96 for metoprolol [8]
For laboratories without MS detection, HPLC with UV or fluorescence detection can be employed, though with potentially higher limits of quantification [19] [27].
Troubleshooting and Quality Control
Common issues and solutions in metoprolol SPE:
- Low Recovery: Verify elution solvent strength and volume. Ensure proper pH adjustment during sample loading. Check sorbent capacity for concentrated samples [9].
- Matrix Effects: Incorporate a thorough washing step after sample loading. Consider PRiME (Process, Robustness, Improvements, Matrix Effects, Ease of use) clean-up procedures to minimize phospholipid interference in LC-MS/MS analysis [20].
- Poor Reproducibility: Standardize flow rates across all steps. Prevent sorbent drying between conditioning and sample loading. Use internal standards to correct for extraction variability [28] [9].
For quality control, include method blanks, fortified samples, and reference materials in each batch to monitor extraction efficiency and minimize background interference.
This application note presents a comprehensively optimized SPE protocol for metoprolol extraction, with detailed guidance on critical parameters including sorbent mass, solvent volumes, and flow rates. The method leverages mixed-mode cationic exchange solid-phase extraction to achieve high recovery (>95%) and effective clean-up of metoprolol from complex matrices. The optimized conditions balance extraction efficiency with practical considerations for implementation in pharmaceutical analysis and clinical research settings. This protocol provides a robust foundation for metoprolol quantification in support of pharmaceutical development, bioequivalence studies, and therapeutic drug monitoring programs.
The optimization of chromatographic conditions is fundamental to developing robust analytical methods for pharmaceutical compounds. For ionizable molecules like metoprolol, a selective β1-adrenergic receptor blocker, mobile phase pH serves as a powerful tool to control retention, selectivity, and peak shape in reversed-phase liquid chromatography [30] [31]. Metoprolol, with a documented pKa of approximately 9.7, exists predominantly in its ionized state at typical analytical pH values, making its chromatographic behavior highly susceptible to subtle pH changes [29]. This application note details the strategic use of pH to optimize solid-phase extraction (SPE) cleanup and subsequent high-performance liquid chromatography (HPLC) analysis of metoprolol tartrate from tablet formulations, providing specific protocols for method development within a research context focused on analytical chemistry.
Theoretical Foundation: pH and Retention of Basic Compounds
In reversed-phase chromatography, the retention of ionizable analytes is governed by their ionization state, which is directly controlled by the mobile phase pH relative to the analyte's pKa.
The Henderson-Hasselbalch Relationship: For a basic compound like metoprolol, the equilibrium between its neutral (B) and protonated (BH⁺) forms is described by:
pH = pKa + log([B]/[BH⁺])At a pH 2 units above the pKa, the base is predominantly neutral and well-retained. Conversely, at a pH 2 units below the pKa, it is predominantly ionized and poorly retained [30]. Metoprolol's pKa of 9.7 indicates it will be largely positively charged across most common HPLC pH ranges [29].Retention Mechanism: The neutral form of metoprolol is hydrophobic and interacts strongly with the non-polar stationary phase (e.g., C18), leading to longer retention times. The protonated, charged form is more hydrophilic and has weaker interactions with the stationary phase, resulting in shorter retention times [30] [31]. Figure 1 illustrates this fundamental relationship between pH and retention for a basic analyte.
Selectivity and Peak Shape: Adjusting pH can significantly alter the selectivity of a separation when analyzing mixtures containing compounds with different pKa values [30]. Furthermore, operating at a pH where the analyte is fully ionized or fully neutral minimizes the presence of multiple species with different retention properties, which can lead to peak tailing or broadening. Optimal peak shape is achieved when the mobile phase pH is at least 1.5-2 pH units away from the analyte's pKa [30] [31].
Figure 1. The influence of mobile phase pH on the retention of a basic compound. At high pH, the neutral base is well-retained, while at low pH, the protonated ion is poorly retained.
Metoprolol-Specific Chemical Profile
A clear understanding of metoprolol's chemical and pharmacological properties informs the analytical strategy.
- Chemical Structure: Metoprolol, chemically known as 1-(isopropylamino)-3-[4-(2-methoxyethyl)phenoxy]propan-2-ol, is a small molecule with a molecular weight of 267.36 g/mol [32].
- pKa and Ionization: The pKa of its secondary amine functional group is 9.7 [29]. This is the critical parameter for pH-based method optimization.
- Pharmacological Relevance: Metoprolol is a cardioselective beta-1 blocker used to treat hypertension, angina, and heart failure. It is typically administered as a racemic mixture of R- and S-enantiomers, with the S-enantiomer possessing most of the beta-blocking activity [32] [33]. The analysis described herein focuses on the racemic mixture.
Experimental Protocols
Protocol 1: SPE Cleanup of Metoprolol from Tablet Formulations
This protocol utilizes mixed-mode cation exchange SPE to leverage metoprolol's basic properties for selective cleanup.
Principle: At a pH below its pKa, metoprolol is positively charged and can be retained on a cation exchange sorbent via electrostatic interactions, while neutral and anionic matrix components are washed away.
Materials:
- Oasis MCX (Mixed-mode Cation Exchange) or equivalent SPE cartridges (e.g., 60 mg, 3 mL)
- Vacuum manifold for SPE
- Metoprolol tartrate tablet formulations
- HPLC-grade methanol, acetonitrile, and water
- Ammonium hydroxide solution (e.g., 25%)
- Formic acid (e.g., 98-100%)
- Volumetric flasks, pipettes, and centrifuge tubes
Procedure:
- Sample Preparation: Crush and homogenize a representative number of tablets. Accurately weigh an amount equivalent to ~50 mg of metoprolol free base into a suitable container. Add 50 mL of a suitable solvent (e.g., water:methanol 70:30), vortex and sonicate for 15 minutes to extract the API. Centrifuge and dilute the supernatant as needed to fit the calibration curve.
- SPE Conditioning: Condition the MCX cartridge with 2 mL of methanol, followed by 2 mL of water. Do not allow the sorbent to dry.
- Sample Loading: Acidify the sample solution to a pH of 2-3 using dilute formic acid. Load an appropriate volume of the acidified sample onto the conditioned cartridge.
- Wash: Wash the cartridge with 2-3 mL of 2% (v/v) formic acid in water to remove neutral and acidic interferents. Optionally, wash with 1-2 mL of methanol to remove strongly retained neutral impurities.
- Elution: Elute metoprolol with 2 x 2 mL of 5% ammonium hydroxide in methanol. This basifies the sorbent, neutralizing metoprolol and breaking the ionic interaction.
- Reconstitution: Evaporate the eluent to dryness under a gentle stream of nitrogen or in a vacuum concentrator. Reconstitute the residue in the initial mobile phase to be used for HPLC analysis (e.g., 1 mL of 20:80 acetonitrile:buffer). Filter through a 0.22 μm syringe filter prior to injection.
Protocol 2: HPLC Method Development and pH Scouting
This protocol outlines a systematic approach to finding the optimal pH for the chromatographic separation of metoprolol.
Materials:
- HPLC system with UV or DAD detector (e.g., set to 220-275 nm)
- Reversed-phase column (e.g., C18, 150 x 4.6 mm, 3.5 μm)
- Mobile phase A: Aqueous buffer (e.g., 25-50 mM ammonium formate or phosphate)
- Mobile phase B: Acetonitrile (HPLC grade)
- Formic acid, ammonium hydroxide, or phosphoric acid for pH adjustment
Procedure:
- Buffer Preparation: Prepare three separate batches of Mobile Phase A at different pH values: 3.0, 7.0, and 10.0. Use phosphate buffer for pH 3.0 and 7.0, and ammonium bicarbonate/formate for pH 10.0, ensuring the pH is compatible with the column's specifications. Adjust pH with dilute acids/bases after buffer preparation. Mix each with Mobile Phase B (acetonitrile) as needed for the gradient or isocratic method.
- Chromatographic Conditions:
- Column Temperature: 35-40 °C
- Flow Rate: 1.0 mL/min
- Detection: UV 225 nm
- Injection: Isocratic elution with 25:75 buffer:acetonitrile or a shallow gradient (e.g., 10-50% B in 10 min).
- pH Scouting Analysis: Inject a standard solution of metoprolol (and any potential impurities/degradants if available) using each of the three mobile phase pH conditions. Keep all other chromatographic parameters constant.
- Data Analysis: Record the retention time, peak area, and peak asymmetry factor for metoprolol at each pH. The results will guide the selection of the optimal pH for the final method, balancing retention, peak shape, and selectivity from other components.
Data Presentation and Analysis
Predicted Retention Behavior of Metoprolol
The data below, derived from general principles of chromatographic behavior, summarizes the expected impact of pH on metoprolol's retention and peak shape [30] [31] [29].
Table 1: Theoretical impact of mobile phase pH on metoprolol chromatography.
| Mobile Phase pH | Ionization State (pKa ~9.7) | Expected Retention (k) | Expected Peak Shape | Recommended for Analysis? |
|---|---|---|---|---|
| 3.0 | >99% Ionized (BH⁺) | Very Low | Good | Yes (but may co-elute) |
| 7.0 | >99% Ionized (BH⁺) | Low | Good | Yes |
| 9.0 | ~80% Ionized | Medium | Potential Tailing | No (too close to pKa) |
| 10.5 | ~85% Neutral (B) | High | Good | Yes (if column stable) |
Experimental pH Scouting Results
The following table presents simulated data from a pH scouting experiment, illustrating the practical outcomes of the protocol in Section 4.2.
Table 2: Simulated HPLC results for metoprolol under different pH conditions (Isocratic: 75% 25mM Buffer, 25% ACN).
| Mobile Phase pH | Retention Time (min) | Retention Factor (k)* | Peak Asymmetry | Resolution from Impurity A |
|---|---|---|---|---|
| 3.0 | 2.5 | 1.1 | 1.1 | 1.5 |
| 7.0 | 3.2 | 1.8 | 1.1 | 2.2 |
| 10.5 | 9.5 | 7.3 | 1.2 | 4.5 |
*Retention Factor (k) = (Retention Time - Void Time) / Void Time; Void Time assumed to be 1.2 min.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key materials and reagents for SPE and HPLC analysis of metoprolol.
| Item Name & Supplier Example | Function in the Analysis |
|---|---|
| Oasis MCX SPE Cartridge (Waters) | Mixed-mode sorbent for selective retention of basic analytes like metoprolol via cation exchange and reversed-phase mechanisms. |
| Ammonium Hydroxide (e.g., 25%) (Sigma-Aldrich) | A volatile base used in the elution solvent to neutralize the analyte and sorbent, enabling efficient release of metoprolol from the SPE cartridge. |
| Ammonium Formate Buffer (Fluka) | Provides buffering capacity for the mobile phase at a wide pH range; it is volatile and compatible with mass spectrometric detection. |
| C18 HPLC Column (e.g., ZORBAX Eclipse Plus, Agilent) | Reversed-phase stationary phase for the chromatographic separation of metoprolol from its impurities and matrix components. |
| Formic Acid (Merck) | Used to acidify the sample and wash solvents in SPE to promote protonation of metoprolol; also used as a mobile phase additive to improve ionization in LC-MS. |
Integrated Workflow for Sample Analysis
The complete analytical procedure, from sample preparation to data analysis, is summarized in Figure 2. This workflow integrates the SPE cleanup and HPLC analysis protocols, highlighting the critical role of pH control at each stage.
Figure 2. Integrated analytical workflow for metoprolol from tablet preparation to quantification, emphasizing critical pH-controlled steps.
Troubleshooting and Optimization Guidelines
- Poor Recovery in SPE: Ensure the sample is adequately acidified before loading onto the MCX cartridge. The pH must be at least 2 units below the pKa (i.e., <7.7) to ensure >99% ionization. Using a stronger elution solvent or ensuring the eluent is sufficiently basic can also improve recovery.
- Peak Tailing in HPLC: This is often observed when the mobile phase pH is too close to the analyte's pKa. Shift the pH to at least 1.5-2 units away from 9.7. Using a high-purity silica column with high surface coverage and endcapping can also mitigate tailing.
- Irreproducible Retention Times: Inconsistent buffer preparation is a common culprit. Precisely measure the pH of the mobile phase after the organic modifier is added, as this can alter the apparent pH. Use fresh, accurately prepared buffers.
- Method Robustness: For a highly robust method, operate at a pH where the ionization state of metoprolol is least sensitive to minor fluctuations. Based on its pKa profile, a pH of 7.0 provides a stable, ionized state, while a pH of 10.5 (if column stability allows) provides a stable, neutral state [30]. The region between ~8.7 and ~10.7 should be avoided for a final method due to high sensitivity to minor pH changes.
Solid-phase extraction (SPE) is a critical sample preparation technique in the bioanalysis of metoprolol tartrate, a selective β1-adrenergic receptor antagonist used to treat cardiovascular conditions like hypertension and heart failure [20]. The primary goal of SPE is to selectively isolate the analyte from the complex biological matrix, thereby concentrating it and reducing interfering substances that can adversely affect the subsequent chromatographic analysis. For metoprolol, which is typically administered as a racemic mixture but with its (S)-enantiomer possessing the primary pharmacological activity, a well-optimized SPE protocol is not just a purification step but a fundamental requirement for accurate pharmacokinetic and pharmacodynamic profiling [20]. The efficiency of this process hinges on the judicious selection of the elution solvent, a choice that directly dictates the balance between maximizing analyte recovery and minimizing co-elution of matrix interferents, particularly phospholipids.
Matrix effects (ME) represent a significant challenge in bioanalysis, especially when using sensitive detection techniques like liquid chromatography-tandem mass spectrometry (LC-MS/MS). These effects cause ion suppression or enhancement, impacting the reproducibility, accuracy, and detection capability of the method [34]. In the context of metoprolol analysis, matrix effects originate from endogenous phospholipids present in human plasma, which can co-elute with the analyte and suppress its ionization signal in the electrospray ion source [20]. Nearly 80% of metoprolol metabolism is mediated by the hepatic cytochrome P450 (CYP) 2D6 enzyme, leading to active metabolites like (S)-α-hydroxymetoprolol, which also need to be accurately quantified [20]. The selection of an optimal elution solvent is, therefore, a critical multivariate optimization problem that aims to achieve the strongest possible elution strength for the target analyte while retaining the majority of the matrix interferences on the sorbent.
Core Principles of Elution Solvent Selection
The fundamental principle guiding elution solvent selection is achieving a higher affinity for the analyte with the solvent than with the sorbent. Metoprolol is an alkaline compound with a pKa of approximately 9.7 and contains a secondary amine functional group, making it particularly suitable for retention on mixed-mode cationic sorbents [20]. These sorbents combine reversed-phase and ion-exchange mechanisms, offering superior selectivity for basic drugs like metoprolol compared to generic reversed-phase sorbents.
The elution process must overcome both these interactions. The solvent strength is primarily responsible for disrupting the reversed-phase (hydrophobic) interactions, while the elution pH is crucial for neutralizing the ion-exchange (electrostatic) interactions. For the mixed-mode cationic extraction of metoprolol, the optimal approach involves using an organic solvent with high elution strength, such as acetonitrile or methanol, that is acidified to protonate the sorbent's functional groups and shift the equilibrium towards the mobile phase [20]. This dual strategy ensures that the strong ionic bonds are broken, allowing the analyte to be released efficiently.
Furthermore, the concept of selectivity is paramount. An ideal elution solvent should have a high eluotropic strength for the target analyte but a low strength for the major phospholipid classes and other endogenous compounds. Phospholipids are a primary source of matrix effects in plasma analysis and can significantly suppress the electrospray ionization signal of metoprolol [20]. The advent of specialized SPE procedures, such as Phospholipid Removal Microelution (PRM)-SPE, has demonstrated that it is possible to virtually cancel phospholipid interference through careful sorbent and solvent selection [20]. The solvent must also be compatible with the subsequent LC-MS/MS analysis to avoid peak broadening or signal instability and should be volatile to facilitate post-extraction concentration if necessary.
Quantitative Data and Solvent Performance
The performance of different elution solvent systems can be quantitatively evaluated based on key bioanalytical validation parameters, primarily recovery (a measure of extraction efficiency) and the matrix effect. The following table summarizes experimental data for metoprolol (MET) and its metabolite, α-hydroxymetoprolol (OH-MET), obtained using a mixed-mode, cationic PRM-SPE approach coupled with LC-MS/MS detection [20].
Table 1: Performance Metrics for Elution Solvents in MET and OH-MET SPE
| Elution Solvent Composition | Analyte | Recovery (%) | Matrix Effect (%) | Key Findings |
|---|---|---|---|---|
| Acidified Methanol | (S)-MET | >85% | Not Specified | Lower recovery compared to acidified acetonitrile in a 3-step SPE method [20]. |
| (e.g., with 2% Formic Acid) | (S)-α-OH-MET | >85% | Not Specified | |
| Acidified Acetonitrile | (S)-MET | >95% | Minimized | Superior elution strength; achieved high recovery and minimal ME in a 4-step PRM-SPE method [20]. |
| (e.g., with 2% Formic Acid) | (S)-α-OH-MET | >95% | Minimized | |
| ACN - 0.1% Formic Acid (50:50) | MET | >89% | 89% | Used in an automated TurboFlow LC-MS/MS method; provided a robust and precise approach [8]. |
The data indicates that acidified acetonitrile generally provides superior performance for eluting metoprolol and its metabolite from mixed-mode cationic sorbents, achieving recoveries greater than 95% while effectively minimizing matrix effects [20]. The high elution strength of acetonitrile, combined with the ionic suppression provided by the acid additive, makes it a preferred choice. Furthermore, an automated online-SPE method using a mobile phase of acetonitrile with 0.1% formic acid demonstrated a matrix effect of 89%, which was considered acceptable, and allowed for a wide linear range of 5 to 1000 ng/L [8].
Detailed Experimental Protocols
Protocol: Mixed-Mode Cationic PRM-SPE for Human Plasma
This protocol is designed for the simultaneous extraction of (S)-metoprolol and (S)-α-hydroxymetoprolol from human plasma, leveraging a 4-step mixed-mode cationic PRM-SPE procedure to maximize recovery and minimize phospholipid-based matrix effects [20].
The Scientist's Toolkit: Research Reagent Solutions
- Mixed-Mode Cationic PRM-SPE Cartridges: Contains a sorbent with both reversed-phase and strong cation-exchange (SCX) functionalities, essential for the selective retention of basic drugs like metoprolol.
- (S)-MET-(d7) and α-OH-MET-(d5): Deuterated internal standards; correct for analyte loss during preparation and quantify matrix effects.
- Methyl-tert-butyl ether (MTBE): A low-polarity solvent used in the wash step to remove hydrophobic interferences without eluting the analyte.
- Acetonitrile (HPLC Grade): The primary organic solvent for elution, chosen for its high elution strength and low UV cutoff.
- Formic Acid (≥95%): Used as an additive to acidify the elution solvent, protonating the sorbent and neutralizing ionic interactions.
- Ammonium Hydroxide (28-30% NH₃): Used to make the conditioning and loading solutions alkaline, ensuring metoprolol is in its neutral form for optimal sorbent interaction.
Step-by-Step Procedure:
- Conditioning: Sequentially load the PRM-SPE cartridge with 500 μL of methanol, followed by 500 μL of water. Do not allow the sorbent to dry out.
- Sample Loading: Acidify the plasma sample with an equal volume of 0.1% formic acid in water. Load the acidified sample onto the conditioned cartridge.
- Washing: Perform two wash steps to remove impurities:
- Wash 1: 500 μL of 2% (v/v) formic acid in water. This removes polar interferences and proteins.
- Wash 2: 500 μL of methyl-tert-butyl ether (MTBE). This removes lipophilic interferences and phospholipids, which is critical for minimizing matrix effects.
- Elution: Elute the analytes with 2 × 500 μL of a freshly prepared solution of 2% formic acid in acetonitrile. Collect the entire eluate in a clean tube.
- Reconstitution: Evaporate the eluate to complete dryness under a gentle stream of nitrogen at 40°C. Reconstitute the dry residue with 100 μL of the initial mobile phase used for the LC-MS/MS analysis (e.g., a water/acetonitrile mixture). Vortex thoroughly and centrifuge before injection.
Protocol: Automated Online SPE (TurboFlow) for Metoprolol
This protocol utilizes an automated dual-column system (TurboFlow and analytical) for online sample clean-up and quantification, enhancing throughput and reproducibility [8].
Step-by-Step Procedure:
- Sample Preparation: Dilute the plasma sample with an internal standard solution (e.g., bisoprolol fumarate). After vortexing, centrifuge the samples.
- System Setup: Configure the LC system with a TurboFlow Cyclone-P column (for extraction) and a C18 analytical column (for separation). The system should include two pumps: one for loading and one for elution/analysis.
- Online Extraction:
- Step 1 (Load & Concentrate): Inject 100 μL of the prepared sample onto the TurboFlow column using a loading pump with 0.1% formic acid in water at 1.5 mL/min. Analytes are retained while matrix components are washed to waste.
- Step 2 (Elute to Analytical Column): Switch the valve to divert the flow. Using the elution pump, transfer the analytes from the TurboFlow column to the analytical column with a gradient of 40% water and 60% methanol (both with 0.1% formic acid).
- Chromatographic Separation & Column Re-equilibration:
- Step 3 (Separate): Perform an isocratic separation on the C18 analytical column with a mobile phase of 0.1% formic acid in water and acetonitrile (50:50, v/v) at a flow rate of 0.5 mL/min.
- Simultaneously, recondition the TurboFlow column with 100% methanol.
- Step 4 (Re-equilibrate): Re-equilibrate the entire system to initial conditions before the next injection.
Visualization of the SPE Workflow and Solvent Selection Logic
The following diagram illustrates the optimized 4-step SPE workflow for the manual mixed-mode extraction of metoprolol, highlighting the role of each solvent.
Diagram 1: Optimized 4-Step Mixed-Mode SPE Workflow for Metoprolol.
The logic behind selecting the final elution solvent is a systematic process, as detailed in the following decision pathway.
Diagram 2: Logic Pathway for Optimal Elution Solvent Selection.
The selection of an elution solvent in solid-phase extraction is a decisive factor in developing a robust and reliable bioanalytical method for metoprolol tartrate. The evidence consistently demonstrates that acidified acetonitrile (e.g., with 2% formic acid) serves as the superior choice for mixed-mode cationic SPE, successfully balancing the dual objectives of high analyte recovery (>95%) and minimal matrix effect. The implementation of specialized protocols, such as the 4-step PRM-SPE or automated TurboFlow systems, provides a clear pathway to achieve this balance. By systematically applying the principles and protocols outlined in this application note, researchers and drug development professionals can significantly enhance the data quality of their pharmacokinetic and clinical studies involving metoprolol, ensuring that results are both accurate and precise.
Solid-phase extraction (SPE) serves as a critical sample preparation step in pharmaceutical analysis, enabling the cleanup, pre-concentration, and purification of target analytes from complex matrices. This integration is particularly valuable in drug development and quality control, where precise and accurate quantification is essential. The selection of an appropriate analytical detection technique—whether HPLC-UV or LC-MS/MS—depends on multiple factors including required sensitivity, specificity, and available instrumentation [35] [36]. This application note details protocols for coupling SPE with both HPLC-UV and LC-MS/MS methodologies, framed within research on metoprolol tartrate tablet analysis.
Technical Background: SPE as a Sample Preparation Tool
SPE utilizes a cartridge or disk containing a chromatographic stationary phase to selectively retain and purify analytes from a liquid sample. This process effectively removes interfering matrix components that could compromise analytical results, while often concentrating the analyte to enhance detection sensitivity [37] [36]. For pharmaceutical analysis in complex biological or formulation matrices, this cleanup step is crucial for achieving reliable quantification.
The combination of SPE with chromatographic techniques addresses key challenges in analytical chemistry:
- Matrix Effect Reduction: Removes proteins and phospholipids that can cause ion suppression in mass spectrometry [36].
- Analyte Enrichment: Improves method sensitivity by concentrating the target analyte [38].
- Chromatographic Performance: Protects the analytical column from contamination and improves peak shape [36].
- Method Robustness: Extends instrument uptime by reducing maintenance requirements [36].
Comparative Technique Selection: HPLC-UV vs. LC-MS/MS
Technique Comparison and Applications
Table 1: Comparison of HPLC-UV and LC-MS/MS Techniques Coupled with SPE
| Parameter | HPLC-UV | LC-MS/MS |
|---|---|---|
| Sensitivity | Moderate (ng to µg range) [35] | High (pg to ng range) [38] |
| Selectivity | Good (based on retention time and UV spectrum) [39] | Excellent (based on mass/charge ratio and fragmentation) [38] |
| Analytical Scope | Targeted analysis, single or few compounds [35] | Multi-analyte methods, complex profiles [38] |
| Equipment Cost | Lower [35] | Higher [35] |
| Operational Complexity | Lower, more accessible [35] | Higher, requires specialized expertise [35] [36] |
| Ideal Application | Routine quality control, therapeutic drug monitoring [35] | Complex matrices, trace analysis, metabolite identification [38] [36] |
Selection Criteria for Metoprolol Tartrate Analysis
For metoprolol tartrate analysis, technique selection depends on the specific research context:
- HPLC-UV is suitable for formulation quality control, dissolution testing, and purity assessment where analyte concentrations are relatively high and matrix effects are manageable [40].
- LC-MS/MS is preferred for biological fluid analysis (pharmacokinetics), metabolite identification, and trace impurity profiling where superior sensitivity and specificity are required [36].
Experimental Protocols
General SPE Procedure for Metoprolol Tartrate
Table 2: Key Research Reagent Solutions for SPE-HPLC-UV Analysis of Metoprolol Tartrate
| Reagent/Material | Function | Specifications/Alternatives |
|---|---|---|
| C18 SPE Cartridge | Primary extraction sorbent | 500 mg, 6 mL capacity [35] |
| Methanol (HPLC grade) | Sorbent conditioning | Maintains sorbent wettability [37] |
| Water (HPLC grade) | Sorbent equilibration | Creates optimal environment for analyte retention [35] |
| Sample Diluent | Matrix modification | Dilute phosphate buffer (pH ~7) [40] |
| Wash Solution | Remove interferents | 5-10% methanol in water [35] |
| Elution Solvent | Analyte recovery | Methanol or acetonitrile [35] [37] |
| Internal Standard | Quantification control | Structural analog with similar properties [35] |
The following protocol describes SPE sample preparation for metoprolol tartrate from tablet formulations or biological matrices:
SPE Cartridge Conditioning: Condition the C18 cartridge (500 mg, 6 mL) with 5 mL methanol followed by 5 mL deionized water [35]. Do not allow the sorbent bed to dry completely.
Sample Preparation: For tablet analysis, dissolve and dilute powdered tablet in appropriate solvent (e.g., mobile phase or buffer). For biological samples, add internal standard and dilute with buffer (pH adjusted to optimize retention) [36].
Sample Loading: Load the prepared sample onto the conditioned SPE cartridge at a controlled flow rate (1-2 mL/min).
Wash Step: Wash with 5 mL of 5% methanol in water to remove interfering compounds [35].
Elution: Elute metoprolol tartrate with 2 × 2.5 mL of methanol into a clean collection tube [35].
Reconstitution: Evaporate the eluent under nitrogen stream and reconstitute in 500 µL of HPLC mobile phase [36].
HPLC-UV Analysis of Metoprolol Tartrate
Building on validated methods for pharmaceutical compounds [35], the following HPLC-UV conditions are recommended:
- Column: C18 reverse-phase (4.6 mm × 150 mm, 3.5 µm) [35]
- Mobile Phase: Acetonitrile:phosphate buffer (pH 3.0) (30:70, v/v) [40]
- Flow Rate: 1.0 mL/min [35]
- Detection: UV at 280 nm [40]
- Injection Volume: 20 µL [35]
- Column Temperature: 35°C [35]
- Run Time: 15-20 minutes
LC-MS/MS Analysis of Metoprolol Tartrate
For enhanced sensitivity, adapt methods from recent SPE-LC-MS/MS applications [38]:
- Column: C18 reverse-phase (2.1 × 50 mm, 1.8 µm) [38]
- Mobile Phase: A: 0.1% formic acid in water; B: 0.1% formic acid in acetonitrile [38]
- Gradient: 5% B to 95% B over 5 minutes [38]
- Flow Rate: 0.3 mL/min [38]
- Ionization: ESI positive mode [38]
- MS Transitions: Monitor metoprolol precursor ion → product ion transitions [36]
Diagram 1: Integrated SPE-HPLC-UV/LC-MS/MS Workflow for Metoprolol Tartrate Analysis. The workflow begins with sample preparation and proceeds through SPE cleanup before branching based on detection requirements.
Method Validation Parameters
When implementing these integrated methods, key validation parameters should be established:
Table 3: Method Validation Parameters for SPE-HPLC-UV and SPE-LC-MS/MS Methods of Metoprolol Tartrate
| Validation Parameter | Acceptance Criteria | Application Notes |
|---|---|---|
| Linearity | r² > 0.99 [35] | Optimize according to expected concentration range [35] |
| Accuracy | ±15% of nominal concentration [35] | Use QC samples at low, medium, high concentrations [35] |
| Precision | RSD < 15% [35] | Evaluate intra-day and inter-day variability [35] |
| Recovery | Consistent and reproducible [35] | Compare extracted vs. non-extracted standards [35] |
| Limit of Quantification | Sufficient for application [40] | LC-MS/MS offers lower LOQ than HPLC-UV [40] [38] |
| Specificity | No interference from matrix [36] | LC-MS/MS provides superior specificity [36] |
Troubleshooting and Optimization
Common challenges in SPE-HPLC-UV and SPE-LC-MS/MS integration include:
- Poor Recovery: Optimize sorbent chemistry, adjust sample pH, modify elution solvent [37].
- Matrix Effects: Incorporate effective cleanup, use stable isotope-labeled internal standards [36].
- Chromatographic Issues: Adjust mobile phase composition, column temperature, or gradient program [35].
- Signal Suppression (MS): Improve sample cleanup, modify ionization parameters [36].
Response surface methodology (RSM) can systematically optimize SPE parameters for maximal recovery and minimal matrix effects [41].
The integration of SPE with either HPLC-UV or LC-MS/MS provides powerful analytical tools for metoprolol tartrate research. HPLC-UV provides a cost-effective solution for routine quality control, while LC-MS/MS offers superior sensitivity and selectivity for challenging applications. Proper method selection, optimization, and validation ensure reliable performance across various research scenarios in pharmaceutical analysis.
Solving Common SPE Problems for Metoprolol Analysis: Low Recovery, Reproducibility, and Cleanup Issues
In the analysis of active pharmaceutical ingredients (APIs) such as metoprolol tartrate from tablet formulations, solid-phase extraction (SPE) is a critical sample preparation technique used to isolate the analyte from complex excipient matrices. Achieving high analyte recovery is paramount for obtaining accurate and reproducible results in downstream analysis. Two of the most prevalent causes of low recovery are sorbent polarity mismatch and insufficient elution strength. This application note provides a structured diagnostic guide and detailed protocols to help researchers systematically identify and resolve these issues, with a specific focus on metoprolol tartrate analysis.
Diagnostic Framework for Low Recovery
The following workflow provides a systematic approach for diagnosing the root causes of low SPE recovery. Adhering to this logical pathway can significantly reduce troubleshooting time.
Understanding and Resolving Sorbent Polarity Mismatch
The Retention Mechanism
Selecting a sorbent with an appropriate retention mechanism for the target analyte is the most critical step in SPE method development. A mismatch here will inevitably lead to poor recovery.
Table 1: SPE Sorbent Selection Guide Based on Analyte and Matrix Properties [42] [43]
| Sorbent Type | Primary Retention Mechanism | Ideal Analyte Properties | Ideal Sample Matrix | Common Sorbent Chemistries |
|---|---|---|---|---|
| Reversed-Phase | Van der Waals (dispersive) forces | Nonpolar, hydrophobic | Aqueous, polar | C18, C8, CN, Phenyl, Polymer-based HLB |
| Normal-Phase | Dipole-dipole, Hydrogen bonding | Polar | Nonpolar organic | Unbonded silica, Diol, Cyano, Amino |
| Cation Exchange | Electrostatic attraction (negative sorbent) | Basic/positively charged (at operating pH) | Aqueous, low ionic strength | SCX, WCX, Carboxylic acid |
| Anion Exchange | Electrostatic attraction (positive sorbent) | Acidic/negatively charged (at operating pH) | Aqueous, low ionic strength | SAX, WAX, Quaternary amine |
Application to Metoprolol Tartrate: Metoprolol is a basic compound containing a secondary amine functional group. Its pKa is approximately 9.7, meaning it will be positively charged in most aqueous solutions with a pH below 8.7 [19]. This property makes it an excellent candidate for retention via a cation-exchange mechanism (e.g., using a strong cation exchanger, SCX). It can also be retained on reversed-phase sorbents via hydrophobic interactions, but this may require pH adjustment to suppress ionization for optimal retention [42] [43].
Diagnostic Protocol: Identifying Polarity Mismatch
Objective: To confirm that the selected sorbent has the appropriate chemistry to retain metoprolol tartrate under the chosen loading conditions.
Materials:
- WelchClean SPE cartridges (e.g., C18 for reversed-phase, SCX for cation-exchange)
- Standard solution of metoprolol tartrate in water (e.g., 0.2 mg/mL) [19]
- Appropriate buffers (e.g., Britton-Robinson buffer, pH 6.0 [19])
- HPLC system with UV detector
Method:
- Conditioning and Equilibration: Condition the test sorbent (e.g., 100 mg/mL) with 2 bed volumes of methanol, followed by 2 bed volumes of water or a buffer compatible with the retention mechanism [44] [45].
- Sample Application: Load a known amount of the metoprolol standard (e.g., 1 mL of 0.2 mg/mL solution) onto the cartridge. Collect the load-through fraction.
- Washing: Pass 2 bed volumes of water or a weak wash buffer (e.g., 5% methanol) through the cartridge. Collect the wash fraction.
- Analysis: Analyze the load-through and wash fractions using HPLC-UV for the presence of metoprolol.
Interpretation: If a significant peak for metoprolol is detected in the load-through or wash fractions, it indicates a retention failure, likely due to a sorbent polarity or chemistry mismatch [44].
Resolution: Selecting the Correct Sorbent
If a mismatch is diagnosed, refer to Table 1 to choose a more appropriate sorbent. For metoprolol tartrate:
- For reversed-phase SPE, ensure the sample is loaded in a predominantly aqueous environment. The pH can be adjusted to ~2 units above its pKa (to ~11.7) to neutralize the compound and enhance hydrophobic retention, though this is often impractical [42] [43].
- For cation-exchange SPE, ensure the sample is loaded at a pH at least 2 units below the pKa (i.e., pH < 7.7) to guarantee the analyte is fully protonated and positively charged. A strong cation-exchange (SCX) sorbent is often the most selective choice for metoprolol [42].
Addressing Insufficient Elution Strength and Volume
The Elution Principle
Elution requires a solvent that can effectively disrupt the specific analyte-sorbent interactions established during the retention phase. Using a solvent that is too weak, or an insufficient volume, will result in incomplete analyte recovery.
Table 2: Elution Solvent Guide for Different Sorbent Chemistries [44] [42] [45]
| Sorbent Type | Analyte-Sorbent Interaction | Recommended Elution Solvents | Mechanism of Elution | Considerations for Metoprolol |
|---|---|---|---|---|
| Reversed-Phase | Hydrophobic | Methanol, Acetonitrile, Isopropanol, Tetrahydrofuran | Disrupts hydrophobic interactions by being less polar than water. | Sufficient organic strength (e.g., ≥80% methanol) is typically needed. |
| Cation Exchange | Ionic/Electrostatic | 1. pH Control: Buffer pH ≥2 above analyte pKa (≥11.7).2. High Ionic Strength: Buffer with high salt concentration.3. Counterions: Use buffers with counterions (e.g., K⁺). | Neutralizes the analyte or sorbent charge; competing ions displace analyte. | A basic eluent (e.g., ammoniated methanol) is highly effective as it neutralizes the charged amine. |
| Mixed-Mode | Hydrophobic & Ionic | Mixtures of organic solvent with acid, base, or salt. | Must disrupt both hydrophobic and ionic interactions simultaneously. | A mixture of, e.g., 80:20 Dichloromethane:Isopropanol with 2% Ammonium Hydroxide is common. |
Diagnostic Protocol: Evaluating Elution Efficiency
Objective: To determine if the current elution solvent and volume are sufficient to completely desorb metoprolol tartrate from the sorbent.
Materials:
- Conditioned SPE cartridge pre-loaded with metoprolol tartrate
- Proposed elution solvent(s) (see Table 2)
- Collection tubes
Method:
- Retention: Load a known quantity of metoprolol standard onto a properly conditioned and matched sorbent. Wash with a weak solvent to remove impurities.
- Fractionated Elution: Elute the analyte using your primary elution solvent, but collect the eluate as multiple sequential fractions (e.g., 1 mL per fraction).
- Analysis: Evaporate each fraction under a gentle stream of nitrogen (avoid excessive heat, e.g., ≤50°C, to prevent degradation [46]), reconstitute in mobile phase, and analyze via HPLC-UV.
Interpretation: Plot the amount of metoprolol found in each fraction. If the analyte is found predominantly in the first 1-2 fractions, the elution volume is likely sufficient. If it is spread over many fractions or is still present in significant amounts in later fractions, the elution strength is too weak. If a substantial amount of analyte is detected in a second elution with a stronger solvent, the primary elution volume or strength was insufficient [44] [45].
Resolution: Optimizing the Elution Step
- Increase Strength: For reversed-phase, increase the organic percentage or switch to a stronger solvent (e.g., from methanol to isopropanol). For ion-exchange, adjust the pH to neutralize the analyte (for metoprolol, this requires a basic pH >11) or increase the ionic strength of the eluent [44] [42].
- Increase Volume: Systematically increase the volume of the elution solvent in 0.5-1 mL increments while monitoring recovery until it plateaus [45].
- Add a Soak Step: Introduce a 1-5 minute soak period after adding the elution solvent to the cartridge. This allows time for the solvent to penetrate the sorbent pores and fully disrupt analyte-sorbent interactions, greatly improving recovery, especially for ion-exchange mechanisms [46] [43].
- Control Flow Rate: A slow, controlled elution flow rate (e.g., 1 mL/min or even slower for ion-exchange) improves recovery by allowing sufficient interaction time between the eluent and the retained analyte [43].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Reagents and Materials for SPE of Metoprolol Tartrate [44] [19] [42]
| Item | Function / Application | Specific Examples / Notes |
|---|---|---|
| C18 SPE Cartridges | Reversed-phase retention of metoprolol via hydrophobic interactions. | WelchClean C18 (100 mg/1mL). Capacity: ~5% of sorbent mass (e.g., 5 mg for 100 mg cartridge) [44]. |
| SCX SPE Cartridges | Cation-exchange retention of protonated metoprolol; offers high selectivity. | Strong Cation Exchanger (e.g., benzenesulfonic acid). Capacity: ~0.25-1.0 mmol/g [44]. |
| Methanol (HPLC Grade) | Universal conditioning and elution solvent for reversed-phase; component of eluent for ion-exchange. | Ensure high purity to prevent introduction of contaminants [42] [45]. |
| Ammonium Hydroxide | Used to create a basic eluent for ion-exchange and mixed-mode SPE to neutralize charged basic analytes. | Typically used as a 2-5% additive in organic eluents [42] [43]. |
| Britton-Robinson Buffer | A universal buffer for precise pH control during complex formation or SPE conditioning/loading. | Used at pH 6.0 for complexation of metoprolol with Cu(II) [19]; can be adapted for SPE pH control. |
| Ammonium Acetate Buffer | Provides buffering and ionic strength for conditioning and washing in ion-exchange SPE. | Useful for maintaining a stable pH during loading and washing steps. |
| SPE Vacuum Manifold | Provides controlled flow and processing of multiple samples simultaneously. | Welch SPE Manifold. Enforces consistent flow rates across samples [44]. |
Integrated Experimental Protocol for Metoprolol Tartrate SPE Cleanup
This protocol assumes the use of a cation-exchange sorbent (SCX), which is highly selective for metoprolol.
Workflow Overview:
Detailed Steps:
- Conditioning: Pass 2 mL of methanol through a 100 mg/1mL SCX cartridge at a flow rate of ~1-2 mL/min. Follow with 2 mL of deionized water. Do not let the sorbent bed run dry. [44] [45].
- Equilibration: Pass 2 x 1 mL of a weak aqueous buffer (e.g., 20 mM ammonium acetate, pH ~6.0) through the cartridge. This sets the pH for optimal retention of the protonated metoprolol.
- Sample Loading:
- Tablet Preparation: Weigh and powder ten tablets. Transfer a portion equivalent to 40 mg of metoprolol tartrate to a conical flask. Extract with 4 x 20 mL of water, filter into a 100 mL volumetric flask, and make up to volume with water [19].
- Load: Adjust the pH of an aliquot of the sample solution to ~6.0. Load this onto the conditioned SPE cartridge at a controlled flow rate not exceeding 2 mL/min [44] [43].
- Washing: Pass 2 x 1 mL of a wash solution (e.g., water or 5% methanol in water) through the cartridge to remove weakly retained matrix interferences. The flow rate can be increased to ~5 mL/min [46].
- Elution:
- Place a collection tube under the cartridge.
- Add 2 mL of a freshly prepared elution solvent (e.g., Methanol with 2% Ammonium Hydroxide) to the cartridge.
- Soak: Allow the solvent to soak the sorbent bed for 2-5 minutes [46] [43].
- Elute: Slowly pass the solvent through the cartridge at a flow rate of ~0.5-1 mL/min and collect the eluate.
- Repeat the elution with a second 2 mL portion of solvent and combine the eluates.
- Post-Processing: Evaporate the combined eluates to dryness under a gentle stream of nitrogen at a moderate temperature (e.g., 40°C). Reconstitute the residue in a suitable HPLC mobile phase (e.g., water or buffer) for analysis [46].
By meticulously following this diagnostic framework and the accompanying protocols, researchers can effectively troubleshoot and optimize SPE methods to achieve high recovery and robust analysis of metoprolol tartrate in pharmaceutical dosage forms.
Solid-phase extraction (SPE) is a critical sample preparation technique in pharmaceutical analysis, yet poor reproducibility in recovery often compromises data reliability. This application note systematically addresses two predominant sources of variability—inconsistent flow rates and improper cartridge drying—within the context of SPE cleanup for metoprolol tartrate tablet analysis. We provide optimized, detailed protocols to help researchers and drug development professionals achieve highly consistent and reproducible results, thereby enhancing the quality and reliability of analytical data.
In the analysis of active pharmaceutical ingredients such as metoprolol tartrate from tablet formulations, SPE provides essential sample cleanup and analyte concentration. However, the technique is susceptible to reproducibility issues that can directly impact method validation and result interpretation. Poor reproducibility manifests as high variability between replicate extractions, often traced to two critical procedural parameters: flow rate during sample loading/washing and completeness of cartridge drying prior to elution [44] [47]. Uncontrolled flow rates lead to inconsistent analyte-sorbent interaction times, while residual water in the sorbent bed after washing can cause inefficient elution and introduce water into final extracts, adversely affecting downstream analysis [48] [49]. This note delineates the causes of these issues and provides optimized, actionable protocols to mitigate them.
Critical Parameters and Their Impact on Reproducibility
Flow Rate Control
The flow rate during sample loading and washing is paramount for achieving equilibrium between the analyte and the sorbent. Excessive flow rates reduce contact time, leading to incomplete retention during loading or premature elution during washing [50] [44]. This is particularly critical for metoprolol, a compound whose retention is highly dependent on pH and ionic interactions.
- Consequence of High Flow Rate: When the flow rate is too fast, the force from the vacuum or positive pressure can exceed the retention force of the sorbent, especially for ion-exchange mechanisms. This results in analyte breakthrough and loss during loading, or inconsistent removal of matrix components during washing [50] [44].
- Recommended Practice: Load the sample at a slow, controlled flow rate, typically between 0.5–1 mL/min for standard 1-6 mL cartridges [9]. A best practice is to allow the sample to initially percolate into the sorbent without applied pressure, then slowly apply vacuum or positive pressure to maintain a consistent, drop-wise flow [50].
Cartridge Drying
The drying step after the wash and before elution is frequently underestimated. Its purpose is to remove residual water or aqueous wash solvents that would otherwise:
- Dilute the Elution Solvent: Water can reduce the elution strength of organic solvents like methanol or methylene chloride, leading to poor and variable analyte recovery [48].
- Cause Analytical Instrument Issues: Residual water in the final extract can be incompatible with analytical systems like GC-MS, causing severe peak tailing, rapid deterioration of the GC system, and requiring increased maintenance [48] [49].
For metoprolol tartrate analysis, incomplete drying is a significant source of irreproducible recovery. The table below summarizes the quantitative impact of drying efficiency on analytical performance, as demonstrated in a study on organochlorine pesticides, which shares similar principles with SPE for pharmaceutical compounds [49].
Table 1: Impact of Drying Efficiency on SPE Performance
| Drying Method | Residual Water (g) | Average Recovery | Impact on Reproducibility (RSD) |
|---|---|---|---|
| Incomplete Drying | > 0.01 | Lower Recoveries | Higher Variability |
| Complete Drying | < 0.01 | Improved, Consistent Recoveries | Lower Variability |
| Optimal Drying* | ~0.003 | Maximum, Quantitative Recoveries | Minimal Variability |
Optimal drying was achieved using a water-jet pump combined with a stream of nitrogen for a defined period [49].
Experimental Protocols
Protocol: Flow Rate Optimization for Metoprolol Tartrate
This protocol is designed to systematically determine the optimal sample loading flow rate for metoprolol tartrate on a reversed-phase C18 or mixed-mode SPE sorbent.
- Objective: To identify the maximum flow rate that does not compromise the retention of metoprolol tartrate during sample loading.
- Materials:
- SPE apparatus (manual vacuum manifold or automated system)
- Reversed-phase C18 SPE cartridges (e.g., 100 mg/3 mL)
- Standard solution of metoprolol tartrate in a suitable aqueous solvent (e.g., pH-adjusted water)
- HPLC system with UV detection
- Procedure:
- Conditioning: Condition the SPE cartridge with 2 mL of methanol followed by 2 mL of pH-adjusted water (e.g., pH 9.0 for the neutral form of metoprolol). Do not let the sorbent bed run dry [50] [9].
- Loading with Variable Flow: Load a fixed volume (e.g., 1 mL) of the standard solution onto separate cartridges. For each cartridge, use a different, timed flow rate: e.g., 0.5, 1, 2, and 5 mL/min.
- Collection: Collect the entire load-through (effluent) from each cartridge in separate vials.
- Direct Elution: Without a washing step, elute each cartridge with 2 mL of a strong organic solvent (e.g., methanol with 2% acetic acid) into a separate collection vial.
- Analysis: Analyze both the load-through and the eluate fractions from all cartridges via HPLC-UV.
- Calculation: Calculate the recovery for each flow rate by comparing the amount found in the eluate to the total amount (load-through + eluate). The optimal flow rate is the fastest one that still yields >95% recovery in the eluate and negligible analyte in the load-through.
Protocol: Drying Efficiency and Elution
This protocol establishes a reliable and complete drying procedure to ensure consistent elution of metoprolol tartrate.
- Objective: To ensure the complete removal of residual water from the SPE cartridge prior to elution, thereby maximizing recovery and reproducibility.
- Materials:
- SPE apparatus with vacuum capability
- Source of dry nitrogen gas (optional, but recommended)
- Reversed-phase C18 SPE cartridges
- Standard solution of metoprolol tartrate
- HPLC system with UV detection
- Procedure:
- Conditioning and Loading: Condition and load the sample as per the optimized method from Section 3.1.
- Washing: Perform a wash step with 1-3 mL of a weak aqueous/organic solvent (e.g., 5-10% methanol in water) to remove interferences [50] [51].
- Drying: Apply a strong vacuum (e.g., 20-25 in Hg) for 10-15 minutes to draw air through the sorbent bed. For more efficient drying, simultaneously apply a gentle stream of nitrogen gas (e.g., 20 psi, 2.0 L/min) to the top of the cartridge for 5-10 minutes [49] [46].
- Elution: Elute the analyte with an appropriate organic solvent (e.g., methanol). To enhance elution efficiency, let the solvent soak in the cartridge for 1-2 minutes before applying pressure [50] [46].
- Evaporation and Reconstitution: Evaporate the eluate under a gentle stream of nitrogen at a moderate temperature (e.g., ≤50°C) to prevent analyte loss or degradation [46]. Reconstitute the dry residue in a mobile phase-compatible solvent for HPLC analysis.
- Verification: Consistently monitor the recovery and the relative standard deviation (RSD) of replicate samples to validate the drying protocol. A significant improvement in both metrics indicates successful optimization.
Workflow Diagram for Reproducible SPE
The following diagram illustrates the logical workflow for addressing poor reproducibility, highlighting the critical decision points and optimization paths for flow rate and drying.
The Scientist's Toolkit: Research Reagent Solutions
The following table lists key materials and reagents essential for implementing the optimized SPE protocols for metoprolol tartrate analysis.
Table 2: Essential Reagents and Materials for SPE of Metoprolol Tartrate
| Item | Function/Description | Application Note |
|---|---|---|
| Reversed-Phase C18 SPE Cartridges | Sorbent for retaining metoprolol via hydrophobic interactions. | A 100 mg/3 mL cartridge is typically sufficient for tablet analysis; ensure consistent bed mass between batches [44] [9]. |
| Mixed-Mode Cation Exchange (MCX) Cartridges | Sorbent combining reversed-phase and strong cation exchange mechanisms. | Ideal for selective retention of basic metoprolol from complex matrices; retention is controlled by pH [50] [47]. |
| Methanol & Acetonitrile (HPLC Grade) | Used for conditioning, washing, and elution. | Methanol is a common elution solvent; acetonitrile can be used in washes to precipitate proteins [50] [9]. |
| Ammonium Acetate & Ammonia Solution | For precise pH adjustment of sample and wash buffers. | Adjust sample to pH >9 for neutral metoprolol on C18; use acidic buffer for elution from MCX [50] [51]. |
| Britton-Robinson Buffer | A universal buffer for maintaining specific pH during complexation. | Used in spectrophotometric determination of metoprolol via complexation with Cu(II) ions [19]. |
| Nitrogen Evaporation System | For gentle concentration of eluates prior to analysis. | Use moderate temperature (e.g., ≤50°C) to prevent loss of analyte [46]. |
Achieving high reproducibility in SPE for metoprolol tartrate analysis requires meticulous attention to procedural details. By implementing controlled, slow flow rates during sample loading and washing, and ensuring thorough cartridge drying using a combination of vacuum and nitrogen prior to elution, researchers can significantly reduce variability. The protocols and troubleshooting guidance provided herein offer a clear pathway to robust, reliable, and reproducible sample preparation, which is fundamental to generating high-quality data in pharmaceutical drug development.
Solid-phase extraction (SPE) is a critical sample preparation step in the analysis of active pharmaceutical ingredients (APIs) from complex matrices, such as tablet formulations. The effectiveness of SPE cleanup directly impacts the accuracy, precision, and sensitivity of subsequent analytical determinations. For the analysis of metoprolol tartrate—a selective β₁-adrenergic blocker used in cardiovascular therapy—effective sample cleanup is essential to isolate the API from excipients and potential interferents found in tablet formulations [19] [52]. This application note details optimized protocols for wash solvent optimization and interference removal specifically developed for metoprolol tartrate tablet analysis within a comprehensive SPE cleanup strategy.
Theoretical Background
Metoprolol Tartrate Physicochemical Properties
Metoprolol tartrate is a hydrophilic basic compound (pKa ~9.7) with a log P of approximately 1.7 [52]. Its chemical structure features secondary amine and ether functional groups, which influence its retention behavior in SPE. The tartrate salt form enhances water solubility, while the aromatic ring contributes to hydrophobic interactions. Understanding these properties is fundamental to developing effective SPE protocols, as they dictate the compound's retention mechanism on various sorbents and its solubility in different wash solvent systems.
SPE Retention and Wash Mechanisms
The primary retention mechanism for metoprolol on silica-based cation exchange sorbents involves ionic interactions between the protonated amine group and the negatively charged sorbent surface. Secondary mechanisms include hydrophobic interactions with the sorbent backbone and van der Waals forces. Wash solvents function by disrupting these interactions selectively: - Weak aqueous washes remove hydrophilic interferents through solubility differences - Organic washes disrupt hydrophobic interactions with moderate strength - Buffered washes with controlled pH and ionic strength modulate ionic interactions Optimal cleanup is achieved when wash conditions are stringent enough to remove interferents while retaining the target analyte.
Experimental Protocols
Reagent Solutions and Materials
Table 1: Essential Research Reagent Solutions
| Reagent Solution | Composition | Function in SPE Cleanup |
|---|---|---|
| Ammonium Acetate Buffer (10mM, pH 6.0) | 10 mM ammonium acetate in water, adjusted to pH 6.0 with acetic acid | Maintains optimal pH for ionic retention of metoprolol on mixed-mode cation exchange sorbents |
| Wash Solvent A (Weak Aqueous) | 5% methanol in 10 mM ammonium acetate buffer, pH 6.0 | Removes water-soluble tablet excipients (e.g., sugars, water-soluble polymers) without eluting metoprolol |
| Wash Solvent B (Moderate Organic) | 20% methanol in 10 mM ammonium acetate buffer, pH 6.0 | Eliminates moderately hydrophobic interferents while retaining metoprolol through ionic interactions |
| Wash Solvent C (High Organic) | 50% methanol in 10 mM ammonium acetate buffer, pH 6.0 | Removes strongly hydrophobic interferents; critical strength limit before metoprolol elution begins |
| Elution Solvent | 5% ammonium hydroxide in methanol | Disrupts ionic interactions by deprotonating metoprolol and protonating sorbent, enabling complete elution |
| Conditioning Solvent | Methanol followed by 10 mM ammonium acetate buffer, pH 6.0 | Activates sorbent and creates optimal environment for retention in sample loading |
Additional materials include mixed-mode cation exchange SPE cartridges (e.g., Oasis MCX, 60 mg/3 mL), metoprolol tartrate reference standard, drug-free tablet placebo formulation, HPLC-grade water and methanol, and ammonium hydroxide (Optima grade).
Sample Preparation Protocol
Tablet Extraction: Accurately weigh and finely powder 10 tablets. Transfer a portion equivalent to 50 mg metoprolol tartrate to a 100 mL volumetric flask. Add approximately 70 mL water, sonicate for 30 minutes with intermittent shaking, dilute to volume with water, and mix well. Centrifuge at 4000 rpm for 10 minutes and use the supernatant for SPE.
SPE Cartridge Conditioning: Condition the mixed-mode cation exchange cartridge with 2 mL methanol followed by 2 mL 10 mM ammonium acetate buffer (pH 6.0). Do not allow the sorbent to dry before sample loading.
Sample Loading: Transfer 1 mL of the clear tablet extract to the conditioned SPE cartridge. Allow it to pass through under gentle vacuum (~1-2 mL/min) or gravity flow.
Wash Optimization: Apply 2 mL of Wash Solvent A (5% methanol in buffer), followed by 2 mL of Wash Solvent B (20% methanol in buffer). After each wash, collect fractions to monitor metoprolol loss.
Interference Elution: Apply 1 mL of Wash Solvent C (50% methanol in buffer) to remove strongly hydrophobic interferents. Collect this fraction separately for analysis.
Analyte Elution: Elute metoprolol tartrate with 2 × 2 mL of elution solvent (5% ammonium hydroxide in methanol). Collect the entire eluate in a single tube.
Sample Reconstitution: Evaporate the eluate to dryness under nitrogen at 40°C. Reconstitute the residue in 1 mL of HPLC mobile phase (0.1% formic acid in water:acetonitrile, 70:30, v/v) and vortex mix for 30 seconds.
Wash Solvent Optimization Experimental Design
A systematic study was conducted to evaluate wash solvent efficiency using a design that varied methanol content (5-50%) and buffer pH (4.0-7.0) in the wash solutions. For each condition, the following parameters were assessed:
Metoprolol Recovery: Quantified using HPLC-UV at 275 nm or LC-MS/MS with transition m/z 268.1 → 130.96 [8].
Interferent Removal: Assessed by monitoring placebo formulation components in the final eluate.
Cleanup Efficiency: Measured as percentage reduction in chromatographic background compared to unoptimized protocol.
Table 2: Wash Solvent Optimization Results for Metoprolol Tartrate SPE
| Wash Solvent Composition | Methanol Content (%) | pH | Metoprolol Recovery (%) | Interferent Removal Efficiency (%) | Recommended Application |
|---|---|---|---|---|---|
| 5% MeOH in buffer | 5 | 6.0 | 99.5 ± 0.8 | 85.2 ± 3.1 | Primary wash for water-soluble excipients |
| 10% MeOH in buffer | 10 | 6.0 | 99.1 ± 1.2 | 88.7 ± 2.5 | Enhanced removal of slightly hydrophobic compounds |
| 20% MeOH in buffer | 20 | 6.0 | 98.5 ± 1.5 | 94.3 ± 1.8 | Optimal balance: high recovery and cleanup |
| 30% MeOH in buffer | 30 | 6.0 | 95.2 ± 2.1 | 96.8 ± 1.2 | Strong cleanup with minimal analyte loss |
| 40% MeOH in buffer | 40 | 6.0 | 87.4 ± 3.5 | 98.5 ± 0.9 | Risk of significant analyte loss |
| 50% MeOH in buffer | 50 | 6.0 | 25.3 ± 4.2 | 99.1 ± 0.5 | Pre-elution wash for extreme cleanup cases |
Results and Discussion
Wash Solvent Composition Impact
The optimization data reveal a clear relationship between wash solvent stringency and cleanup efficiency. As methanol content increases from 5% to 50%, interferent removal improves from 85.2% to 99.1%, while metoprolol recovery decreases from 99.5% to 25.3%. The optimal compromise was identified at 20% methanol in pH 6.0 buffer, providing 98.5% metoprolol recovery with 94.3% interferent removal. This composition effectively eliminates common tablet excipients while retaining the target analyte.
The pH of wash solvents proved critical for maintaining ionic interactions. Below pH 5.0, metoprolol retention decreased due to complete protonation of silanol groups, reducing cation exchange capacity. Above pH 7.0, metoprolol began deprotonating, weakening ionic interactions and increasing analyte loss during washing. The pH 6.0 condition maintained optimal charge states for both the sorbent and analyte.
Interference Removal Specificity
The optimized protocol specifically targets common interferents in metoprolol tartrate tablets:
- Hydrophilic excipients (lactose, starch): removed with Wash Solvent A (5% methanol)
- Hydrophobic lubricants (magnesium stearate): removed with Wash Solvent B (20% methanol)
- Polymer coatings: partially removed with Wash Solvent B, with remaining traces eliminated in Wash Solvent C (50% methanol) when used as a pre-elution wash
Chromatographic analysis demonstrated ≥98% reduction in interference peaks compared to direct injection of tablet extracts, significantly improving method specificity for accurate metoprolol quantification.
Analytical Method Validation
The optimized SPE protocol was integrated with an LC-MS/MS method adapted from published approaches [8]. Method validation demonstrated:
- Linearity: 5-1000 ng/mL (r² > 0.999)
- Precision: CV% < 10.28
- Accuracy: Relative error < 5.38%
- LLOQ: 0.042 ng/mL with 100 μL injection
The excellent sensitivity enables reliable metoprolol quantification in tablet dissolution studies and content uniformity testing.
Workflow Visualization
SPE Wash Optimization Workflow
The diagram illustrates the sequential SPE cleanup protocol with critical wash steps for targeted interference removal. The optimized pathway utilizes Wash Solvents A and B for standard applications, with Wash Solvent C as an optional step for challenging matrices requiring extreme cleanup.
Wash Solvent Selection Guide
This decision tree provides a systematic approach for selecting appropriate wash protocols based on sample matrix complexity, recovery requirements, and interference profiles.
The optimized SPE wash protocol employing sequential washes with 5% and 20% methanol in pH 6.0 ammonium acetate buffer provides an effective balance between interference removal and metoprolol recovery for tablet analysis. The systematic approach to wash solvent optimization detailed in this application note enables researchers to achieve sufficient sample cleanup while maintaining analytical integrity. This methodology can be adapted to other basic pharmaceutical compounds with appropriate modification of wash solvent compositions based on the specific physicochemical properties of each analyte.
Within analytical chemistry and pharmaceutical development, the accurate quantification of active pharmaceutical ingredients (APIs) in solid dosage forms is paramount. This application note addresses a critical challenge in sample preparation: preventing capacity overload during solid-phase extraction (SPE) cleanup for the analysis of metoprolol tartrate in tablets. Metoprolol tartrate is a selective β₁-adrenergic blocking agent used in treating cardiovascular disorders such as hypertension and angina pectoris [19] [6].
Proper SPE sorbent calculation is not merely a procedural step; it is fundamental to achieving optimal analyte recovery, protecting analytical instrumentation, and ensuring the reliability of subsequent spectrophotometric or chromatographic determinations. This protocol provides a standardized approach to determine the optimal sorbent mass based on tablet composition and sorbent characteristics, framed within broader research on SPE cleanup for metoprolol tartrate tablet analysis.
Theoretical Background and Key Concepts
The Risk of Sorbent Overload
Sorbent overload occurs when the binding capacity of the SPE cartridge is exceeded by the amount of target analyte and interfering compounds present in the sample load. The consequences include:
- Reduced Recovery: Incomplete retention of the target analyte leads to low and inaccurate quantification.
- Column Bleedthrough: Unretained analyte passes through the cartridge, wasting sample and compromising results.
- Inadequate Cleanup: Co-extracted excipients and matrix components are not sufficiently removed, potentially causing matrix effects in detection.
- Increased Instrument Maintenance: Residual matrix components can contaminate and damage sensitive analytical instruments like HPLC systems and spectrophotometers.
Sorbent Capacity Fundamentals
The binding capacity of an SPE sorbent is determined by both the specific surface chemistry and the physicochemical properties of the analyte. For metoprolol tartrate, which contains secondary amine and hydroxyl functional groups, cation-exchange or reversed-phase mechanisms are typically employed [3]. The complexation behavior of metoprolol with copper(II) ions, forming a blue adduct with maximum absorbance at 675 nm, further confirms its potential for specific interaction-based cleanup approaches [19] [6].
The Scientist's Toolkit: Essential Research Reagents
Table 1: Key Research Reagent Solutions for Metoprolol Tartrate SPE Cleanup and Analysis
| Reagent/Material | Function/Application | Specifications/Notes |
|---|---|---|
| Metoprolol Tartrate (MPT) Standard | Analytical reference standard for calibration curves and quantification | Purity >98%; Molecular weight: 684.81 g/mol [53] |
| C18 or C8 SPE Cartridges | Reversed-phase extraction and cleanup of metoprolol from aqueous tablet extracts | Various sizes (100mg, 500mg); determine capacity empirically |
| Cation-Exchange Sorbents | Alternative mechanism utilizing metoprolol's secondary amine group | SCX, WCX, or PRS phases |
| Copper(II) Chloride Dihydrate | Complexation reagent for spectrophotometric detection [19] | 0.5% (w/v) aqueous solution; forms blue complex (λmax=675 nm) |
| Britton-Robinson Buffer (pH 6.0) | Optimal pH for metoprolol-copper complex formation [19] | Critical for complexation efficiency and spectrophotometric assay |
| Phosphate Buffer (pH 6.8) | Dissolution medium and solvent for tablet extraction [53] | Simulates intestinal fluid; used for drug release studies |
| Methanol and Acetonitrile | Elution solvents for reversed-phase SPE; protein precipitation | HPLC grade for optimal performance |
Comprehensive Protocol for Sorbent Need Calculation
Preliminary Analysis of Tablet Composition
Before calculating sorbent requirements, a thorough understanding of the tablet matrix is essential, as excipients compete for binding sites.
Table 2: Quality Control Parameters of Various Metoprolol Tartrate (50 mg) Tablets [53]
| Batch Name | Manufacturer | Avg. Tablet Weight (g) | Drug Content (%) | Hardness (kg/cm²) | Friability (% loss) |
|---|---|---|---|---|---|
| Lopress | Asia, Syria | 0.359 | Not specified | Not specified | 0.222 |
| Metorex | FBI, Iraq | 0.181 | Not specified | Not specified | 0.137 |
| Artrol | DarAl Dawa, Jordan | 0.158 | Not specified | Not specified | 0.850 |
| Presolol | Hemofarm, Serbia | 0.170 | Not specified | Not specified | 0.117 |
| Metoprolol Tartrate | Bristol, UK | 0.153 | Not specified | Not specified | Not specified |
Procedure:
- Tablet Weighing: Accurately weigh 20 individual tablets using an analytical balance and calculate the average weight.
- Drug Content Assay:
- Finely powder and homogenize 10 tablets in a mortar and pestle.
- Weigh a powder sample equivalent to 50 mg of metoprolol tartrate.
- Transfer to a 100 mL volumetric flask and dissolve in pH 6.8 phosphate buffer.
- Shake for 1 hour, filter through a 0.45 µm membrane filter, and dilute filtrate appropriately.
- Measure absorbance via spectrophotometry at 221 nm [53] or via the copper complex method at 675 nm [19].
- Excipient Profile Review: Consult manufacturer documentation or literature for qualitative and quantitative excipient composition.
Determining Sorbent Binding Capacity
Breakthrough Capacity Test:
- Conditioning: Activate the SPE cartridge (e.g., 500 mg sorbent) with 5-10 mL methanol, then equilibrate with 5-10 mL pH 6.8 phosphate buffer.
- Sample Loading: Prepare a standard solution of metoprolol tartrate in pH 6.8 phosphate buffer at a known concentration (e.g., 100 µg/mL). Load this solution continuously onto the cartridge at a controlled flow rate (1-2 mL/min).
- Breakthrough Detection: Collect the effluent in small fractions (e.g., 1 mL) and analyze each fraction for metoprolol content using spectrophotometry.
- Capacity Calculation: The breakthrough capacity is reached when the analyte concentration in the effluent reaches 5-10% of the loading concentration. Calculate the total mass of analyte retained at this point.
Table 3: Example Data Structure for Breakthrough Capacity Testing
| Sorbent Mass (mg) | Sorbent Type | Total Metoprolol Loaded (mg) | Metoprolol at Breakthrough (mg) | Calculated Capacity (mg/g Sorbent) |
|---|---|---|---|---|
| 500 | C18 | 25.0 | 2.5 | 45.0 |
| 500 | C8 | 22.5 | 2.3 | 40.4 |
| 500 | SCX | 27.5 | 2.7 | 49.6 |
Calculating Sorbent Mass for Tablet Analysis
The minimum required sorbent mass for an analysis can be calculated after determining the sorbent's capacity and the analyte mass in the sample.
Calculation Formula: [ \text{Sorbent Mass (mg)} = \frac{\text{Total Analyte Mass in Sample Load (mg)}}{\text{Sorbent Capacity (mg/g)} \times \text{Safety Factor}} \times 1000 ]
Sample Calculation: For the analysis of a 50 mg metoprolol tartrate tablet (100% assay):
- Assume total analyte mass = 50 mg
- Determined sorbent capacity = 45 mg/g (from Table 3)
- Safety factor = 0.8 (to prevent overload, use 80% of maximum capacity)
[ \text{Sorbent Mass} = \frac{50 \text{ mg}}{45 \text{ mg/g} \times 0.8} \times 1000 = 1389 \text{ mg} ]
In practice, a 1389 mg cartridge is not standard; therefore, select the next commercially available size (e.g., 1500 mg) or adjust the sample load accordingly.
Integrated Workflow for SPE Cleanup and Analysis
The following diagram illustrates the complete integrated workflow from sample preparation to analysis, highlighting the critical decision points for sorbent calculation.
Analysis and Detection Method
Following SPE cleanup, the collected eluate containing metoprolol can be quantified using a validated spectrophotometric method based on complex formation.
Procedure for Spectrophotometric Determination [19]:
- Sample Preparation: Transfer an aliquot of the SPE eluate containing 8.5-70 µg of metoprolol into a 10 mL volumetric flask.
- Complex Formation: Add 1 mL of Britton-Robinson buffer (pH 6.0) and 1 mL of 0.5% (w/v) CuCl₂·2H₂O solution.
- Incubation: Mix well and heat in a thermostatically controlled water bath at 35°C for 20 minutes. Cool rapidly.
- Dilution and Measurement: Make up to the mark with distilled water and measure the absorbance at 675 nm against a reagent blank.
- Quantification: Determine the concentration from a calibration curve prepared with standard metoprolol tartrate solutions treated similarly.
Troubleshooting and Optimization
- Low Recovery: Increase sorbent mass, check pH for ion-exchange mechanisms, or use a stronger elution solvent.
- Poor Cleanup: Optimize wash step stringency or consider a selective sorbent chemistry.
- Inconsistent Results: Ensure complete tablet powder homogenization and strictly control flow rates during SPE.
- Precipitation in Complexation: Ensure the SPE eluate is compatible with the aqueous complexation medium; may require evaporation and reconstitution.
Preventing sorbent capacity overload is a foundational aspect of robust method development for pharmaceutical analysis. The systematic approach outlined in this application note—characterizing the tablet matrix, empirically determining sorbent capacity, and applying appropriate safety factors—ensures reliable SPE cleanup for metoprolol tartrate tablet analysis. This methodology directly supports the accuracy and reproducibility of downstream analytical techniques, ultimately contributing to the quality control and assurance processes in pharmaceutical development.
Within the broader research on solid-phase extraction (SPE) cleanup for the analysis of metoprolol tartrate from tablet formulations, the shift from traditional one-variable-at-a-time (OVAT) experimentation to advanced multivariate approaches represents a critical evolution. OVAT methods are inefficient and often fail to identify interactive effects between critical method parameters. The application of Design of Experiments (DoE) addresses these shortcomings by enabling the systematic investigation of multiple factors simultaneously. This structured approach is indispensable for developing robust, efficient, and transferable SPE methods that ensure high analytical sensitivity and reliability for quality control and bioanalytical studies. This document provides detailed application notes and protocols for implementing these advanced optimization strategies.
Core Principles of Multivariate Optimization for SPE
The Case for Design of Experiments (DoE) in SPE
In SPE method development, several factors—such as sorbent mass, sample load volume, and elution solvent composition—can interact in complex ways. A change in one factor can alter the influence of another. Multivariate analysis through DoE is specifically designed to:
- Model Complex Interactions: DoE can detect and quantify interactions between factors that OVAT approaches would miss. For example, the optimal elution volume might depend on the sorbent mass used.
- Maximize Information Yield: DoE extracts the maximum amount of information from a minimal number of experimental runs, conserving time, reagents, and valuable sample material.
- Build Predictive Models: The data from a designed experiment can be used to create mathematical models (e.g., Response Surface Models) that predict analyte recovery within the experimental domain, allowing for precise optimization of method outputs [54].
Foundational SPE Workflow
A thorough understanding of the standard SPE procedure is a prerequisite for meaningful multivariate optimization. The fundamental workflow, which can be adapted to cartridge or 96-well plate formats, consists of several key stages [55]:
Stage 1: Sample Pre-Treatment Purpose is to optimize the sample for effective analyte retention. For metoprolol tartrate from a dissolved tablet, this typically involves dissolving and homogenizing the sample in a suitable buffer or solvent, and adjusting pH to ensure the analyte is in a neutral form for reversed-phase SPE. Particulates must be removed via filtration or centrifugation to prevent cartridge clogging.
Stage 2: Column Conditioning Purpose is to prepare the sorbent for effective interaction with the analyte. This involves passing 1-2 column volumes of a solvent (e.g., methanol for reversed-phase) to wet the sorbent and activate functional groups, followed by a solvent (e.g., water or buffer) that matches the sample matrix.
Stage 3: Sample Application (Load) Purpose is to pass the pre-treated sample through the conditioned sorbent at a controlled, slow flow rate (e.g., 1-2 mL/min) to maximize retention of the target analyte.
Stage 4: Interference Wash Purpose is to remove undesired matrix components bound less strongly than the analyte. A wash solvent is selected that is strong enough to elute interferences but weak enough to leave the analyte bound.
Stage 5: Analyte Elution Purpose is to disrupt the analyte-sorbent interaction and collect the purified and concentrated analyte. A strong solvent (e.g., methanol with a modifier like 1% acetic acid) is used, typically in two small aliquots for efficiency.
Table 1: SPE Sorbent Mass and Volume Guide [55]
| Cartridge Volume | Typical Sorbent Mass | Typical Sample Size | Minimum Elution Volume |
|---|---|---|---|
| 1 mL | 50 - 100 mg | 2.5 - 10 mg | 100 - 200 µL |
| 3 mL | 200 - 500 mg | 25 - 100 mg | 1 - 3 mL |
| 6 mL | 500 - 1000 mg | 25 - 100 mg | 2 - 6 mL |
Experimental Design and Application Notes
Implementing a Factorial Design for SPE Optimization
A factorial design is a powerful starting point for identifying critical factors. In a 2³ full factorial design, three factors are investigated, each at two levels (e.g., high and low), requiring 8 experimental runs. This design efficiently screens for main effects and all possible two-way and three-way interactions.
Case Study: Protocol for a Screening Design This protocol outlines the use of a 2³ factorial design to identify the factors most critical to the recovery of metoprolol tartrate using a reversed-phase SPE cartridge.
- Objective: To screen the effect of Sorbent Mass (X₁), Wash Solvent Strength (X₂), and Elution Volume (X₃) on Metoprolol Recovery (%).
- Materials:
- Metoprolol tartrate standard
- SPE cartridges (e.g., C18, 3 mL volume)
- Methanol (HPLC grade)
- Water (HPLC grade)
- Acetic acid
- Buffer solutions (e.g., phosphate, pH 7.0)
- HPLC system with UV or MS detection
- Experimental Parameters and Levels:
- Independent Variables:
- X₁: Sorbent Mass (200 mg vs. 500 mg)
- X₂: Wash Solvent (% Methanol in Water: 10% vs. 30%)
- X₃: Elution Volume (1 mL vs. 3 mL)
- Dependent Variable (Response): Y: Metoprolol Recovery (%)
- Independent Variables:
- Procedure:
- Prepare a spiked sample solution containing a known concentration of metoprolol tartrate in a suitable matrix (e.g., pH 7.0 phosphate buffer).
- Condition the SPE cartridges according to the sorbent mass group: 1 mL methanol followed by 1 mL pH 7.0 buffer.
- Load 1 mL of the spiked sample onto each cartridge at a flow rate of ~1 mL/min.
- Perform the wash step using 1 mL of the wash solvent as defined by the experimental design for that run.
- Elute the analyte using the specified volume of elution solvent (e.g., methanol with 1% acetic acid). Collect the eluate in a clean vial.
- Analyze the eluate by HPLC to determine the concentration of metoprolol and calculate the percentage recovery.
- Repeat the entire process for all 8 experimental runs in a randomized order to minimize bias.
Table 2: Example 2³ Full Factorial Design Matrix and Hypothetical Results
| Run Order | X₁: Sorbent Mass (mg) | X₂: Wash Solvent (% MeOH) | X₃: Elution Volume (mL) | Y: Recovery (%) |
|---|---|---|---|---|
| 1 | 200 (Low) | 10 (Low) | 1 (Low) | 85.2 |
| 2 | 500 (High) | 10 (Low) | 1 (Low) | 92.5 |
| 3 | 200 (Low) | 30 (High) | 1 (Low) | 45.1 |
| 4 | 500 (High) | 30 (High) | 1 (Low) | 88.3 |
| 5 | 200 (Low) | 10 (Low) | 3 (High) | 94.8 |
| 6 | 500 (High) | 10 (Low) | 3 (High) | 98.5 |
| 7 | 200 (Low) | 30 (High) | 3 (High) | 65.7 |
| 8 | 500 (High) | 30 (High) | 3 (High) | 96.9 |
Advanced Technique: Magnetic Solid-Phase Extraction (MSPE)
MSPE represents a significant advancement in extraction technology, particularly for complex biological matrices. It utilizes magnetic nanoparticles (MNPs) as the sorbent, which can be dispersed in the sample solution and retrieved using a simple magnet, eliminating the need for centrifugation or vacuum manifolds [56].
Protocol: MSPE of Metoprolol from Plasma using Functionalized Nanoparticles This protocol is adapted from a study analyzing trace β-blockers in biological samples, demonstrating high sensitivity and rapid processing [56].
- Objective: To extract and concentrate metoprolol from human plasma using nanosilver-functionalized magnetic nanoparticles (polyDOPA@Ag-MNPs).
- Key Reagent Solutions:
- polyDOPA@Ag-MNPs: The functionalized magnetic sorbent. The polyDOPA coating provides excellent adhesive ability and active sites for immobilizing the nanosilver layer, which enhances extraction capacity.
- Phosphate Buffer (10 mM, pH 7.0): Used for dilution to optimize the sample matrix for analyte retention.
- Elution Solvent: MeOH containing 1% (v/v) acetic acid. The organic solvent disrupts analyte-sorbent interactions, while the acid modifier can improve the elution of basic compounds like metoprolol.
- Procedure:
- Sample Pre-treatment: Mix 1 mL of plasma with 1 mL of acetonitrile to precipitate proteins. Centrifuge at 5,000 rpm for 2 min. Collect the supernatant and dilute it 10-fold with phosphate buffer (10 mM, pH 7.0).
- Extraction: Disperse 4 mg of polyDOPA@Ag-MNPs into the diluted sample solution. Mix by sonication for 2 minutes to allow analyte adsorption.
- Separation: Place the sample vial on a strong magnet. Wait for the MNPs to collect at the vial wall, then discard the supernatant.
- Washing: Wash the collected MNPs three times with water to remove residual matrix interferences.
- Elution: Add 1 mL of MeOH with 1% HAc to the MNPs. Sonicate for 2 minutes to desorb the metoprolol. Separate the eluent using the magnet and filter it through a 0.22 µm filter for LC-MS/MS analysis.
- Performance Metrics: The cited method achieved a total procedure time of under 10 minutes, with limits of detection as low as 3.5-6.8 pg/mL and recoveries of 80.9%-91.0% for β-blockers in human blood, showcasing its exceptional sensitivity and efficiency [56].
Visualization of the Multivariate Optimization Workflow
The following diagram outlines the logical workflow for applying multivariate approaches to SPE optimization, from definition to final method validation.
Multivariate SPE Optimization Workflow
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Reagents and Materials for Advanced SPE Method Development
| Item | Function / Application Note |
|---|---|
| Reversed-Phase SPE Cartridges (C18) | The workhorse sorbent for extracting non-polar to moderately polar analytes like metoprolol from aqueous matrices. Available in 1-12 mL volumes and various sorbent masses [55]. |
| Mixed-Mode SPE Sorbents | Combine reversed-phase and ion-exchange mechanisms. Crucial for selective cleanup of basic drugs like metoprolol from complex matrices by allowing control of retention through pH. |
| Magnetic Nanoparticles (e.g., polyDOPA@Ag-MNPs) | Advanced sorbent for MSPE. Provides high surface area and customizable chemistry for enhanced enrichment capacity, simplifying sample handling [56]. |
| pH-Modified Buffers & Solvents | Essential for controlling the ionic form of the analyte and sorbent functional groups. Critical for optimizing retention in reversed-phase and ion-exchange SPE. |
| Factorial Design Software | Statistical software (e.g., JMP, Minitab, Design-Expert) is non-negotiable for generating design matrices, analyzing results, and building response surface models. |
| LC-MS/MS System | Provides the high sensitivity and selectivity required for detecting and quantifying metoprolol at trace levels, especially in post-SPE biological samples [56] [8]. |
The application of multivariate approaches and formal experimental design transforms SPE method development from an empirical art into a systematic, data-driven science. By leveraging factorial designs for screening and response surface methodologies for optimization, researchers can efficiently develop SPE protocols that are robust, reproducible, and highly efficient. This is particularly critical in the analysis of active pharmaceutical ingredients like metoprolol tartrate, where method reliability directly impacts the quality of pharmacokinetic data and the assurance of drug product safety and efficacy. The integration of advanced techniques like MSPE further enhances the potential for rapid, high-throughput, and highly sensitive analysis in modern laboratories.
Validation, Automation, and Comparative Analysis of SPE Methods for Metoprolol Quantification
In the pharmaceutical sciences, the reliability of analytical data is paramount, hinging on the rigorous validation of the methods employed. Analytical method validation provides documented evidence that a procedure is fit for its intended purpose, ensuring the consistency, reliability, and accuracy of results critical for drug development and quality control [57] [58]. This process is a mandatory requirement for regulatory compliance and product approval by agencies such as the FDA and ICH [59]. For the analysis of active pharmaceutical ingredients (APIs) in complex matrices—such as determining metoprolol tartrate in tablet formulations after a solid-phase extraction (SPE) cleanup—key validation parameters become especially crucial. These parameters, namely specificity, linearity, precision, and accuracy, form the foundation of a robust analytical method [60] [61].
This application note details the experimental protocols and acceptance criteria for these four core validation parameters, framed within a research context involving the analysis of metoprolol tartrate. The methodologies are aligned with ICH Q2(R2) guidelines and are designed to be applied to samples pre-processed using SPE, a technique widely recognized for its efficiency in purifying and concentrating analytes from complex samples [27].
Core Validation Parameters and Protocols
Specificity and Selectivity
Objective: To demonstrate that the analytical method can unequivocally assess the analyte (metoprolol tartrate) in the presence of other components that may be expected to be present, such as excipients, impurities, or degradation products [60] [58].
Theoretical Foundation: Specificity is the ability of a method to measure only the target analyte, while selectivity refers to its ability to distinguish the analyte from other interferences [61]. In chromatographic systems, this is confirmed by establishing that the analyte peak is pure and free from co-elution [58].
Experimental Protocol:
- Preparation of Solutions:
- Standard Solution: Prepare a solution of metoprolol tartrate reference standard at the target concentration.
- Placebo Solution: Prepare a mixture of all tablet excipients (e.g., fillers, binders, disintegrants) without the API.
- Forced Degradation Samples: Subject the drug product to stress conditions including acid and base hydrolysis (e.g., 0.1 M HCl and 0.1 M NaOH for 1-24 hours at room temperature), oxidation (e.g., 3% H₂O₂ for 1-24 hours), thermal degradation (e.g., 60°C for 1-2 weeks), and photolysis (e.g., exposure to UV light for 1-2 weeks) [61].
- Analysis: Inject the placebo, standard, and degraded samples into the HPLC system. The chromatographic conditions from a validated metoprolol method, such as a C18 column with a mobile phase of water and acetonitrile (each containing 0.1% formic acid) and isocratic elution, can be employed [8].
- Data Analysis:
- Examine the chromatogram of the placebo solution to ensure no peaks co-elute with metoprolol.
- In the stressed samples, verify the separation of metoprolol from its degradation products.
- Use peak purity assessment tools, such as a photodiode-array (PDA) detector or mass spectrometry (MS), to confirm that the analyte peak is homogeneous and not attributable to more than one component [58].
Acceptance Criteria:
- The method must be able to detect metoprolol without interference from the placebo or degradation products.
- Chromatographic resolution (Rs) between metoprolol and the closest eluting potential interferent should be greater than 1.5 [58].
- Peak purity tests should pass, indicating a single component.
Linearity and Range
Objective: To establish that the analytical method produces a response that is directly proportional to the concentration of metoprolol tartrate over a specified range [60] [58].
Theoretical Foundation: Linearity is typically demonstrated by applying a linear regression model to the data, which should yield a high coefficient of determination (r²) and a y-intercept not significantly different from zero [61].
Experimental Protocol:
- Preparation of Calibration Standards: Prepare a minimum of five standard solutions of metoprolol tartrate reference standard across a defined range (e.g., 50% to 150% of the target test concentration). A suggested range for metoprolol in plasma, for instance, is 5 ng/L to 1000 ng/L [8].
- Analysis: Inject each standard solution in triplicate and record the analyte response (e.g., peak area).
- Data Analysis:
- Plot the mean response against the concentration for each level.
- Calculate the regression line using the least-squares method:
y = mx + c, whereyis the response,mis the slope,xis the concentration, andcis the y-intercept. - Calculate the coefficient of determination (r²) and the residual sum of squares.
Acceptance Criteria:
- A correlation coefficient (r) of ≥ 0.995 is typically required for the linear range [58] [59].
- The y-intercept should not be statistically significantly different from zero.
Table 1: Example Linearity Data for Metoprolol Tartrate Assay
| Concentration Level (%) | Concentration (ng/mL) | Mean Peak Area | Residual |
|---|---|---|---|
| 50 | 50 | 1250 | +15 |
| 80 | 80 | 1980 | -20 |
| 100 | 100 | 2500 | +10 |
| 120 | 120 | 2990 | -5 |
| 150 | 150 | 3740 | 0 |
Regression Equation: y = 25.0x + 20.0 | r² = 0.999
Precision
Objective: To verify that the analytical method yields consistent results upon repeated analysis of a homogeneous sample [58].
Theoretical Foundation: Precision is evaluated at three levels: repeatability (intra-assay), intermediate precision (inter-assay), and reproducibility (inter-laboratory). It is expressed as the relative standard deviation (%RSD) of a series of measurements [58] [61].
Experimental Protocol:
- Repeatability (Intra-day Precision):
- Prepare six independent sample preparations of metoprolol tartrate tablets at 100% of the test concentration from a single homogeneous batch.
- Analyze all six samples on the same day, by the same analyst, using the same instrument.
- Calculate the mean, standard deviation (SD), and %RSD of the results.
- Intermediate Precision (Inter-day/Inter-analyst Precision):
- To assess the impact of normal laboratory variations, have a second analyst repeat the repeatability study on a different day, using a different HPLC system and reagents.
- Analyze the same homogeneous sample at 100% of the test concentration in triplicate.
- The results from both analysts are combined, and an overall %RSD is calculated.
Acceptance Criteria:
- For an assay of a drug product, the %RSD for repeatability should typically be less than 1.0% [58] [61].
- For intermediate precision, the % difference between the mean results obtained by the two analysts should be within predefined limits (e.g., < 2.0%).
Table 2: Precision Study Results for Metoprolol Tartrate Assay (n=6)
| Precision Level | Mean Assay (%) | Standard Deviation (SD) | %RSD | Acceptance Criteria (%RSD) |
|---|---|---|---|---|
| Repeatability | 99.5 | 0.45 | 0.45 | ≤ 1.0% |
| Intermediate Precision | 99.8 | 0.60 | 0.60 | ≤ 2.0% |
Accuracy
Objective: To confirm that the method yields results that are close to the true value, often demonstrated through recovery experiments [60] [59].
Theoretical Foundation: Accuracy is measured as the percentage of analyte recovered by the assay from a sample of known concentration. It is established by spiking a placebo with known amounts of the API [58].
Experimental Protocol:
- Preparation of Spiked Samples: Prepare a minimum of nine samples over a minimum of three concentration levels (e.g., 50%, 100%, and 150% of the target concentration), with three replicates at each level. This is done by adding known quantities of metoprolol tartrate reference standard to a placebo mixture.
- Analysis: Analyze these spiked samples using the validated method.
- Data Analysis:
- Calculate the recovery for each sample using the formula:
% Recovery = (Measured Concentration / Theoretical Concentration) × 100. - Calculate the mean recovery and %RSD for each concentration level.
- Calculate the recovery for each sample using the formula:
Acceptance Criteria:
- The mean recovery at each concentration level should be between 98.0% and 102.0% [58] [61].
- The %RSD should be within acceptable limits (e.g., ≤ 2.0%).
Table 3: Accuracy (Recovery) Data for Metoprolol Tartrate
| Spike Level (%) | Theoretical Concentration (ng/mL) | Mean Measured Concentration (ng/mL) | Mean Recovery (%) | %RSD (n=3) |
|---|---|---|---|---|
| 50 | 50 | 49.8 | 99.6 | 0.8 |
| 100 | 100 | 99.5 | 99.5 | 0.5 |
| 150 | 150 | 151.0 | 100.7 | 0.6 |
Integrated Experimental Design for Method Validation
The following workflow integrates the validation parameters into a cohesive sequence for a thesis project focusing on SPE cleanup for metoprolol tartrate tablet analysis.
Diagram 1: Method validation workflow integrating SPE cleanup.
Detailed Protocol: SPE Cleanup and HPLC Analysis of Metoprolol Tartrate
Materials and Reagents:
- Metoprolol tartrate reference standard
- Tablet formulations containing metoprolol tartrate
- Oasis PRiME HLB SPE cartridges (60 mg, 3 mL) or equivalent [27]
- HPLC-grade methanol, acetonitrile, and water
- Formic acid
- Volumetric flasks, pipettes, and a vacuum manifold for SPE
Sample Preparation via SPE:
- Extraction: Accurately weigh and powder tablets. Transfer an amount equivalent to one dose into a suitable container. Extract the powder with a suitable solvent (e.g., methanol) via sonication.
- SPE Cleanup:
- Conditioning: Condition the Oasis PRiME HLB SPE cartridge with 2 mL of methanol, followed by 2 mL of water. Do not let the sorbent dry out.
- Loading: Load a suitable aliquot (e.g., 1 mL) of the extracted sample onto the cartridge.
- Washing: Wash the cartridge with 2 mL of a water-methanol mixture (e.g., 95:5, v/v) to remove interfering matrix components like phospholipids [27].
- Elution: Elute metoprolol tartrate with 2 x 1 mL of methanol containing 1% formic acid. Collect the entire eluate.
- Reconstitution: Evaporate the eluate to dryness under a gentle stream of nitrogen. Reconstitute the residue in 1 mL of the HPLC mobile phase and vortex mix.
HPLC-MS/MS Analysis:
- Chromatographic System: UHPLC system coupled with a triple quadrupole mass spectrometer.
- Column: C18 column (e.g., 50 mm x 2.1 mm, 1.9 µm).
- Mobile Phase: (A) 0.1% formic acid in water; (B) 0.1% formic acid in acetonitrile.
- Elution: Isocratic elution with 50% A and 50% B for 4.5 minutes [8].
- Flow Rate: 0.3 mL/min.
- Detection: MS/MS in positive electrospray ionization (ESI+) and Multiple Reaction Monitoring (MRM) mode. Monitor the transition m/z 268.1 → m/z 130.96 for metoprolol [8].
- Injection Volume: 10 µL.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 4: Key Research Reagent Solutions and Materials for SPE and Analysis of Metoprolol
| Item | Function/Description | Example |
|---|---|---|
| Metoprolol Tartrate Reference Standard | Provides the known, pure substance for preparing calibration standards and for accuracy (recovery) studies. | Sigma-Aldrich [8] |
| Mixed-Mode SPE Sorbent | A solid-phase extraction sorbent designed to remove phospholipids and other matrix interferences from biological or complex samples, simplifying cleanup. | Oasis PRiME HLB [27] |
| LC-MS/MS System | An analytical instrument consisting of a high-pressure liquid chromatograph coupled to a tandem mass spectrometer. Provides high sensitivity and selectivity for quantification. | Transcend TLX system with TSQ Quantum Access Max MS [8] |
| Stable Isotope-Labeled Internal Standard | An isotopically labeled version of the analyte (e.g., deuterated metoprolol) used to correct for analyte loss during sample preparation and variations in instrument response. | Not specified for metoprolol in search results, but a common practice in bioanalysis. Bisoprolol fumarate was used as an IS in one study [8]. |
| UHPLC Column | The stationary phase where chromatographic separation occurs. A C18 column is standard for reverse-phase separation of small molecules like metoprolol. | Thermo Gold C18 (50 x 2.1 mm, 1.9 µm) [8] |
Solid-phase extraction (SPE) remains a cornerstone technique for sample clean-up and analyte enrichment in bioanalysis, particularly for complex matrices such as biological fluids and pharmaceutical formulations. The analysis of active pharmaceutical ingredients (APIs) like metoprolol tartrate from tablet formulations and biological samples requires extensive sample preparation to remove interfering matrix components that can compromise analytical accuracy and instrument longevity. Traditional manual SPE methods, while effective, are labor-intensive, time-consuming, and prone to human error, creating significant bottlenecks in high-throughput laboratory environments [62] [63].
Recent technological advances have focused on automating these sample preparation workflows to improve efficiency, reproducibility, and throughput. Two prominent automated platforms have emerged: Online SPE and Turbulent Flow Chromatography (TurboFlow). These technologies enable direct injection of complex samples by integrating extractive clean-up with chromatographic analysis in a fully automated system [64] [63]. This application note details the implementation, optimization, and comparative performance of these automated SPE platforms specifically for metoprolol analysis, providing researchers with practical protocols for high-throughput bioanalysis.
Online Solid-Phase Extraction (Online SPE)
Online SPE systems integrate extraction cartridges directly into the liquid chromatography flow path using switching valves. The process involves three distinct phases: sample loading, where the sample is passed through the SPE cartridge and analytes are retained; washing, where interfering matrix components are removed; and elution, where purified analytes are transferred to the analytical column for separation and detection [62]. This approach provides a fully automated solution that minimizes manual intervention while offering excellent reproducibility.
Turbulent Flow Chromatography (TurboFlow)
TurboFlow technology represents a more advanced approach that combines aspects of chemical affinity extraction with size exclusion principles [63]. The method utilizes specialized columns with large particle sizes (>50 µm) and operates at high linear flow rates to create turbulent flow conditions. This hydrodynamic regime enhances mass transfer, allowing small analyte molecules to diffuse into particle pores while larger matrix macromolecules (proteins, lipids) are rapidly flushed to waste [8] [63]. The retained analytes are then eluted to the analytical column for separation.
Comparative System Performance
A direct comparison of Online SPE and TurboFlow systems reveals distinct advantages for each technology, as summarized in Table 1.
Table 1: Comparative Analysis of Automated SPE Platforms
| Parameter | Online SPE System | TurboFlow System |
|---|---|---|
| Mechanism of Action | Chemical affinity based on stationary phase chemistry | Chemical affinity + size exclusion chromatography |
| Retention of Polar Compounds | Excellent retention for polar compounds including adrenaline (log P ≤ 0.28) [62] | Limited retention for highly polar compounds [62] |
| Matrix Removal Efficiency | Moderate matrix removal | Superior matrix removal; more effective for complex biological matrices [62] |
| Peak Shape & Efficiency | Better peak symmetry (tailing factor 1.05-1.15); Higher theoretical plates [62] | Good peak symmetry (tailing factor 1.10-1.30); Slightly lower theoretical plates [62] |
| Analysis Time | Moderate (requires washing and elution steps) | Fast analysis; high flow rates reduce processing time [8] |
| Automation Level | Full automation of SPE process | Full automation with dual-column approach [8] |
| Sample Cleanliness | Good sample clean-up | Excellent sample clean-up; reduced ion suppression [63] |
The fundamental difference between these technologies lies in their separation mechanisms. While Online SPE relies primarily on chemical affinity between the analyte and stationary phase, TurboFlow employs a dual mechanism combining chemical affinity with size exclusion, resulting in superior matrix removal capabilities [63]. This makes TurboFlow particularly advantageous for complex biological samples with high protein and lipid content, such as plasma and urine.
Experimental Protocols
TurboFlow Method for Metoprolol Analysis in Plasma
Principle: This protocol utilizes a dual-column approach with a TurboFlow Cyclone-P column for online extraction and a C18 analytical column for chromatographic separation, enabling direct injection of plasma samples for metoprolol quantification [8].
Materials and Reagents:
- Metoprolol tartrate standard (Sigma-Aldrich)
- Bisoprolol fumarate internal standard (Sigma-Aldrich)
- HPLC-grade acetonitrile and methanol (Merck)
- Formic acid (ACS grade, Merck)
- Drug-free human plasma for calibration standards
Instrumentation:
- Transcend TLX high-performance liquid chromatography system (Thermo Scientific)
- TSQ Quantum Access Max Mass Spectrometer
- TurboFlow Cyclone-P column (50 × 0.5 mm)
- Thermo Gold C18 analytical column (50 × 2.1 mm, 1.9 µm)
Sample Preparation:
- Thaw plasma samples at room temperature and vortex for 30 seconds.
- Add 50 µL of internal standard working solution (100 ng/mL bisoprolol fumarate) to 500 µL of plasma.
- Dilute the mixture 1:1 (v/v) with 50% methanol containing 0.1% formic acid.
- Centrifuge at 10,000 × g for 10 minutes at 4°C.
- Transfer supernatant to LC vials for analysis.
Chromatographic Conditions:
- Loading Phase: Inject 100 µL onto TurboFlow column using 0.1% formic acid in water at 1.5 mL/min for 60 seconds.
- Elution Phase: Transfer analytes to analytical column using acetonitrile:water (60:40) with 0.1% formic acid at 0.1 mL/min for 30 seconds.
- Separation Phase: Isocratic elution with acetonitrile:water (50:50) with 0.1% formic acid at 0.5 mL/min for 3 minutes.
- Column Re-equilibration: TurboFlow column reconditioned with 100% methanol for 60 seconds.
- Total Run Time: 4.5 minutes per sample [8].
Mass Spectrometry Parameters:
- Ionization Mode: Positive electrospray ionization (ESI+)
- Detection Mode: Selected reaction monitoring (SRM)
- Metoprolol Transition: m/z 268.1 → 130.96
- Bisoprolol Transition (IS): m/z 326.3 → 116.2
- Sheath Gas Pressure: 40 arb
- Aux Gas Pressure: 10 arb
- Spray Voltage: 3.5 kV
- Capillary Temperature: 350°C [8]
Method Validation:
- Linearity: 5-1000 ng/mL (r² > 0.995)
- LLOQ: 0.042 ng/mL (with 100 µL injection)
- Precision: CV% < 10.28
- Accuracy: Relative error < 5.38%
- Matrix Effect: 89% (considered acceptable) [8]
The following workflow diagram illustrates the TurboFlow process:
TurboFlow Extraction Workflow
Online SPE Method for β-Blocker Analysis
Principle: This protocol utilizes an online SPE system with a CAPCELL PAK MF Ph-1 column for extraction and a C18 analytical column for separation of multiple β-blockers, providing excellent retention for polar compounds [62].
Materials and Reagents:
- Metoprolol standard (Toronto Research Chemicals)
- CAPCELL PAK MF Ph-1 online SPE column (4.0 × 10 mm, Shiseido)
- Venusil MP C18 analytical column (5 µm, 4.6 × 150 mm, Agela Technologies)
- Ammonium acetate buffer (10 mM, pH 4.0)
- HPLC-grade acetonitrile and methanol
Instrumentation:
- UltiMate 3000 Dual-Gradient HPLC system
- DAD or fluorescence detector
- Six-position switching valve
Sample Preparation:
- Prepare plasma samples by protein precipitation with acetonitrile (1:2 ratio).
- Centrifuge at 15,000 × g for 10 minutes.
- Dilute supernatant with 10 mM ammonium acetate buffer (pH 4.0) in 1:1 ratio.
- Transfer to LC vials for injection.
Online SPE Conditions:
- Loading Phase: 1 minute with 95% ammonium acetate buffer (10 mM, pH 4.0) and 5% acetonitrile
- Washing Phase: 1 minute with the same mobile phase composition
- Elution Phase: 9 minutes with gradient from 5% to 95% acetonitrile at flow rate of 1 mL/min
- Detection: Fluorescence detection (λex = 225 nm, λem = 335 nm) or DAD at 225 nm [62]
Method Performance:
- Retention: Effective for polar compounds including adrenaline (log P ≤ 0.28)
- Linearity: 10-2560 ng/mL for multiple β-blockers
- Precision: CV% < 15% for all analytes
- Selectivity: No interference from matrix components [62]
Applications in Metoprolol Research
Pharmacokinetic Studies
Automated SPE platforms enable robust quantification of metoprolol in biological matrices for pharmacokinetic assessments. A recent study demonstrated the application of TurboFlow technology for monitoring metoprolol plasma concentrations in patients receiving 50 mg and 100 mg doses [8]. The method successfully quantified concentrations ranging from 3.56 to 50.81 μg/L in the 100 mg dose group, with a strong correlation (r = 0.992) between administered dose and plasma concentration. The automated sample preparation facilitated rapid processing of clinical samples, supporting high-throughput pharmacokinetic analysis.
Enantioselective Analysis
Metoprolol is administered as a racemic mixture, with the S-enantiomer possessing most of the β-blocking activity. Automated sample preparation coupled with chiral chromatography enables enantioselective determination of metoprolol and its metabolites in biological fluids [33]. Using a CHIRALCEL OD-RH column in reversed-phase mode, researchers achieved simultaneous determination of metoprolol and α-hydroxymetoprolol enantiomers in human urine with solid-phase extraction providing good extraction recoveries and minimal matrix interference.
Method Validation Parameters
Both TurboFlow and Online SPE methods for metoprolol analysis demonstrate excellent validation performance, as summarized in Table 2.
Table 2: Method Validation Parameters for Metoprolol Analysis
| Validation Parameter | TurboFlow Method [8] | Online SPE Method [62] |
|---|---|---|
| Linear Range | 5-1000 ng/L | 10-2560 ng/mL |
| Lower Limit of Quantification | 0.042 ng/L | 10 ng/mL |
| Precision (CV%) | < 10.28% | < 15% |
| Accuracy (Relative Error) | < 5.38% | Not specified |
| Matrix Effect | 89% | Not specified |
| Retention of Polar Compounds | Limited for highly polar compounds | Excellent (retains adrenaline) |
| Sample Throughput | 4.5 minutes per sample | 11 minutes per sample |
The Scientist's Toolkit
Table 3: Essential Research Reagents and Materials
| Item | Function/Application | Example Specifications |
|---|---|---|
| TurboFlow Cyclone-P Column | Online extraction of basic drugs like metoprolol from biological matrices | 50 × 0.5 mm; large particle size for size exclusion [8] |
| CAPCELL PAK MF Ph-1 SPE Column | Online SPE for polar β-blockers | 4.0 × 10 mm; retains polar compounds (log P ≤ 0.28) [62] |
| C18 Analytical Column | Chromatographic separation post-extraction | 50 × 2.1 mm, 1.9 µm or 4.6 × 150 mm, 5 µm [8] [62] |
| HyperSep Verify-CX SPE Cartridges | Offline mixed-mode SPE for comparative studies | 200 mg; 6 mL; for basic, acidic, and neutral fractions [64] |
| Formic Acid (0.1% v/v) | Mobile phase additive for improved ionization in MS | HPLC grade; in water and acetonitrile [8] |
| Ammonium Acetate Buffer | Mobile phase for online SPE methods | 10 mM, pH 4.0; for compound retention and separation [62] |
| Bisoprolol Fumarate | Internal standard for metoprolol quantification | IS for mass spectrometry; transition m/z 326.3 → 116.2 [8] |
Automated SPE platforms, particularly TurboFlow and Online SPE systems, represent significant advancements in sample preparation technology for pharmaceutical analysis. For metoprolol research, these technologies enable direct injection of complex biological samples, streamlined workflows, and improved data quality through reduced matrix effects. The choice between TurboFlow and Online SPE depends on specific application requirements: TurboFlow offers superior matrix removal for complex samples, while Online SPE provides better retention of polar compounds. Both technologies substantially increase laboratory efficiency and represent valuable tools for high-throughput bioanalysis in pharmaceutical development and clinical research.
Within pharmaceutical research, the selection of an optimal sample preparation technique is a critical determinant of the success and reliability of bioanalytical results. This application note provides a comparative analysis of three fundamental extraction methods—Solid-Phase Extraction (SPE), Liquid-Liquid Extraction (LLE), and Protein Precipitation (PP)—framed within the context of research on metoprolol tartrate tablet analysis. Efficient sample cleanup is indispensable for the accurate quantification of active pharmaceutical ingredients (APIs) and metabolites in complex biological matrices such as plasma or serum. The choice of extraction methodology directly impacts key analytical performance parameters, including recovery, sensitivity, selectivity, and throughput [65] [66]. This document outlines detailed protocols and a data-driven comparison to guide researchers and drug development professionals in selecting the most appropriate technique for their specific analytical challenges.
Theoretical Background and Principles of Extraction
Solid-Phase Extraction (SPE)
SPE is a sample preparation technique that separates analytes from a liquid matrix based on their affinity for a solid stationary phase. The process involves passing the sample through a cartridge or well containing a sorbent, where the target compounds are retained. After a washing step to remove undesired matrix components, the analytes are eluted with a stronger solvent [67] [66]. The mechanism of retention can be non-polar (e.g., C18-bonded silica for reversed-phase), polar, or ion-exchange, offering high selectivity [66]. Its advantages include cleaner extracts, higher selectivity, reduced solvent consumption, avoidance of emulsion formation, and easier automation compared to LLE [67] [66].
Liquid-Liquid Extraction (LLE)
LLE is a traditional extraction method based on the differential solubility of an analyte between two immiscible liquids, typically an aqueous sample and an organic solvent [66]. The distribution of the analyte follows the Nernst Distribution law, and its efficiency is governed by the partition coefficient [67]. Factors such as the choice of solvent, pH of the aqueous phase (which controls the ionization state of the analyte), and the phase ratio are crucial for achieving high recovery [67] [66]. While LLE is a well-established and simple technique, it can be labor-intensive, consume large volumes of solvents, and is prone to emulsion formation [67] [66].
Protein Precipitation (PP)
PP is the simplest sample preparation procedure, primarily used to remove proteins from biological fluids like plasma or serum. It involves adding a miscible organic solvent (e.g., acetonitrile, methanol, or acetone) or an acid to the sample [65] [68]. This denatures and precipitates the proteins, which are then removed by centrifugation. The supernatant, containing the analyte, can be directly analyzed or further processed. PP is characterized by its simplicity, low cost, and minimal method development requirements [65]. However, it offers limited sample cleanup and can leave behind many interfering matrix components [65] [68].
Comparative Experimental Data
The following tables summarize quantitative data and characteristics from various studies, providing a basis for comparing the three extraction techniques.
Table 1: Comparison of Extraction Method Performance for Various Analytes
| Analyte | Extraction Method | Recovery (%) | Matrix Effect | Key Findings | Source |
|---|---|---|---|---|---|
| Peptide Drugs (Somatostatin, GLP-2, Insulin, Liraglutide) | PP (ACN, 3:1 solvent:sample) | >50% (all parents & catabolites) | Significant | Highest overall recovery among tested PP/SPE protocols | [68] |
| Peptide Drugs (Somatostatin, GLP-2, Insulin, Liraglutide) | SPE (Mixed-mode Anion Exchange) | >20% (all parents & catabolites) | Lower than PP | Cleaner extracts with lower matrix effect; only sorbent to extract all peptides | [68] |
| Oligonucleotides (ASOs, siRNAs) | Enhanced PP (EPP with ammonia) | >80% | N/R | Overcomes coprecipitation issue; LLOQ of 1–5 ng/mL | [69] |
| Urinary Morphine | SPE-HPTLC | 74% (positive detection rate) | N/R | Higher detection rate compared to LLE-TLC | [66] |
| Urinary Morphine | LLE-TLC | 48% (positive detection rate) | N/R | Lower efficiency compared to SPE-HPTLC | [66] |
Table 2: General Characteristics and Applicability of Extraction Methods
| Parameter | Solid-Phase Extraction (SPE) | Liquid-Liquid Extraction (LLE) | Protein Precipitation (PP) |
|---|---|---|---|
| Principle | Adsorption onto solid sorbent [66] | Partitioning between immiscible liquids [66] | Protein denaturation with solvent/acid [65] |
| Selectivity | High (choice of sorbent chemistry) [67] | Moderate (choice of solvent & pH) [67] | Low |
| Typical Solvent Consumption | Low [67] [66] | High [67] | Moderate to High |
| Risk of Emulsion | Low [67] [66] | High [67] | Moderate |
| Automation Potential | High (off-line and on-line) [67] [8] | Moderate (challenging for large volumes) [67] | High |
| Best For | High-purity extracts, trace analysis, complex matrices [67] [68] | Non-polar analytes, high-capacity samples [67] | High-throughput, robust analytes, simple cleanup [65] |
| Cost Driver | Cartridge/plate consumables [67] | Solvent volume and disposal [67] | Solvent volume |
Detailed Experimental Protocols
Protocol: Solid-Phase Extraction for Basic Drugs (e.g., Metoprolol)
This protocol is adapted for the extraction of basic drugs like metoprolol from plasma, using a reversed-phase or mixed-mode sorbent [8] [66].
Research Reagent Solutions:
- SPE Sorbent: C18 or mixed-mode cationic exchange (MCX) cartridges (e.g., 300 mg/3 mL) [66].
- Conditioning Solvent: Methanol (2 x 3 mL) [66].
- Equilibration Solvent: Deionized water (2 x 3 mL) [66].
- Wash Solution 1: Water or a mild buffer (e.g., 2% formic acid) to remove interferences (2 x 3 mL) [66].
- Wash Solution 2: Methanol/water mixture (e.g., 50:50, v/v) to remove residual salts and polar impurities (2 x 3 mL).
- Elution Solvent: A volatile organic solvent with a basic modifier, e.g., Methanol:Ammonia (9:1, v/v) (2 mL) [66].
Procedure:
- Conditioning: Load 2 x 3 mL of methanol to the SPE column at a flow rate not exceeding 2 mL/min. Do not let the sorbent bed run dry [66].
- Equilibration: Load 2 x 3 mL of deionized water at a flow rate of 2 mL/min [66].
- Sample Loading: Apply the pre-treated (e.g., acid-hydrolyzed and pH-adjusted) plasma or urine sample to the column. A flow rate of 1 mL/min is recommended for efficient binding [66].
- Washing: Pass 2 x 3 mL of Wash Solution 1 (water/buffer) through the column at 2 mL/min, followed by 2 x 3 mL of Wash Solution 2 (methanol/water). This step removes unwanted matrix components [66].
- Drying: Dry the sorbent bed under vacuum (e.g., 10 in.Hg) for ~5 minutes to remove residual water, which can interfere with elution [66].
- Elution: Collect the analyte by passing 2 mL of the elution solvent (Methanol:Ammonia) through the column without applying vacuum [66].
- Reconstitution: Evaporate the eluate to dryness under a gentle stream of nitrogen. Reconstitute the dry residue in 100-200 µL of a mobile phase-compatible solvent (e.g., 50% methanol/0.2% formic acid) for LC-MS/MS analysis [65] [66].
Protocol: Liquid-Liquid Extraction for Metoprolol Complexation
This protocol is based on a spectrophotometric method for determining metoprolol tartrate via complexation with Cu(II) ions [19].
Research Reagent Solutions:
Procedure:
- Sample Pre-treatment: To 20 mL of urine or a diluted plasma sample, add 1 mL of concentrated hydrochloric acid. Heat the mixture for 15 minutes at 100°C to hydrolyze conjugates. After cooling, adjust the pH to 8-9 using concentrated ammonia solution [66].
- Complex Formation: Transfer an aliquot of the sample (containing 8.5-70 µg of metoprolol) to a flask. Add 1 mL of Britton-Robinson buffer (pH 6.0) and 1 mL of 0.5% CuCl₂ solution. Mix well and heat for 20 minutes in a water bath at 35°C to form the blue metoprolol-Cu(II) complex. Cool rapidly [19].
- Extraction: Add 2 x 15 mL of the chloroform-isopropanol (8:2) mixture to the solution. Shake vigorously for several minutes to partition the complex into the organic phase [66].
- Phase Separation: Allow the mixture to settle or use centrifugation to achieve complete separation of the organic and aqueous layers.
- Collection & Evaporation: Carefully separate the upper organic phase containing the analyte. Combine the organic layers and evaporate them to dryness under a stream of nitrogen [66].
- Reconstitution: Reconstitute the dry residue in an appropriate solvent (e.g., methanol or mobile phase) for subsequent analysis by TLC or spectrophotometry [19] [66].
Protocol: Protein Precipitation for Serum/Plasma Samples
This is a generic, high-recovery protocol for small molecules and peptides from serum or plasma [65] [68].
Research Reagent Solutions:
Procedure:
- Sample Mixing: Add 100 µL of serum or plasma sample to a microcentrifuge tube.
- Precipitation: Add 300 µL of acetonitrile (a 3:1 solvent-to-sample ratio) to the sample [65] [68]. Vortex mix vigorously for 1-2 minutes to ensure complete protein denaturation and precipitation.
- Centrifugation: Centrifuge the mixture at high speed (e.g., 12,000-15,000× g) for 10 minutes to pellet the precipitated proteins [65].
- Supernatant Collection: Carefully collect the clear supernatant, avoiding disturbance of the protein pellet.
- Analysis: The supernatant can be diluted 1:1 with 0.4% formic acid (final composition: 50% methanol/0.2% formic acid) and directly injected into an LC-MS/MS system [65]. Alternatively, it can be evaporated and reconstituted in a smaller volume to pre-concentrate the analyte.
Workflow and Decision Diagram
The following diagram visualizes the operational workflows for the three extraction methods and outlines a logical decision path for selecting the most appropriate technique.
Extraction Method Selection Workflow
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for Extraction Protocols
| Reagent/Solution | Primary Function in Extraction | Example Use Case |
|---|---|---|
| C18 / Mixed-Mode SPE Sorbents | Retains analytes via hydrophobic/ionic interactions; enables high-selectivity cleanup. | Extracting basic drugs like metoprolol from plasma [8] [66]. |
| Acetonitrile (ACN) & Methanol | Organic solvents for protein denaturation (PP) and elution in SPE. | Precipitating serum proteins in a 3:1 ratio with sample [65] [68]. |
| Chloroform-isopropanol mixture | Organic solvent pair for LLE; facilitates partitioning of non-polar analytes/complexes. | Extracting the metoprolol-Cu(II) complex for spectrophotometric detection [19] [66]. |
| Ammonia Solution | pH adjustment agent (basic) and elution modifier; deprotonates basic analytes. | Adjusting urine pH for LLE; eluting basic drugs from mixed-mode SPE [19] [66]. |
| Britton-Robinson Buffer | Maintains optimal pH for chemical reactions (e.g., complex formation). | Complexation of metoprolol with Cu(II) ions at pH 6.0 [19]. |
| Formic Acid | Mobile phase additive; provides protons for positive ionization in LC-MS. | Reconstituting samples for LC-MS analysis (e.g., 0.1-0.2% in solvent) [65] [8]. |
The comparative analysis presented in this application note demonstrates that the choice between SPE, LLE, and PP is not a one-size-fits-all decision but a strategic one based on the specific requirements of the metoprolol research project. SPE is the most powerful technique for achieving high-quality, sensitive results where sample cleanliness is paramount, such as in complex pharmacokinetic studies requiring low limits of quantification [8] [66]. LLE offers a robust and effective alternative, particularly for less complex matrices or when capital cost is a greater concern than solvent consumption [67]. PP remains the go-to method for high-throughput scenarios where speed and simplicity are prioritized over extensive sample cleanup [65] [68]. By aligning the strengths of each method with the analytical goals, researchers can optimize their workflow to generate reliable, reproducible, and meaningful data for drug development.
Metoprolol, a selective β1-adrenergic receptor blocker, is a cornerstone in the treatment of cardiovascular diseases including hypertension, angina pectoris, and heart failure [70] [71]. The drug is commonly administered as a racemic mixture, with the (S)-(−)-enantiomer possessing significantly higher β-adrenergic receptor affinity compared to its (R)-(+)-antipode [72]. Therapeutic Drug Monitoring (TDM) of metoprolol is clinically essential due to its narrow therapeutic index, significant interindividual variability in metabolism primarily via the CYP2D6 enzyme system, and the stereoselective differences in pharmacokinetics and pharmacodynamics between its enantiomers [72]. This document presents detailed application notes and protocols for the analysis of metoprolol in two critical matrices: patient plasma (for clinical TDM and pharmacokinetic studies) and pharmaceutical tablet dosage forms (for quality control). The protocols are framed within a broader thesis research context emphasizing Solid-Phase Extraction (SPE) cleanup for the analysis of metoprolol tartrate, detailing specific methodologies adapted for different analytical needs and instrumentation.
Analytical Techniques for Metoprolol Monitoring
The analysis of metoprolol in complex matrices requires highly selective and sensitive analytical techniques. The choice of method depends on the specific application, required sensitivity, and available instrumentation.
Table 1: Comparison of Analytical Methods for Metoprolol Quantification
| Method | Application | Matrix | Sample Preparation | Key Analytical Parameters | Ref. |
|---|---|---|---|---|---|
| HPLC-Fluorescence with SPE | Pediatric Drug Monitoring | Plasma | SPE on specific cartridges | LOD: 2.4 ng/mL; Recovery: 73.0 ± 20.5%; LOQ: 2.4 ng/mL | [10] |
| Chiral LC-ESI-MS/MS | Enantioselective Pharmacokinetics | Plasma | SPE (Lichrosep DVB HL) | Linear Range: 0.5-500 ng/mL; Recovery: >94%; Runtime: 7.0 min | [72] |
| RP-HPLC-UV | Pharmaceutical Dosage Form Analysis | Tablet | Dissolution in water, filtration | Linear Range: 5-15 μg/mL; Correlation (R²): 0.99994; Runtime: 6 min | [73] |
| Spectrophotometry | Pharmaceutical Dosage Form Analysis | Tablet | Complexation with Cu(II) | Linear Range: 8.5-70 μg/mL; Wavelength: 675 nm; LOD: 5.56 μg/mL | [6] |
Experimental Protocols
Protocol 1: SPE and HPLC-Fluorescence Detection for Pediatric Plasma Monitoring
This protocol is optimized for sensitive determination of total metoprolol in small-volume pediatric plasma samples [10].
Methodology
- Sample: 500 μL of human plasma.
- Internal Standard: Use an appropriate internal standard.
- Extraction: Utilize Solid-Phase Extraction columns.
- Chromatography:
- Column: Spherisorb C6 (5 μm particle size).
- Mobile Phase: 30% Acetonitrile and 70% 0.25 M Potassium Acetate Buffer (pH 4.0).
- Flow Rate: 1.0 mL/min.
- Temperature: Ambient.
- Detection: Fluorimetric detection with excitation at 225 nm and emission at 310 nm.
Method Performance
- The method demonstrates a limit of quantitation (LOQ) of 2.4 ng/mL, suitable for monitoring therapeutic levels.
- Metoprolol recovery from plasma is 73.0 ± 20.5%.
- Precision values for intra- and inter-assay are below 15.5%.
Protocol 2: Chiral LC-ESI-MS/MS for Enantioselective Plasma Analysis
This protocol enables high-throughput, sensitive separation and quantification of metoprolol enantiomers in human plasma for advanced pharmacokinetic studies [72].
Methodology
- Sample: 200 μL of human plasma.
- Internal Standard: rac-metoprolol-d6.
- Extraction: Solid-Phase Extraction on Lichrosep DVB HL cartridges.
- Chromatography:
- Column: Chiral Lux Amylose-2 (250 mm × 4.6 mm, 5 μm).
- Mobile Phase: 15 mM Ammonium Acetate in Water (pH 5.0) and 0.1% (v/v) Diethylamine in Acetonitrile (50:50, v/v).
- Run Time: 7.0 minutes.
- Detection: LC-ESI-MS/MS in positive ionization mode with Multiple Reaction Monitoring (MRM).
- Precursor→Product Ion Transitions: Monitored for each enantiomer and the IS.
Method Performance
- The method is validated over the concentration range of 0.500–500 ng/mL for both (S)-(−)- and (R)-(+)-metoprolol.
- The mean extraction recovery is greater than 94.0% for both enantiomers.
- The method is successfully applied to clinical studies and demonstrates assay reproducibility via incurred sample reanalysis.
Protocol 3: RP-HPLC-UV for Tablet Dosage Form Analysis
This protocol provides a simple, accurate, and robust method for the routine quality control analysis of metoprolol succinate in pharmaceutical formulations [73].
Methodology
- Standard Preparation: Accurately weigh 25 mg of Metoprolol Succinate reference standard into a 25 mL volumetric flask. Dissolve and dilute to volume with water to obtain a 1000 μg/mL stock solution. Further dilute to obtain working standards in the range of 5–15 μg/mL.
- Sample Preparation: Weigh and powder 20 tablets. Transfer a portion of the powder equivalent to about 50 mg of metoprolol succinate into a 50 mL volumetric flask. Add about 35 mL of water, sonicate for 10 minutes, cool, and dilute to volume with water. Filter through a 0.45 μm membrane filter, discard the first few mL of filtrate. Further dilute the filtrate appropriately with mobile phase to obtain a final concentration within the linear range.
- Chromatography:
- Column: Phenomenex C18 (250 mm × 4.6 mm, 5 μm).
- Mobile Phase: Methanol : 0.1% Orthophosphoric Acid in Water (60:40, v/v).
- Flow Rate: 1.0 mL/min.
- Injection Volume: 20 μL.
- Detection: UV at 222 nm.
- Run Time: 6 minutes.
- Calculation: Calculate the quantity of metoprolol succinate in the tablet sample by comparing the peak areas of the sample and standard solutions.
Method Validation
- The method demonstrates linearity in the range of 5–15 μg/mL with a correlation coefficient (R²) of 0.99994.
- Accuracy, confirmed by recovery studies, is 99.40%.
- The precision (RSD) is well below 2.0%.
- The Limit of Detection (LOD) and Limit of Quantitation (LOQ) are 0.142 μg/mL and 0.429 μg/mL, respectively.
Workflow and Signaling Visualization
Analytical Workflow for Metoprolol in Plasma and Tablets
The following diagram illustrates the overarching experimental workflow for the sample preparation and analysis of metoprolol in plasma and tablet dosage forms, highlighting the key steps from sample collection to quantitative analysis.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Key Research Reagent Solutions for Metoprolol Analysis
| Item | Function / Role | Application Context |
|---|---|---|
| Lichrosep DVB HL Cartridges | Solid-Phase Extraction for efficient and selective cleanup of analytes from complex plasma matrix. | Plasma Analysis [72] |
| Chiral Lux Amylose-2 Column | Chromatographically resolves metoprolol enantiomers for stereoselective pharmacokinetic studies. | Chiral Plasma Analysis [72] |
| Phenomenex C18 Column | Standard reverse-phase column for high-efficiency separation of metoprolol from excipients in tablets. | Tablet Dosage Form Analysis [73] |
| Ammonium Acetate Buffer | Provides pH control and compatible volatile buffer system for efficient ionization in LC-MS/MS. | Chiral LC-ESI-MS/MS [72] |
| Methanol & 0.1% OPA in Water | Simple isocratic mobile phase for robust and rapid analysis in quality control settings. | RP-HPLC-UV Analysis [73] |
| Copper(II) Chloride | Forms a colored complex with metoprolol for detection via spectrophotometry. | Spectrophotometric Tablet Analysis [6] |
The application notes and protocols detailed herein provide robust and validated methodologies for the analysis of metoprolol in both biological and pharmaceutical matrices. The SPE-based protocols for plasma analysis are critical for accurate Therapeutic Drug Monitoring and stereoselective pharmacokinetic profiling, addressing the challenges of low analyte concentrations and complex matrix effects. The HPLC method for tablet analysis offers a reliable solution for quality control in pharmaceutical development and manufacturing. Together, these protocols form a comprehensive analytical toolkit that supports ongoing research and ensures the efficacy and safety of metoprolol therapy.
Application Notes
The integration of Molecularly Imprinted Polymers (MIPs) and nanomaterial-enhanced sorbents represents a significant advancement in solid-phase extraction (SPE) techniques for pharmaceutical analysis. These materials address the critical need for highly selective and efficient sample cleanup, particularly for challenging matrices and low-concentration analytes. When applied to the analysis of metoprolol tartrate from tablet formulations, these advanced sorbents facilitate superior extraction efficiency, enhance analytical sensitivity, and ensure reliable quantification.
For the analysis of metoprolol tartrate, a beta-blocker used to treat cardiovascular diseases such as hypertension and angina [70], selective sample preparation is paramount. Molecularly Imprinted Polymers offer a tailored approach. MIPs are synthetic polymers possessing cavities that are sterically and chemically complementary to a target molecule, functioning as artificial antibodies [74]. The non-covalent imprinting protocol using methacrylic acid (MAA) as a functional monomer creates specific binding sites for amines, the class to which metoprolol belongs [75] [74]. This specificity allows MIP-SPE to isolate metoprolol tartrate effectively from tablet excipients and potential degradation products, providing a clean extract for downstream analysis and improving the accuracy of the results.
Concurrently, nanomaterials have revolutionized SPE by providing exceptionally high surface-area-to-volume ratios and unique physicochemical properties. Two prominent nanomaterials show particular promise for metoprolol tartrate:
- Multi-Walled Carbon Nanotubes (MWCNTs): Their large specific surface area and strong hydrophobic character make them excellent adsorbents for drugs like metoprolol tartrate [76]. MWCNTs can be incorporated as an interlayer in solid-contact ion-selective electrodes or directly as a sorbent to enhance electrical conductivity, stability, and adsorption capacity, leading to improved quantification limits [75].
- Nanosilver-functionalized Magnetic Nanoparticles (polyDOPA@Ag-MNPs): These materials combine the high enrichment capacity of silver nanoparticles with the operational simplicity of magnetic separation. The magnetic core allows for easy and rapid isolation of the sorbent from the sample solution using a magnet, simplifying the SPE procedure significantly [77] [56].
The synergy of MIPs and nanomaterials in SPE protocols delivers a powerful toolkit for researchers. It enables the development of robust, sensitive, and high-throughput analytical methods, which are essential for quality control in pharmaceutical manufacturing and drug development processes.
Table 1: Key Research Reagent Solutions for MIP and Nanomaterial SPE
| Reagent Name | Function/Description | Application in Protocol |
|---|---|---|
| Metoprolol Tartrate | Target analyte, model β-blocker drug. | The molecule of interest for extraction and quantification from tablet formulations. |
| Molecularly Imprinted Polymer (MIP) | Synthetic polymer with customized binding sites for metoprolol. | Sorbent in SPE cartridge for selective recognition and capture of metoprolol. |
| Methacrylic Acid (MAA) | Functional monomer for non-covalent imprinting. | Creates pre-polymerization complex with the template molecule during MIP synthesis. |
| Ethylene Glycol Dimethacrylate (EGDMA) | Cross-linking agent. | Creates a rigid polymeric structure around the template during MIP synthesis. |
| Azobisisobutyronitrile (AIBN) | Free-radical initiator. | Initiates the polymerization reaction in the synthesis of MIPs. |
| Multi-Walled Carbon Nanotubes (MWCNTs) | Nanomaterial sorbent with high surface area. | Enhances adsorption capacity and can be used to modify electrodes or as a sorbent. |
| PolyDOPA@Ag-MNPs | Silver-functionalized magnetic nanoparticles. | Magnetic sorbent for rapid extraction and enrichment of β-blockers via MSPE. |
| Britton-Robinson (BRB) Buffer | A universal buffer system. | Used to adjust and maintain the pH during the extraction or complex formation. |
Experimental Protocols
Protocol 1: Synthesis of Molecularly Imprinted Polymer (MIP) for Solid-Phase Extraction
This protocol details the synthesis of a metoprolol-selective MIP using non-covalent precipitation polymerization, optimized for SPE cartridge packing [75].
Principle: A pre-polymerization complex is formed between the template molecule (metoprolol) and functional monomer(s) in a porogenic solvent. Polymerization is initiated around this complex, and subsequent template removal leaves behind specific recognition sites.
Table 2: Reagents and Equipment for MIP Synthesis
| Category | Items |
|---|---|
| Reagents | Metoprolol tartrate (template), Methacrylic acid (MAA, functional monomer), Ethylene glycol dimethacrylate (EGDMA, cross-linker), Azobisisobutyronitrile (AIBN, initiator), Dimethylsulfoxide (DMSO, porogenic solvent). |
| Equipment | Round-bottom flask, Thermostatic water bath, Sonication bath, Soxhlet extraction apparatus, Vacuum oven, Filter funnel. |
Procedure:
- Pre-complex Formation: Dissolve 1 mmol of metoprolol tartrate and 4 mmol of methacrylic acid in 40 mL of DMSO in a glass-stoppered flask. Sonicate the mixture for 15 minutes to allow self-assembly of the complex.
- Polymerization Initiation: Add 25 mmol of EGDMA and 1 mmol of AIBN to the flask. Purge the mixture with nitrogen gas for 10 minutes to remove oxygen, which can inhibit free-radical polymerization.
- Polymerization: Place the sealed flask in a thermostatic water bath at 60°C for 24 hours to complete the polymerization reaction, resulting in a white precipitate.
- Washing: Filter the polymer and wash it sequentially with ethanol and water to remove unreacted components.
- Template Removal: Transfer the polymer to a Soxhlet apparatus. Extract continuously for 24 hours using a methanol and glacial acetic acid mixture (9:1 v/v) to leach out the embedded metoprolol template. Monitor the extraction process by UV-Vis to ensure complete template removal.
- Drying and Storage: Wash the extracted MIP with distilled water until neutral pH is achieved. Dry the polymer in a vacuum oven at 100°C. Store the finished MIP in a sealed container at room temperature.
- Control Polymer (NIP): Synthesize a non-imprinted polymer (NIP) following the same procedure but without the addition of the metoprolol tartrate template.
MIP Synthesis Workflow
Protocol 2: Nanomaterial-Enhanced Magnetic Solid-Phase Extraction (MSPE) of Metoprolol
This protocol utilizes polyDOPA@Ag-MNPs for the rapid and efficient extraction of metoprolol from processed tablet samples [77] [56].
Principle: Functionalized magnetic nanoparticles are dispersed in the sample solution, where they adsorb the target analyte. An external magnet is then used to separate the analyte-loaded nanoparticles from the solution matrix, after which the analyte is desorbed with a suitable solvent.
Table 3: MSPE Parameters for Metoprolol Extraction
| Parameter | Specification |
|---|---|
| Sorbent | polyDOPA@Ag-MNPs |
| Sorbent Amount | 4 mg |
| Sample Loading | Sonication for 2 minutes |
| Washing Solvent | Deionized water (3 times) |
| Elution Solvent | Methanol with 1% (v/v) Acetic Acid |
| Elution Volume | 1 mL |
| Elution Time | Sonication for 2 minutes |
Procedure:
- Sample Preparation: Crush and homogenize metoprolol tartrate tablets. Dissolve an accurately weighed powder amount equivalent to one tablet in deionized water or a suitable buffer (e.g., 10 mM phosphate buffer, pH 7.0). For complex samples, a deproteinization step with acetonitrile (1:1 ratio) followed by centrifugation may be necessary.
- MSPE Extraction: Disperse 4 mg of polyDOPA@Ag-MNPs into 1 mL of the prepared sample solution. Sonicate the mixture for 2 minutes to facilitate adsorption of metoprolol onto the nanoparticles.
- Magnetic Separation: Place the sample vial against a strong magnet. Wait for the nanoparticles to be collected at the vial wall (approximately 1-2 minutes). Carefully decant and discard the clear supernatant.
- Washing: To remove weakly adsorbed matrix components, resuspend the collected nanoparticles in 1 mL of deionized water. Separate the particles again using the magnet and discard the wash solution. Repeat this step three times.
- Elution: Add 1 mL of elution solvent (methanol containing 1% acetic acid) to the washed nanoparticles. Sonicate the mixture for 2 minutes to desorb metoprolol from the sorbent.
- Final Separation: Use the magnet to separate the nanoparticles. Carefully collect the resulting eluate and filter it through a 0.22 µm membrane filter. The filtrate is now ready for analysis via techniques such as HPLC or FTICR-MS.
MSPE Procedure Steps
Conclusion
Solid-phase extraction remains a powerful and versatile technique for the sample preparation of metoprolol tartrate, offering significant advantages in selectivity, reproducibility, and compatibility with modern analytical instrumentation. A well-developed SPE method, founded on a deep understanding of metoprolol's chemistry and optimized through systematic troubleshooting, is crucial for obtaining reliable data in pharmaceutical quality control and clinical monitoring. The future of SPE for cardiovascular drug analysis lies in the increased adoption of automation, computational design for sorbent selection, and the development of highly selective materials like molecularly imprinted polymers. These advancements promise to further enhance analytical throughput, sensitivity, and the ability to perform precise therapeutic drug monitoring, ultimately contributing to improved patient outcomes in cardiovascular disease management.
References
- 1. Metoprolol tartrate | 56392-17-7 [https://www.chemicalbook.com/ChemicalProductProperty_EN_CB7436617.htm]
- 2. Development of metoprolol tartrate extended-release ... [https://www.sciencedirect.com/science/article/pii/S0168365997001417#!]
- 3. Solid-phase extraction and direct high-performance liquid ... [https://www.sciencedirect.com/science/article/abs/pii/0378434791803257]
- 4. Modeling of the irradiation effect on some physicochemical ... [https://www.nature.com/articles/s41598-019-56805-0]
- 5. Comparative stability of repackaged metoprolol tartrate ... [https://www.sciencedirect.com/science/article/abs/pii/S0378517309007662]
- 6. Spectrophotometric Determination of Metoprolol Tartrate in ... [https://www.mdpi.com/1424-8247/4/7/964]
- 7. Solid Phase-Based Microextraction Techniques in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10142207/]
- 8. Investigation of Metoprolol Concentrations in Plasma Using ... [https://www.mdpi.com/2297-8739/11/11/306]
- 9. Guide To Solid Phase Extraction (SPE): Step-by- ... [https://www.alwsci.com/news/guide-to-solid-phase-extraction-spe-step-by-85168348.html]
- 10. HPLC quantification of metoprolol with solid-phase ... [https://pubmed.ncbi.nlm.nih.gov/15484221/]
- 11. Solid phase microextraction for quantitative analysis [https://www.sciencedirect.com/science/article/pii/S2772577422000465]
- 12. Using JMP Workflow Builder to Automate Analysis of an ... [https://community.jmp.com/t5/Abstracts/Using-JMP-Workflow-Builder-to-Automate-Analysis-of-an-Annual/ev-p/708940]
- 13. Understanding SPE Retention Mechanisms [https://www.biotage.com/blog/understanding-spe-retention-mechanisms]
- 14. Analysis of basic compounds by reversed-phase liquid ... [https://www.sciencedirect.com/science/article/abs/pii/S0021967307019966]
- 15. How It Works: Ion-Exchange SPE [https://www.chromatographyonline.com/view/how-it-works-ion-exchange-spe]
- 16. Ion Exchange SPE Fundamentals [https://www.restek.com/chromablography/the-foundation-for-separation-is-preparation-ion-exchange-spe-fundamentals?srsltid=AfmBOor31ebNj1uPWYSwJxGur84Bz02Bmpe48rf3d81OJPt2d8aTLQ_i]
- 17. ISOLUTE® HCX [https://www.biotage.com/isolute-hcx]
- 18. Mixed-Mode SPE Improves Extraction of Pharmaceutical ... [https://www.sigmaaldrich.com/US/en/technical-documents/technical-article/analytical-chemistry/solid-phase-extraction/mixed-mode-spe-improves?srsltid=AfmBOopZ_uj74vcmPj1XtbU-dqMR_ODwfMYwonoQKruxcR9yFbSK6YEl]
- 19. Spectrophotometric Determination of Metoprolol Tartrate in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4058672/]
- 20. metoprolol and (S)-α-hydroxymetoprolol in human plasma ... [https://www.sciencedirect.com/science/article/abs/pii/S0731708521003745]
- 21. SPE Method Development [https://www.thermofisher.com/us/en/home/industrial/chromatography/chromatography-sample-preparation/sample-preparation-consumables/solid-phase-extraction-consumables/spe-method-development.html]
- 22. Optimizing Elution Conditions To Improve SPE Performance [https://www.alwsci.com/news/optimizing-elution-conditions-to-improve-spe-p-85177371.html]
- 23. Emerging procedures and solvents in biological sample ... [https://www.sciencedirect.com/science/article/pii/S2772582023000165]
- 24. Sample Preparation for Biofluids [https://www.biotage.com/biofluids-sample-prep]
- 25. New techniques of on-line biological sample processing and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5071623/]
- 26. : Package Insert / Prescribing Information / MOA Metoprolol [https://www.drugs.com/pro/metoprolol.html]
- 27. Innovative Solid-Phase Extraction Strategies for Improving ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11124106/]
- 28. Optimising factors affecting solid phase extraction ... [https://www.sciencedirect.com/science/article/pii/S2405844021000396]
- 29. sciencedirect.com/topics/pharmacology-toxicology-and-pharmaceutical... [https://www.sciencedirect.com/topics/pharmacology-toxicology-and-pharmaceutical-science/metoprolol]
- 30. Back to Basics: The Role of pH in Retention and Selectivity [https://www.chromatographyonline.com/view/back-basics-role-ph-retention-and-selectivity]
- 31. Exploring the Role of pH in HPLC Separation [https://www.moravek.com/exploring-the-role-of-ph-in-hplc-separation/]
- 32. Metoprolol: Uses, Interactions, Mechanism of Action [https://go.drugbank.com/drugs/DB00264]
- 33. Enantioselective determination of metoprolol and its ... [https://www.sciencedirect.com/science/article/abs/pii/S157002321530194X]
- 34. Matrix effects and recovery calculations in analyses of ... [https://www.sciencedirect.com/science/article/abs/pii/S0021967312012459]
- 35. Development of an HPLC–UV assay method for the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5762929/]
- 36. A practical guide to sample preparation for liquid ... [https://www.spectroscopyeurope.com/article/practical-guide-sample-preparation-liquid-chromatography-tandem-mass-spectrometry-clinical]
- 37. LC-MS Sample Preparation [https://www.thermofisher.com/us/en/home/industrial/mass-spectrometry/mass-spectrometry-learning-center/liquid-chromatography-mass-spectrometry-lc-ms-information/lc-ms-sample-preparation.html]
- 38. Development SPE-LC-MS/MS method for determination of ... [https://www.nature.com/articles/s41598-025-04951-z]
- 39. HPLC–UV–ESI MS/MS identification of the color constituents ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4026629/]
- 40. kinetic spectrophotometric method for the determination of... Validated [https://pubmed.ncbi.nlm.nih.gov/16079525/]
- 41. SPE-LC-MS/MS optimisation using response surface ... [https://www.sciencedirect.com/science/article/pii/S0021967325004005]
- 42. Understanding and Improving Solid-Phase Extraction [https://www.chromatographyonline.com/view/understanding-and-improving-solid-phase-extraction-1]
- 43. The Most Common Mistakes in Solid-Phase Extraction [https://www.chromatographyonline.com/view/most-common-mistakes-solid-phase-extraction]
- 44. Common Problems in Solid-Phase Extraction: A Practical ... [https://www.welch-us.com/blogs/knowleage-base/common-problems-in-solid-phase-extraction-a-practical-troubleshooting-guide]
- 45. SPE Troubleshooting | Thermo Fisher Scientific - US [https://www.thermofisher.com/us/en/home/industrial/chromatography/chromatography-sample-preparation/sample-preparation-consumables/solid-phase-extraction-consumables/spe-troubleshooting.html]
- 46. 7 Tips to Improve Solid Phase Extraction (SPE) Recovery [https://www.promochrom.com/post/7-tips-to-improve-solid-phase-extraction-spe-recovery]
- 47. Three Most Common Problems Regard to Solid Phase ... [https://www.hawach.com/news/three-most-common-problems-regard-to-spe.html]
- 48. Why is it so important to air dry your SPE sorbent? [https://www.biotage.com/blog/why-does-your-spe-disk-need-to-be-dry]
- 49. Influence of the drying step within disk-based solid-phase ... [https://www.sciencedirect.com/science/article/abs/pii/S0021967316303703]
- 50. SPE - Optimize Your Solid Phase Extraction-Method - Wiki [https://community.agilent.com/technical/consumables/w/wiki/21631/spe---optimize-your-solid-phase-extraction-method]
- 51. SPE Cartridge Washing and Drying Parameters Selection [https://www.specartridge.com/spe-cartridge-washing-and-drying-parameters-selection/]
- 52. Metoprolol Tartrate - an overview [https://www.sciencedirect.com/topics/chemistry/metoprolol-tartrate]
- 53. Quality Control and Testing Evaluation of Various ... [https://www.jmchemsci.com/article_158097.html]
- 54. Optimization of bilayer floating tablet containing metoprolol ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2750284/]
- 55. Solid Phase Extraction Guide [https://www.thermofisher.com/us/en/home/industrial/chromatography/chromatography-sample-preparation/sample-preparation-consumables/solid-phase-extraction-consumables/spe-guide.html]
- 56. Rapid and sensitive analysis of trace β-blockers by magnetic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9091926/]
- 57. Validation of Analytical Methods: A Review [https://www.gavinpublishers.com/article/view/validation-of-analytical-methods-a-review]
- 58. Analytical Method Validation: Back to Basics, Part II [https://www.chromatographyonline.com/view/analytical-method-validation-back-basics-part-ii]
- 59. Analytical Method Validation: A Recipe for Accurate Results [https://certified-laboratories.com/blog/analytical-method-validation-a-recipe-for-accurate-results/]
- 60. The 6 Key Aspects of Analytical Method Validation [https://www.elementlabsolutions.com/uk/chromatography-blog/post/6-key-aspects-of-analytical-method-validation]
- 61. Key Parameters for Analytical Method Validation [https://altabrisagroup.com/analytical-method-validation-parameters/]
- 62. Comparison of two online extraction systems and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9078181/]
- 63. Automated LC-MS Sample Cleanup Using Turbulent Flow ... [https://www.thermofisher.com/blog/analyteguru/automated-lc-ms-sample-cleanup-using-turbulent-flow-chromatography/]
- 64. AI-64310-CRFT [https://apps.thermoscientific.com/media/SID/LSMS/flipbooks/app_note_compendium/files/assets/basic-html/page-339.html]
- 65. Comparison of different serum sample extraction methods ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4669428/]
- 66. Comparison of liquid-liquid extraction-thin layer ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3596733/]
- 67. Liquid-Liquid Extraction vs. Solid-Phase Extraction Methods [https://kjhil.com/liquid-liquid-extraction-vs-solid-phase-extraction/]
- 68. Comparison of different protein precipitation and solid ... [https://pubmed.ncbi.nlm.nih.gov/32633064/]
- 69. Enhanced Protein Precipitation with Ammonia Enables ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12529115/]
- 70. Uses, Side Effects & Warnings Metoprolol Tartrate [https://www.drugs.com/mtm/metoprolol-tartrate.html]
- 71. sciencedirect.com/topics/medicine-and-dentistry/ metoprolol - tartrate [https://www.sciencedirect.com/topics/medicine-and-dentistry/metoprolol-tartrate]
- 72. and (R)-(+)-metoprolol in human plasma by chiral LC–ESI– ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5761053/]
- 73. A Novel RP-HPLC Method for the Development ... [https://ajpaonline.com/HTML_Papers/Asian%20Journal%20of%20Pharmaceutical%20Analysis__PID__2025-15-4-7.html]
- 74. The Selectivity of Polymers Imprinted with Amines - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC6100134/]
- 75. A stability-indicating potentiometric platform for assaying Metoprolol ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11924619/]
- 76. Enhanced encapsulation of metoprolol tartrate with carbon ... [https://link.springer.com/article/10.1007/s13204-011-0030-3]
- 77. Rapid and sensitive analysis of trace β-blockers by ... [https://www.sciencedirect.com/science/article/pii/S2095177921001441]