The Evolution of Solid-Phase Extraction: From Historical Foundations to Modern Biomedical Applications
This article provides a comprehensive exploration of the early development and evolution of solid-phase extraction (SPE) techniques, tailored for researchers, scientists, and drug development professionals.
The Evolution of Solid-Phase Extraction: From Historical Foundations to Modern Biomedical Applications
Abstract
This article provides a comprehensive exploration of the early development and evolution of solid-phase extraction (SPE) techniques, tailored for researchers, scientists, and drug development professionals. It traces the foundational journey from initial concepts in the 1940s and 1950s using active carbon to the revolutionary introduction of pre-packed cartridges in the 1970s. The content covers core methodological principles, diverse sorbent chemistries, and configurations critical for modern applications. It further delivers practical troubleshooting guidance for common challenges and concludes with a comparative analysis of SPE performance against other techniques, validating its indispensable role in achieving high-quality, reproducible data in complex biomedical and clinical research.
The Foundational Era: Tracing the Origins and Key Milestones of SPE Technology
The period from the 1940s to the 1950s marked a transformative era for activated carbon, transitioning it from a specialized material with limited applications to a cornerstone of modern industrial and environmental processes. This "Age of Active Carbon" was characterized by pioneering experimental applications that exploited the material's exceptional adsorptive properties, laying the foundational principles for what would later be formalized as solid-phase extraction (SPE) techniques. While activated carbon itself had been known for centuries—with historical uses ranging from Egyptian preservatives to Bronze Age metal purification [1]—the mid-20th century witnessed its systematic application to address complex chemical separation challenges. The development of these early carbon-based methods represented a significant departure from traditional liquid-liquid extraction, offering improved efficiency and practicality that would eventually influence the creation of modern SPE protocols [2] [3].
This article examines the groundbreaking experimental applications of activated carbon during its formative years, with particular focus on its role in the evolution of sample preparation methodology. By exploring the technical protocols, material properties, and scientific contexts of this period, we aim to establish the historical continuity between these early carbon-based techniques and contemporary solid-phase extraction principles that now underpin much of analytical chemistry and drug development.
Historical and Technical Background
The Material: Understanding Activated Carbon
Activated carbon, also referred to as activated charcoal, is a porous form of carbon characterized by an exceptionally high surface area that enables it to control millions of cataloged chemicals through adsorption processes [1]. The material's structure consists of graphitic plates connected by carbon-carbon bonds, creating an extensive internal network of pores of varying sizes [4]. This microstructure provides the foundation for its adsorptive capacity, with one pound of carbon offering over six million square feet of surface area in some formulations [1].
The activation process, which involves treatment with carbon dioxide or super-heated steam at high temperatures, creates a matrix of tiny pores that increase surface area and create more sites for molecule trapping [1]. A critical distinction in the mechanism of action lies in the difference between adsorption and absorption: while absorption involves substances being taken up throughout the bulk of a material, adsorption refers to the adherence of molecules specifically to the internal and external surfaces of the carbon [4]. This surface-based interaction enables activated carbon to remove organic compounds from liquids and gases with remarkable efficiency.
The Pre-1940s Foundation
Prior to its systematic application in the mid-20th century, activated carbon had already accumulated a diverse history of uses. Evidence suggests that prehistoric humans may have used primitive forms of carbon to remove impurities from smelted metals during bronze manufacturing [1]. Ancient Egyptian papyri from around 1500 B.C. record the use of carbon for medicinal purposes, while around 400 B.C., ancient Hindu and Phoenician civilizations discovered its water-purification properties [1].
Significant milestones in the modern development of activated carbon included its use in gas masks during World War I to protect against toxic gases, though widespread commercial production and application only emerged after World War II [1]. The first coal-based activated carbon products were developed in the 1940s, marking the beginning of controlled, industrial-scale production and setting the stage for the experimental applications that would define the early "Age of Active Carbon" [4].
Experimental Applications and Methodologies (1940s-1950s)
The post-World War II era witnessed the systematic application of activated carbon across diverse fields, with researchers developing experimental protocols that would establish fundamental principles for separation science.
Environmental and Air Pollution Research
The emergence of severe air pollution issues in Los Angeles during the 1940s served as a catalyst for innovative applications of activated carbon in environmental research. Beginning in July 1943, Los Angeles experienced repeated "smog" episodes characterized by eye irritation, plant damage, and reduced visibility [5]. Unlike traditional coal-derived pollution, this photochemical smog exhibited distinct diurnal patterns, forming during daytime hours and dissipating at night.
To identify the causative agents responsible for plant damage, researchers at the California Institute of Technology (Caltech) and the Los Angeles County Air Pollution Control District (LACAPCD) established a comprehensive research program utilizing the state-of-the-art Earhart Plant Research Laboratory (Phytotron) [5]. The experimental methodology employed activated carbon as a purification medium:
- Air Filtration System Setup: Ambient air from Pasadena was drawn through activated carbon filters to remove pollutant species.
- Controlled Fumigation Experiments: Plants were grown under controlled conditions and exposed to both filtered and unfiltered air.
- Comparative Damage Assessment: Researchers documented the presence or absence of plant damage across different exposure scenarios.
The experiments demonstrated that plant damage could be eliminated when ambient air was first purified through activated carbon filters, providing crucial evidence that the harmful species were adsorbable contaminants [5]. This research represented one of the first systematic applications of activated carbon for the selective removal of unknown organic compounds from complex environmental samples.
Table 1: Key Experimental Applications of Activated Carbon (1940s-1950s)
| Application Area | Time Period | Experimental Purpose | Key Researchers/Institutions |
|---|---|---|---|
| Air Pollution Analysis | Late 1940s-1950s | Identify plant-damaging agents in photochemical smog | Caltech, LACAPCD |
| Water Treatment | Early 1960s (building on 1950s research) | Taste and odor removal from drinking water | Calgon Carbon Corporation |
| Military Protection | 1940s | Development of coal-based activated carbon for protective equipment | Calgon Carbon Corporation |
| Sugar Decolorization | 1950s | Industrial purification using "Pittsburgh Pulse Bed" system | Calgon Carbon Corporation |
Industrial and Water Treatment Applications
Parallel to environmental research, industrial applications of activated carbon advanced significantly during the 1950s. Calgon Carbon Corporation pioneered several industrial-scale processes, including the "Pittsburgh Pulse Bed" for sugar decolorization in the 1950s [4]. This system represented an early example of continuous-flow adsorption processes that would later influence solid-phase extraction cartridge design.
In the early 1960s, building directly on research from the previous decade, Calgon Carbon achieved the first use of granular activated carbon (GAC) for taste and odor removal from drinking water [4]. The experimental protocols developed for these applications established fundamental parameters for carbon-based purification, including:
- Contact time optimization between water and carbon
- Flow rate studies to maximize adsorption efficiency
- Capacity testing to determine carbon saturation points
- Reactivation methods to extend material lifespan
These industrial applications demonstrated the economic viability of activated carbon for large-scale separation processes and provided practical experience that would inform later laboratory-scale extraction techniques.
Emerging Solid-Phase Extraction Principles
While the term "solid-phase extraction" would not be formally established until later, the foundational principles of SPE began emerging through these early activated carbon applications. The first experimental uses of animal charcoal as an adsorbent in column-based separations during the 1940s represented a direct precursor to modern SPE [2]. These early carbon-based methods shared key characteristics with contemporary SPE:
- Selective retention of target compounds based on chemical affinity
- Interference removal through differential adsorption
- Sample purification prior to analysis
- Compound concentration through adsorption and elution
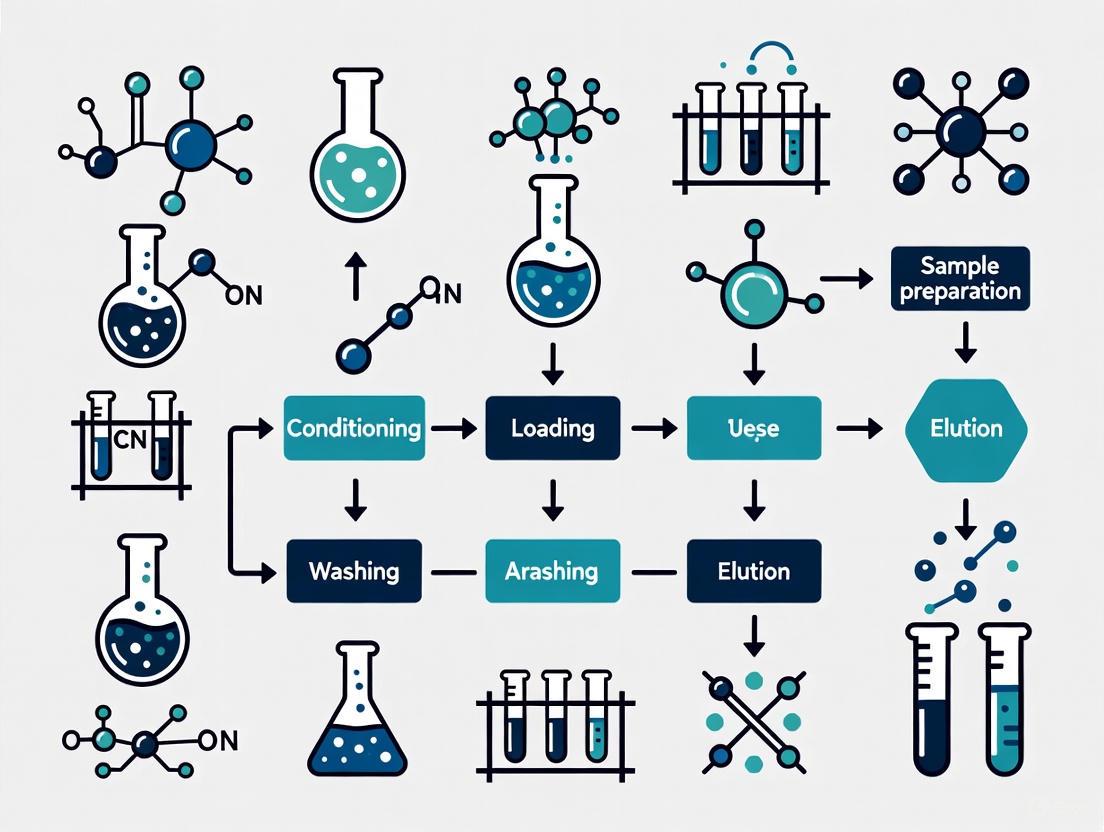
The methodology developed during this period established the basic workflow that would later be formalized in SPE protocols: sample pretreatment, column conditioning, sample loading, washing, and elution [6].
The Scientist's Toolkit: Key Research Reagents and Materials
The experimental work during the early "Age of Active Carbon" relied on a focused set of materials and reagents that defined the technological capabilities of the period.
Table 2: Essential Research Materials in Early Activated Carbon Applications
| Material/Reagent | Function | Specific Examples & Applications |
|---|---|---|
| Bituminous Coal-Based Carbon | Primary adsorbent with controlled pore structure | Calgon Carbon's granular products for water treatment [4] |
| Powdered Activated Carbon (PAC) | Fine-particle adsorbent for liquid-phase applications | FLUEPAC for mercury treatment (later development) [4] |
| Granular Activated Carbon (GAC) | Particle form for packed-bed adsorption systems | Taste and odor removal from drinking water [4] |
| Activated Carbon Filters | Air purification and contaminant removal | Caltech smog research filtration systems [5] |
| Pelletized Activated Carbon | Compressed cylinders for specialized applications | Volatile organic compound removal from gas streams [4] |
The predominance of coal-derived activated carbon during this period reflected both manufacturing capabilities and the limited understanding of alternative stationary phases. Unlike modern SPE with its diverse sorbent chemistries, early researchers worked primarily with unmodified carbon, relying on its inherent non-polar characteristics and extensive surface area for adsorption. The reusability of activated carbon through thermal reactivation processes represented an important economic consideration, with spent carbon being heated in oxygen-devoid furnaces using steam to restore adsorptive capacity [4].
Methodology: Detailed Experimental Protocols
This section reconstructs the core experimental methodologies that defined the application of activated carbon during the 1940s and 1950s, providing insight into the technical practices of the period.
Activated Carbon Filtration for Air Purification
The protocol developed at Caltech for identifying plant-damaging agents in smog exemplifies the systematic application of activated carbon in environmental research [5]:
Apparatus Setup:
- Construct a filtration chamber with sufficient volume for air flow rates supporting plant growth.
- Pack the chamber with granular activated carbon (typically bituminous coal-based, 50-100 mesh size).
- Install pre-filters to remove particulate matter that could clog the carbon bed.
System Conditioning:
- Purge the carbon bed with dry, clean air or nitrogen to remove manufacturing residues.
- Humidity the air stream to maintain appropriate conditions for plant health.
Sample Processing:
- Draw ambient air through the activated carbon bed at controlled flow rates (typically 5-15 L/min).
- Direct the filtered air to enclosed plant growth chambers.
- Maintain parallel control chambers receiving unfiltered ambient air.
Quality Assessment:
- Monitor plant health indicators including leaf damage, growth rates, and visible lesions.
- Conduct comparative analysis between filtered and unfiltered exposures.
- Periodically test carbon efficiency by measuring breakthrough of known contaminants.
This methodology confirmed that activated carbon could effectively remove the causative agents of plant damage, providing critical evidence for the organic chemical nature of photochemical smog constituents [5].
Early Column-Based Extraction Using Carbon
The use of activated carbon in column configurations for liquid extraction established foundational principles for modern SPE [2]:
Column Preparation:
- Pack glass columns with slurry-activated carbon in water or organic solvent.
- Use column diameters ranging from 1-5 cm with bed heights of 10-30 cm.
- Place glass wool or sintered glass frits at both ends to retain carbon particles.
Column Conditioning:
- Pre-wet the carbon bed with 5-10 bed volumes of the intended sample solvent.
- Ensure no air bubbles remain in the column bed that could cause channeling.
Sample Loading:
- Adjust sample pH if necessary to optimize compound retention.
- Apply sample to the column at controlled flow rates (1-5 mL/min for laboratory-scale columns).
- Collect effluent for analysis if monitoring compound breakthrough.
Wash and Elution:
- Wash with 2-5 bed volumes of solvent matching sample matrix to remove weakly adsorbed interferents.
- Elute retained compounds with stronger solvents (e.g., methanol, acetone, or acetonitrile).
- Use stepwise or gradient elution for complex mixtures.
Diagram 1: Early Carbon Column Extraction Workflow. This diagram illustrates the sequential steps for solid-phase extraction using activated carbon in column configurations, as practiced in the 1940s-1950s.
Technological Impact and Evolution to Modern SPE
The experimental applications of activated carbon during the 1940s and 1950s established fundamental principles that would directly influence the development of modern solid-phase extraction techniques. The demonstrated effectiveness of carbon-based adsorption for selective compound isolation from complex matrices provided a practical alternative to liquid-liquid extraction, offering reduced solvent consumption, simpler apparatus, and potential for automation [2] [3].
The transition from these early carbon methods to contemporary SPE occurred through several key developments:
Introduction of Standardized Formats: The 1970s saw the introduction of pre-filled cartridges containing silica sorbents, making the procedure more convenient and reproducible [2]. The first commercial Sep Pak C18 cartridge in 1977 represented a direct evolution from the packed carbon columns of the previous decades.
Diversification of Sorbent Chemistry: While early work relied almost exclusively on activated carbon, modern SPE incorporated reversed-phase, normal-phase, ion-exchange, and mixed-mode sorbents to address a wider range of analytical challenges [2] [6].
Miniaturization and Automation: The development of 96-well plate formats in the 1980s and pipette-tip SPE enabled higher throughput processing of smaller sample volumes, building on the fundamental principles established with carbon columns [2].
Table 3: Comparative Analysis: Early Activated Carbon vs. Modern SPE Techniques
| Parameter | 1940s-1950s Activated Carbon Methods | Modern Solid-Phase Extraction |
|---|---|---|
| Primary Sorbents | Animal charcoal, coal-based activated carbon | C18, silica, ion-exchange, polymer-based, mixed-mode |
| Common Formats | Packed glass columns, filter systems | Cartridges, 96-well plates, pipette tips, disks |
| Scale | Large sample volumes (50mL-1L) | Wide range (μL to mL volumes) |
| Mechanistic Understanding | Empirical observations, limited theoretical framework | Well-established principles of adsorption chromatography |
| Application Scope | Limited to non-polar/medium polarity compounds | Comprehensive range from non-polar to ionic compounds |
| Throughput | Low (manual processing of few samples) | High (automation of hundreds of samples) |
Despite these advancements, the fundamental adsorption principles demonstrated in the early activated carbon applications remain central to modern sample preparation. The "bind and elute" and "removal/trapping" strategies formalized in contemporary SPE [3] directly mirror the approaches pioneered with activated carbon columns in the mid-20th century.
The experimental applications of activated carbon during the 1940s and 1950s represent a critical chapter in the history of separation science, forming a conceptual and methodological bridge between traditional extraction techniques and modern solid-phase extraction. The pioneering work in environmental analysis, industrial processing, and laboratory-scale purification established the fundamental adsorption principles, column-based formats, and methodological workflows that would later be refined into contemporary SPE protocols.
For today's researchers and drug development professionals, understanding this historical foundation provides valuable context for current sample preparation techniques. The evolution from empirical carbon-based methods to highly specialized SPE sorbents and formats demonstrates how fundamental materials science, when coupled with innovative experimental applications, can transform analytical capabilities across diverse fields. The early "Age of Active Carbon" thus stands not as a historical curiosity, but as a formative period that established core principles still relevant in modern laboratories.
Within the framework of research on the early development of solid-phase extraction (SPE) techniques, understanding the foundational work of chromatography's pioneers is not merely historical but fundamentally practical. The principles established by Mikhail Tsvet and later refined by Archer Martin and Richard Synge form the essential theoretical and operational bedrock upon which modern sample preparation and separation science is built. This article traces the influential contributions of these key figures, demonstrating how their work on column and partition chromatography directly enabled the creation of SPE. It provides a detailed technical examination of their methodologies, illustrating the direct lineage to contemporary SPE protocols used by researchers and drug development professionals today. The separation of complex mixtures is a prerequisite for accurate analysis in pharmaceutical science, and the evolution from Tsvet's color bands to today's high-throughput SPE disks represents a continuous refinement of core chromatographic principles.
The Foundational Work of Mikhail Tsvet
Historical Context and Experimental Breakthrough
At the turn of the 20th century, Mikhail Tsvet, a Russian-Italian botanist, sought to separate and study the complex pigments found in plants [7] [8]. His innovative solution, developed around 1900, was the invention of column chromatography [9] [10]. In his seminal experiments, Tsvet used a vertical glass column packed with a solid adsorbent—calcium carbonate—through which he passed a plant extract dissolved in an organic solvent, such as petroleum ether combined with ethanol [7] [8]. As the solution moved through the column, the different plant pigments, including chlorophylls and carotenoids, separated into distinct, colored bands [7]. This powerful visual demonstration of separation led Tsvet to name the technique "chromatography," from the Greek words chroma (color) and graphein (to write) [7] [8]. Tragically, due to factors including global unrest and the language barrier of his initial publications, Tsvet's work remained largely unrecognized for several decades, delaying widespread adoption of his technique [8].
Detailed Experimental Protocol: Tsvet's Column Chromatography
The following protocol reconstructs the key methodology employed by Tsvet for the separation of plant pigments.
- Objective: To separate and isolate the various pigment components from a leaf extract using adsorption column chromatography.
- Principle: Separation is achieved based on the differential adsorption of the mixture's components onto a solid stationary phase.
Materials and Reagents:
| Research Reagent/Material | Function in the Experiment |
|---|---|
| Calcium Carbonate (CaCO₃) | Solid stationary phase/adsorbent; provides the surface for differential adsorption of pigments. |
| Glass Column | Housing for the stationary phase; allows visual observation of band separation. |
| Plant Leaf Extract (e.g., spinach) | Source of the target analyte mixture (chlorophylls, carotenoids). |
| Petroleum Ether & Ethanol | Organic solvents acting as the mobile phase to carry the extract through the column. |
Procedure:
- Column Preparation: Pack a clean, dry glass column with finely powdered calcium carbonate. Ensure the packing is uniform to prevent channeling.
- Sample Application: Gently apply the concentrated plant extract, dissolved in a minimal volume of petroleum ether, to the top of the calcium carbonate bed.
- Elution and Separation: Allow the sample to adsorb onto the top layer of the adsorbent. Then, carefully pass a mixture of petroleum ether and ethanol through the column via gravity flow. The mobile phase is often contained in a reservoir attached to the top of the column.
- Observation: As the mobile phase traverses the column, observe the formation of distinct colored bands corresponding to separated pigments (e.g., green chlorophylls and yellow-orange carotenoids) [7].
- Fraction Collection (if intended for isolation): Once the bands are fully separated, the column may be extruded, and the individual bands physically excised, or the pigments may be eluted separately by continued solvent washing.
Underlying Mechanism: Tsvet correctly identified the mechanism as adsorption, where the pigments were retained to different degrees on the solid calcium carbonate surface based on their chemical properties [7]. The stronger the adsorption, the slower the pigment migrated down the column.
Technical Visualization: Tsvet's Column Workflow
The following diagram illustrates the logical workflow and core components of Tsvet's column chromatography experiment.
The Partition Chromatography Revolution by Martin and Synge
Advancing the Theoretical Foundation
The next pivotal advancement in chromatography was made by Archer John Porter Martin and Richard Laurence Millington Synge in the early 1940s. While Tsvet's work leveraged solid-liquid adsorption, Martin and Synge introduced liquid-liquid partition chromatography [7] [11]. Their key insight was to use a solid support (like silica gel or filter paper) to hold one liquid stationary, while a second, immiscible liquid mobile phase flowed past it [7]. Separation occurred based on the differential partition of solutes between the two liquid phases, governed by their distinct partition coefficients [7] [12]. This principle was elegantly summarized by the partition coefficient (Kd), where Kd = Concentration of solute in stationary phase (Cs) / Concentration of solute in mobile phase (Cm) [12]. For their groundbreaking work, which made the efficient separation of delicate biochemicals like amino acids possible, they were awarded the Nobel Prize in Chemistry in 1952 [11].
Detailed Experimental Protocol: Martin and Synge's Partition Chromatography
This protocol outlines their method for separating amino acids, a cornerstone of biochemical analysis.
- Objective: To separate a mixture of amino acids using partition chromatography on a silica gel column.
- Principle: Separation is achieved based on the differential partition of analytes between a water-based stationary phase immobilized on silica gel and a water-saturated organic mobile phase.
Materials and Reagents:
| Research Reagent/Material | Function in the Experiment |
|---|---|
| Silica Gel | Inert solid support for immobilizing the aqueous stationary phase. |
| Water (H₂O) | Stationary phase; analytes partition between this and the mobile phase. |
| Chloroform & n-Butanol | Organic solvents composing the mobile phase, saturated with water. |
| Amino Acid Mixture | Target analytes for separation. |
| Methyl Red | Indicator used to visualize the movement of the amino acid zones in the column. |
Procedure:
- Stationary Phase Preparation: Impregnate the silica gel with an aqueous solution to form the stationary phase. The gel must be thoroughly mixed with water to ensure it is uniformly saturated.
- Column Packing: Pack the water-laden silica gel into a chromatographic column. Care must be taken to avoid drying the gel, which would disrupt the stationary phase.
- Equilibration and Sample Loading: Pre-equilibrate the column with the water-saturated organic mobile phase (e.g., chloroform/n-butanol). Apply the amino acid mixture, dissolved in a small volume of the mobile phase, to the top of the column.
- Elution and Development: Continuously pass the water-saturated organic mobile phase through the column. The amino acids will travel down the column at different rates determined by their partition coefficients between the immobilized water and the moving organic solvent [7].
- Detection: Since amino acids are colorless, an indicator like methyl red was added to the silica gel. As the amino acids migrated, they would displace the indicator, creating visible zones that could be tracked [7].
- Analysis: Upon completion, the column could be extruded, and the segments containing the separated amino acids could be cut out for further analysis or elution.
This work directly led to the development of paper chromatography by the same researchers, where filter paper served as the support for the stationary water phase [7].
Technical Visualization: The Partition Principle
The diagram below contrasts the adsorption mechanism of Tsvet with the partition mechanism pioneered by Martin and Synge, highlighting the fundamental conceptual shift.
Quantitative Comparison of Foundational Techniques
The following table provides a structured, quantitative comparison of the key parameters and components of the chromatographic methods developed by Tsvet and Martin & Synge, underscoring their distinct approaches and contributions.
Table: Quantitative Comparison of Foundational Chromatographic Techniques
| Parameter | Tsvet's Adsorption Chromatography | Martin & Synge's Partition Chromatography |
|---|---|---|
| Primary Mechanism | Solid-Liquid Adsorption | Liquid-Liquid Partition |
| Stationary Phase | Calcium Carbonate (Solid) | Water immobilized on Silica Gel (Liquid) |
| Mobile Phase | Petroleum Ether, Ethanol (Organic Solvent) | Water-saturated Chloroform/n-Butanol |
| Key Separation Factor | Differential Adsorption to Solid Surface | Partition Coefficient (Kd) between two liquids |
| Typical Analytes | Plant Pigments (e.g., Chlorophyll, Carotenoids) | Biochemically Relevant Molecules (e.g., Amino Acids) |
| Visualization Method | Native Color of Bands | Chemical Indicator (e.g., Methyl Red) |
The Direct Lineage to Solid-Phase Extraction
The principles established by Tsvet, Martin, and Synge are not historical artifacts; they are the direct progenitors of modern Solid-Phase Extraction (SPE). SPE, first applied in the 1940s and developed significantly in the 1970s, is a sample preparation technique that exploits the very same interactions [2]. It simplifies and miniaturizes column chromatography for the specific purpose of extracting, purifying, and concentrating analytes from a complex matrix before instrumental analysis [13].
The standard SPE protocol is a direct reflection of this lineage:
- Conditioning: Prepares the solid sorbent (modern equivalent of Tsvet's calcium carbonate or Martin's silica gel) for interaction with the sample.
- Sample Loading: The liquid sample passes through the sorbent, where analytes are retained based on adsorption, partition, ion-exchange, or affinity interactions—all principles explored by the pioneers.
- Washing: Removes weakly retained interferents, refining the separation.
- Elution: A strong solvent disrupts the analyte-sorbent interaction, desorbing the purified analytes in a small volume, analogous to eluting a band from a classic column [2] [13].
The core innovation of SPE was the commercialization of pre-packed columns or cartridges containing a variety of functionalized sorbents (e.g., C18 for reversed-phase, ion-exchange resins), which operationalized the theories of partition and adsorption for routine, robust, and high-throughput sample preparation [2]. This made sophisticated chromatographic cleanup accessible for countless applications in pharmaceutical, environmental, and biological analysis.
Technical Visualization: From Classic Columns to Modern SPE
The evolution from the foundational chromatography techniques to the modern SPE workflow is summarized in the following diagram.
The pioneering work of Mikhail Tsvet, Archer Martin, and Richard Synge provided the indispensable foundation for the field of separation science. Tsvet's demonstration of adsorption chromatography and Martin and Synge's theoretical and practical introduction of partition chromatography created the vocabulary and grammar of molecular separation. Their meticulous experiments, which can be reconstructed and studied today, established the core principles of interaction between analytes, stationary phases, and mobile phases. These principles were directly translated into the powerful sample preparation technique of Solid-Phase Extraction, which is now an irreplaceable tool in the researcher's toolkit. For today's scientist working on drug development or complex mixture analysis, understanding these chromatographic roots is to understand the very mechanism of the techniques they rely on daily, enabling more informed and innovative application of these methods to future challenges.
Prior to the 1970s, the field of analytical chemistry was grappling with a significant bottleneck in sample preparation. The dominant technique, liquid-liquid extraction (LLE), was plagued by numerous inefficiencies that limited analytical throughput and reliability. LLE required the use of large volumes of organic solvents, was time-consuming and labor-intensive, and often led to the formation of emulsions that compromised phase separation and analytical recovery [2]. These challenges were particularly acute in emerging fields such as environmental monitoring and pharmaceutical analysis, where researchers needed to isolate trace analytes from complex matrices like water, blood, and urine. The search for a more efficient, reliable, and less cumbersome sample preparation method set the stage for the rapid development and adoption of solid-phase extraction (SPE) during the 1970s [14].
The transformation of sample preparation during this period was not merely a technical improvement but a paradigm shift that fundamentally changed how analytical methods were developed and implemented. SPE technology emerged as a synthesis of liquid-solid extraction and column liquid chromatography principles, offering a new approach that could overcome the limitations of traditional LLE [14]. This in-depth technical guide examines the key developments, methodological advances, and practical implementations of SPE during this critical decade, providing context for its enduring impact on modern analytical science.
Historical Development: The Emergence of SPE Technology
The origins of SPE technology can be traced to broader developments in chromatography that began in the early 20th century with Mikhail Tsvet's work on column chromatography [15]. However, the specific concept of using solid sorbents for selective extraction and purification of analytes gained significant momentum during the 1960s and 1970s, as researchers sought alternatives to traditional liquid-liquid extraction [15]. The first experimental applications of SPE beginning in the 1950s focused primarily on analyzing organic traces in water samples [2]. Throughout the 1960s, researchers utilized various synthetic polymers, particularly styrenedivinylbenzene resins, in early SPE applications [2].
A critical turning point came in October 1977 with the commercial introduction of pre-filled cartridges/columns containing silica sorbents, which dramatically improved the convenience and reproducibility of the technique [2]. This development marked the beginning of a new phase in sample preparation technology, making SPE accessible to a broader range of analytical laboratories. The following year, in May 1978, this technology was featured on the cover of a prominent laboratory equipment publication, signaling its growing importance in the analytical community [2]. The first published application using silica-based SPE described the use of a Sep Pak C18 cartridge to clean histamine from wine, demonstrating the practical utility of this new approach for solving real-world analytical problems [2].
The development of stable, covalently bound chromatographic adsorbents, especially those with a reverse-phase character, opened new applications in environmental, clinical, and pharmaceutical markets [2]. The 1970s also saw the introduction of disk formats that incorporated sorbent particles between Teflon or fiberglass pads, creating extremely short yet accurate SPE cartridges that enabled good extraction recoveries with high flow rates [2]. These innovations collectively addressed the sample preparation bottleneck by providing a more efficient, reliable, and less solvent-intensive approach to sample preparation.
Table 1: Key Milestones in SPE Development During the 1970s
| Year | Development | Significance |
|---|---|---|
| 1940s | First applications of SPE | Animal charcoal used as first adsorbent for removing pigments from mixtures [2] |
| 1950s | Early experimental applications | Analysis of organic traces in water samples [2] |
| 1960s-1970s | Use of synthetic polymers | Styrenedivinylbenzene resins employed in early SPE applications [2] |
| 1977 | Introduction of pre-filled cartridges | Commercial availability of silica sorbent columns greatly improved convenience [2] |
| 1978 | First published silica SPE application | Sep Pak C18 used to clean histamine from wine [2] |
| 1970s | Development of stable bonded phases | Covalently bound reverse-phase adsorbents expanded applications [2] |
Technical Principles and Mechanisms of SPE
Fundamental Operating Principles
Solid-phase extraction operates on principles similar to liquid-liquid extraction, involving the distribution of analytes between two phases [2]. However, instead of using two immiscible liquid phases as in LLE, SPE utilizes the distribution of analytes between a liquid sample medium and a solid adsorbent phase [2] [16]. The fundamental process involves passing a liquid sample through adsorbent particles to which the analytes have greater affinity than the bulk liquid, followed by extraction of the retained analytes through elution with an appropriate solvent [2]. This approach simplifies analysis by removing much of the interfering sample matrix, while simultaneously concentrating the analytes of interest.
The core mechanism of SPE depends on the selective retention of target compounds on the sorbent material, which can be controlled through careful selection of sorbent chemistry and optimization of operational parameters. The retention occurs through various interactions including hydrophobic interactions, van der Waals forces, polar interactions, hydrogen bonding, π-π interactions, and ionic attraction, depending on the specific sorbent and analyte characteristics [14]. This versatility in retention mechanisms enables SPE to be adapted to a wide range of analytical challenges, contributing to its rapid adoption during the 1970s.
Modes of Separation
The 1970s saw the establishment of three primary modes of SPE separation, each leveraging different chemical interactions between analytes and sorbents:
Reversed-phase SPE: Utilizes a polar liquid phase and non-polar solid phase, with retention primarily governed by hydrophobic interactions and van der Waals forces [14]. This mode is particularly effective for extracting non-polar organic compounds from aqueous matrices.
Normal-phase SPE: Employs a non-polar liquid phase and polar solid phase, with retention based on hydrophilic interactions including hydrogen bonding, π-π interactions, dipole-dipole interactions, and dipole-induced dipole interactions [14]. This approach is suitable for extracting polar compounds from non-polar solvents.
Ion-exchange SPE: Relies on electrostatic attraction between charged compounds and oppositely charged functional groups on the adsorbent surface [14]. The retention in this mode is highly dependent on the pH of the sample and pretreatment solutions, which control the ionization state of both the analytes and the functional groups on the sorbent.
The following diagram illustrates the generalized workflow for solid-phase extraction, highlighting the key steps involved in processing samples through an SPE cartridge:
SPE Configurations and Formats in the 1970s
Cartridge-Based Systems
The most common SPE configuration developed during the 1970s was the disposable cartridge format [2]. These early cartridges typically consisted of high-density polypropylene syringe barrels filled with varying amounts of sorbent bed material (typically between 4-30 mg) positioned between two frits [2]. The sample was forced through the sorbent bed either by positive pressure from a piston or by reduced pressure from a vacuum source [2]. The most popular configurations were 500 mg SPE cartridges packed in 3 and 5 mL syringe barrels, capable of processing sample volumes ranging from 500 μL up to 50 mL [2]. Smaller mass cartridges (containing 100 mg of sorbent in a 1 mL syringe) were also developed for faster cleanup procedures and improved analytical sensitivity through reduced elution volumes, particularly for biological samples [2].
Alternative Formats
While cartridges dominated the early SPE landscape, the late 1970s saw the introduction of alternative formats that addressed specific limitations of conventional cartridges. The disk format emerged as an important innovation, featuring sorbent material placed between Teflon or fiberglass pads, or within a specialized matrix [2]. This design created extremely short yet accurate SPE cartridges with greater cross-sectional areas that enabled faster flow rates while maintaining good extraction efficiency [2]. Another significant development was the introduction of pipette-tip SPE (PT-SPE), which utilized very small amounts of sorbent (4-400 μg) and was designed for processing biological samples in the 0.5-1 mL volume range [2]. This format offered simplicity, shorter extraction times, high sensitivity, and did not require conditioning steps, making it particularly amenable to automation using available micropipette tools [2].
Table 2: Comparison of SPE Configurations Available in the 1970s
| Parameter | Cartridge | Pipette-Tip SPE | Disk |
|---|---|---|---|
| Sorbent Weight | 4-30 mg | 4-400 μg | 4-200 mg |
| Sample Volume | 500 μL - 50 mL | 0.5-1 mL | Up to 1 L |
| Primary Applications | Wide variety of sample matrices | Biological samples | Substantial samples |
| Key Benefits | Easy to assemble, wide range of uses, low cost | Simplicity, shorter extraction time, no conditioning required | Greater cross-sectional area, fast flow rates |
| Limitations | Small cross-section, sluggish flow rate, plugging | Restricted flow rates, limited sample capacity | Costly with large numbers of samples |
Key Methodological Advances and Experimental Protocols
Sorbent Development and Evolution
The 1970s witnessed significant advances in sorbent technology that fundamentally improved SPE performance. Early sorbents included silica-based materials, carbon-based resins, and clay-based materials [16]. A critical innovation was the development of reversed-phase sorbents, particularly octadecylsilyl-derivatized silica (C18), which became one of the most widely used sorbents for extracting non-polar organic compounds like pesticides, drugs, and industrial pollutants from aqueous matrices [16]. The introduction of polymeric sorbents such as styrenedivinylbenzene resins offered enhanced retention capabilities compared to silica-based materials for certain applications [2]. For instance, research by Huck et al. demonstrated that poly(styrene-co-divinylbenzene) achieved an average recovery of 77% for pesticides, compared to 69% for octadecyl silica phases [2].
The protocol for developing and testing new sorbents typically involved several key steps. First, researchers would synthesize or modify sorbent materials, often employing chemical modification to promote crosslinking of polymer chains using agents such as glutaraldehyde and epichlorohydrin to improve mechanical strength and chemical stability [17]. The synthesized sorbents were then characterized using techniques including Fourier transform infrared spectrometry (FT-IR) to confirm chemical interactions, scanning electron microscopy (SEM) and transmission electron microscopy (TEM) to examine morphology and particle size, and X-ray diffraction (XRD) to determine crystalline structure [17]. Finally, the extraction efficiency of new sorbents was evaluated using factorial experimental designs that systematically investigated the effects of parameters such as pH, analyte concentration, agitation time, and temperature on adsorption efficiency [17].
Standardized SPE Protocols
The following section outlines a representative experimental protocol for pesticide extraction from water samples using SPE cartridges, based on methodologies developed during the 1970s:
Materials and Reagents:
- SPE cartridges (C18, 500 mg/3 mL)
- Water samples (100-1000 mL, depending on analyte concentration)
- HPLC-grade methanol and acetonitrile
- Reagent water (HPLC-grade)
- Glacial acetic acid
- Standard solutions of target pesticides
Sample Preparation Procedure:
- Conditioning: Activate the sorbent by passing 5-10 mL of methanol through the cartridge at a flow rate of approximately 5 mL/min, followed by 5-10 mL of reagent water or initial mobile phase without allowing the cartridge to dry. [16]
- Sample Loading: Apply the prepared water sample to the cartridge at a controlled flow rate of 5-10 mL/min using positive pressure or vacuum. For large sample volumes, this step may require 30-60 minutes.
- Washing: Remove weakly retained interferents by passing 5-10 mL of a weak solvent (typically 5-20% methanol in water) through the cartridge.
- Drying: Remove residual water by drawing air or nitrogen through the cartridge for 10-20 minutes, or by centrifugation.
- Elution: Recover the retained pesticides by passing 2-5 mL of appropriate elution solvent (typically 70-100% methanol or acetonitrile, sometimes with modifiers) through the cartridge into a collection vessel.
- Concentration (if needed): Evaporate the eluate to dryness under a gentle stream of nitrogen and reconstitute in a smaller volume of solvent compatible with the subsequent analytical method.
Table 3: Research Reagent Solutions for SPE in the 1970s
| Reagent/Material | Function | Application Notes |
|---|---|---|
| C18 Bonded Silica | Reversed-phase sorbent for non-polar compounds | Most common commercial phase; used for pesticides, drugs, industrial pollutants [16] |
| Silica Gel | Normal-phase sorbent for polar compounds | Used with non-polar sample matrices [16] |
| Polystyrene-divinylbenzene Resins | Polymeric sorbent with enhanced retention | Offered better recovery for certain compounds compared to C18 [2] |
| Glutaraldehyde | Crosslinking agent for polymer sorbents | Improved mechanical strength and hardness of chitosan particles [17] |
| Epichlorohydrin | Crosslinking agent for hydroxyl groups | Enhanced chemical stability in acidic media without eliminating amine function [17] |
Advantages Over Traditional Methods
The rapid adoption of SPE during the 1970s was driven by its significant advantages over traditional liquid-liquid extraction, which it progressively replaced in many application areas [14]. A direct comparison of the two techniques reveals why SPE represented such a substantial improvement in addressing the sample preparation bottleneck:
The transition from LLE to SPE offered multiple quantifiable benefits that directly addressed the sample preparation bottleneck. SPE techniques dramatically reduced organic solvent consumption to only 10-20% of the volumes required for LLE, decreasing costs and environmental impact while improving laboratory safety [14]. The elimination of emulsion formation problems that frequently plagued LLE methods resulted in more reproducible extractions and higher quantitative recovery [14]. SPE also enabled batch processing of multiple samples, significantly increasing throughput and reducing the hands-on time required for sample preparation [14]. Furthermore, SPE provided capability for on-site sample processing, allowing stabilization of unstable analytes immediately after collection rather than requiring rapid transport to the laboratory [14]. The technique also offered superior extraction efficiency for polar compounds that were poorly recovered by traditional LLE methods [2].
Applications and Impact in Drug Development and Environmental Analysis
The implementation of SPE technology during the 1970s had transformative effects across multiple scientific disciplines, particularly in pharmaceutical and environmental analysis. In the pharmaceutical field, SPE revolutionized the extraction of drug components from complex biological matrices such as blood and urine samples [14]. The technique's ability to selectively adsorb drug compounds while removing interfering matrix components made it ideally suited for large-scale batch purification operations in clinical and research settings [14]. The consistency of SPE cartridges enabled more reproducible sample preparation, which was particularly valuable for pharmacokinetic studies and therapeutic drug monitoring programs that required processing of large numbers of patient samples.
In environmental analysis, SPE provided breakthrough capabilities for monitoring trace organic contaminants in water samples [2]. The technology enabled concentration and purification of pesticides, industrial chemicals, and other pollutants from large water volumes (up to liters), significantly improving detection limits and analytical reliability compared to previous methods [16]. The development of SPE disks specifically designed for processing large sample volumes addressed the need for monitoring regulated contaminants in drinking water, wastewater, and natural waters [2]. The ability to perform on-site processing using portable SPE equipment also solved the long-standing challenge of analyte instability during transport from sampling locations to analytical laboratories, particularly for volatile or degradable compounds [14].
The impact of SPE extended beyond these primary application areas to include analysis of natural products, foods and beverages, and clinical specimens [2]. In each of these fields, SPE provided a versatile platform for sample cleanup and concentration that could be adapted to specific analytical requirements through selection of appropriate sorbent chemistries and processing conditions. This flexibility, combined with the consistent performance characteristics of commercial SPE products, established solid-phase extraction as a fundamental sample preparation technique that would continue to evolve in subsequent decades through further innovations in sorbent technology and automation.
The year 1977 marked a transformative juncture in analytical chemistry with the commercial launch of the Sep-Pak cartridge by Waters Associates. This innovation addressed a critical bottleneck in sample preparation by miniaturizing and simplifying liquid chromatography principles for routine laboratory use. This whitepaper details the technical specifications, operational protocols, and profound impact of this development, framing it within the broader context of solid-phase extraction (SPE) history. The introduction of a standardized, convenient, pre-packed cartridge format catalyzed the shift from laborious, solvent-intensive liquid-liquid extraction to a more efficient, robust, and accessible sample preparation methodology, thereby accelerating research and standardization in pharmaceutical and environmental analysis.
Prior to the 1970s, the preparation of samples for analysis was often the most tedious and problematic aspect of the analytical process. Techniques like liquid-liquid extraction (LLE) were dominant, requiring large volumes of high-purity solvents, being time-consuming and labor-intensive, and posing risks of emulsion formation and analyte loss [2]. The theoretical foundation for a better method existed within liquid chromatography (LC), but the practical application was confined to column chromatography, which was not optimized for rapid sample clean-up and concentration.
The conceptual evolution of SPE can be divided into distinct historical periods as identified by researchers. The "age of active carbon" represented the first analytical attempts to use solid surfaces like activated carbon filters for recovering organic compounds from water, dating back to the 1940s and 1950s [18]. This was followed by the "age of search for the most appropriate material" (late 1960s to early 1980s), where investigators explored a wide variety of new sorbents, including styrenedivinylbenzene resins and functionalized silicas, seeking a universal material for analyte recovery [18] [2]. While the principles were being established, a lack of standardization and convenient formats limited widespread adoption.
It was within this context that Waters Associates, a leader in LC technology, identified sample preparation as a major challenge for laboratories. The company challenged its scientist, Patrick McDonald, to "find new, faster, more convenient ways to do traditional sample preparation operations" using their existing LC technology [19]. The solution was not a new polymer, but a new paradigm in packaging and application.
The Sep-Pak Innovation: Technical Specifications and Workflow
The commercial introduction of the Sep-Pak cartridge in October 1977 was a watershed moment because it packaged established chromatographic sorbent technology into a ready-to-use, disposable, and user-friendly format. The core innovation was the application of triaxial bed compression and individual cartridge packaging to create a reliable and reproducible miniature column [19].
Cartridge Configuration and Design
The classic Sep-Pak design revolutionized laboratory practice by creating a self-contained, disposable extraction unit. Its configuration, which has become the archetype for thousands of subsequent SPE products, is detailed below.
The cartridge's design was pivotal for its performance. The compressed sorbent bed was key to preventing channeling—a common issue in loosely packed columns where the sample liquid finds paths of least resistance, leading to inefficient and inconsistent extraction. Compression ensured uniform flow and maximum contact between the analyte and the sorbent particles. The frits at either end served as physical barriers to retain the fine sorbent particles while allowing liquids to pass through freely. The Luer tip design enabled a secure, leak-free connection to vacuum manifolds or syringe barrels, facilitating the controlled flow of solvents.
Foundational Experimental Protocol
The standardized procedure for using a reverse-phase C18 Sep-Pak cartridge, as established in early applications, involves a sequence of critical steps to condition the sorbent, load the sample, wash away interferences, and finally elute the purified analytes. This foundational protocol is summarized in the table below.
Table: Foundational Protocol for C18 Sep-Pak Cartridge Use
| Step | Purpose | Typical Solvents/Volumes | Critical Parameters |
|---|---|---|---|
| 1. Conditioning | To solvate the hydrophobic C18 chains and create an active surface. | 5-10 mL Methanol | Ensures high analyte recovery by wetting the sorbent. |
| 2. Equilibration | To replace the organic solvent with an aqueous matrix compatible with the sample. | 5-10 mL Water or aqueous buffer | Prevents sample precipitation and ensures proper retention. |
| 3. Sample Loading | To pass the aqueous sample through the cartridge, retaining analytes on the C18 phase. | Sample in aqueous solution | Flow rate control (<10 mL/min) is crucial to prevent breakthrough. |
| 4. Washing | To remove weakly adsorbed matrix interferences while retaining analytes. | 5-10 mL Water or mild buffer (e.g., 5% methanol) | Uses a solvent strong enough to remove impurities but too weak to elute the target analytes. |
| 5. Elution | To desorb the purified analytes from the sorbent for collection. | 5-10 mL Organic solvent (e.g., Acetonitrile, Methanol) | Uses a strong solvent to disrupt hydrophobic interactions; collected in a clean vial. |
This protocol, enabled by the Sep-Pak design, provided a dramatic improvement in reproducibility and ease of use over previous methods. An early published application demonstrated this efficiency by using a Sep-Pak C18 cartridge to clean histamine from wine, showcasing its utility in complex matrices [2].
The Scientist's Toolkit: Essential Research Reagent Solutions
The successful implementation of SPE, as pioneered by the Sep-Pak, relies on a suite of key materials and reagents. The following table details these essential components and their functions within the SPE workflow.
Table: Essential Reagents and Materials for Solid-Phase Extraction
| Item | Function | Typical Examples |
|---|---|---|
| SPE Sorbent | The active media that selectively interacts with and retains analytes based on its chemistry. | C18 (Reversed-Phase), Silica (Normal-Phase), Cation/Anion Exchange Resins [18] [2]. |
| Conditioning Solvent | Prepares the sorbent surface for interaction with the sample. | Methanol, Acetonitrile. |
| Equilibration Solvent | Creates a compatible environment for the application of the sample. | Water, aqueous buffer (e.g., phosphate, acetate). |
| Elution Solvent | Disrupts the analyte-sorbent interaction to recover the target compounds. | Methanol, Acetonitrile, Ethyl Acetate, often with modifiers (e.g., acid, base). |
| Vacuum Manifold | A device that holds multiple cartridges and uses negative pressure to process samples in parallel. | 12- or 24-port manifolds with collection tubes. |
Impact and Evolution in Scientific Research
The commercial availability of Sep-Pak cartridges had an immediate and profound impact, triggering a "boom situation" in the adoption of SPE [18]. Its influence can be traced through several key areas:
- Standardization and Methodology: The consistency offered by pre-packed, quality-controlled cartridges allowed SPE to be incorporated into official standardized methods by environmental and pharmaceutical regulatory bodies [18]. This was a critical step in establishing the technique's legitimacy.
- Accelerated Research: The convenience of the format drastically reduced sample preparation time. As noted in the historical review, analysts were spending up to two-thirds of their analysis time on sample collection and preparation prior to such innovations [18]. Sep-Pak and its successors freed up significant resources.
- Proliferation of Sorbent Chemistry: While the first Sep-Pak cartridges utilized silica-based C18 and other classic materials, the commercial success of the format spurred intense development of new sorbents. This led to the creation of more specialized phases, including polymeric sorbents like poly(styrene-co-divinylbenzene) (PS-DVB), which offered higher capacity and better retention for certain non-polar compounds [2].
- Foundation for Future Innovation: The cartridge format itself evolved, leading to the introduction of SPE disks in 1989 for handling larger sample volumes, and later, 96-well plate formats to meet the high-throughput demands of modern drug development and clinical laboratories [2]. The principles established by the Sep-Pak cartridge directly enabled subsequent micro-extraction techniques like Solid-Phase Microextraction (SPME) [19].
The historical trajectory of SPE technology, from its early experimental roots to its modern automated implementations, was fundamentally redirected by the commercialization of the Sep-Pak cartridge. This transition is illustrated in the following timeline.
The introduction of the Sep-Pak cartridge in 1977 was far more than a simple product launch; it was a pivotal event that fundamentally reoriented the practice of sample preparation. By packaging established LC sorbent chemistry into a convenient, reliable, and accessible format, Waters Associates addressed a critical pain point for researchers and catalyzed a paradigm shift from LLE to SPE. This innovation laid the groundwork for half a century of advancement, enabling the standardization, automation, and miniaturization that today's drug development professionals and research scientists rely upon for efficient and reliable analysis. The Sep-Pak cartridge truly earned its title as a "watershed moment," channeling the scattered streams of early SPE research into a powerful and enduring technological river.
The accurate measurement of trace-level organic contaminants in complex environmental matrices represents a significant challenge for modern analytical science. This challenge is primarily driven by stringent environmental legislation from regulatory agencies like the European Community (CEE) and the U.S. Environmental Protection Agency (US-EPA), which mandate increasingly lower concentration limits for contaminants such as pesticides and polycyclic aromatic hydrocarbons (PAHs) in natural waters and sediments [20]. Achieving these required detection limits necessitates sophisticated sample preparation techniques that can isolate and enrich analytes while removing matrix interferences. Within this framework, Solid-Phase Extraction (SPE) has evolved from a simple preparatory step into a critical technology enabling compliance with regulatory standards and ensuring environmental and public health safety.
This technical guide examines the key drivers behind the development of SPE techniques, focusing on the interplay between environmental legislation and analytical innovation. It details the experimental protocols and material choices that underpin reliable trace analysis, providing researchers and drug development professionals with a comprehensive resource for method development and implementation.
The Legislative Imperative and Analytical Challenges
Environmental legislation directly shapes analytical methodologies by defining the required performance characteristics for monitoring programs. Regulatory agencies establish maximum contaminant levels to protect water quality and ecosystem health, compelling laboratories to develop methods capable of detecting contaminants at parts-per-billion (ppb) or even parts-per-trillion (ppt) concentrations [20].
The primary analytical challenges in meeting these requirements include:
- Matrix Complexity: Environmental samples such as surface water and sediments contain abundant interfering substances, notably humic materials, which co-elute with target analytes and create significant background interference in chromatographic analysis [20].
- Analyte Diversity: Monitoring programs must simultaneously track contaminants with diverse chemical properties, from non-polar PAHs to polar pesticides and their degradation products [20].
- Throughput Demands: Regulatory compliance often requires processing large numbers of samples, necessitating methods that balance sensitivity with efficiency and throughput.
Without selective sample preparation, the matrix effects from co-extracted interferents can severely compromise the sensitivity and accuracy of chromatographic techniques such as Gas Chromatography (GC) and High-Performance Liquid Chromatography (HPLC) [20].
The Evolution of Solid-Phase Extraction Materials
The development of SPE sorbents reflects a continuous effort to enhance selectivity and efficiency in sample preparation. The table below summarizes the key stages in this evolution, from conventional to advanced materials.
Table 1: Evolution of Solid-Phase Extraction Sorbents
| Sorbent Generation | Key Materials | Retention Mechanism | Advantages | Limitations |
|---|---|---|---|---|
| Conventional | C18-bonded silica, polymeric styrene-divinylbenzene [20] [2] | Reversed-phase (van der Waals, hydrogen bonding) [20] | Wide applicability, well-understood protocols | Limited selectivity for polar compounds, susceptible to silica-based issues [20] |
| Improved Polymeric | Cross-linked styrene-divinylbenzene polymers [20] [2] | Reversed-phase, enhanced surface area | Better retention of polar compounds, higher capacity, pH-stable [20] | Still largely non-selective, leading to co-extraction of interferences [20] |
| Selective & Mixed-Mode | Layered sorbents, ion-exchange materials [20] [21] | Multiple mechanisms (polarity, ion-exchange) [21] | Class-selective isolation, cleaner extracts | More complex method development |
| Advanced Molecular Recognition | Immunosorbents (IS), Molecularly Imprinted Polymers (MIPs) [20] | Antibody-antigen binding, template-specific cavities [20] | High specificity, excellent clean-up, suitable for complex matrices [20] | High cost (IS), complex synthesis (MIPs), limited to known analytes [20] |
The Shift to Selective Mechanisms
The limitations of conventional C18 sorbents, particularly their non-selectivity, drove the development of materials with targeted interaction mechanisms. Ion-exchange sorbents leverage electrostatic interactions, ideal for analytes that can be ionized, such as acidic or basic compounds. Method development requires careful consideration of the analyte's pKa and the sample pH to ensure both the analyte and sorbent are in their charged states for effective retention [21].
The most significant advance in selectivity came with molecular recognition sorbents. Immunosorbents (IS) use immobilized antibodies to capture specific analytes or classes with exceptional precision, dramatically reducing matrix effects [20]. Molecularly Imprinted Polymers (MIPs) offer a synthetic alternative, creating polymer networks with cavities complementary to the target molecule in shape, size, and functional groups. MIPs provide similar selectivity to antibodies but with greater chemical and physical robustness [20].
Experimental Protocols for Reliable Trace Analysis
A robust SPE-based analytical method involves a systematic sequence of critical steps. The following workflow diagram and detailed protocol ensure reproducible and reliable results for trace analysis.
Diagram 1: Solid-Phase Extraction Workflow
Detailed SPE Procedure Using a Reversed-Phase Cartridge
This protocol is adapted for the analysis of non-polar to mid-polar pesticides in water samples using a polymeric sorbent [20] [21].
Step 1: Sorbent Conditioning
- Purpose: To solvate the sorbent and create a uniform interface for analyte interaction, removing any potential contaminants from the manufacturing or storage process.
- Procedure: Pass 5-10 mL of methanol (or acetonitrile) through the SPE cartridge under gentle vacuum (~1-2 mL/min). Follow immediately with 5-10 mL of ultra-pure water or a buffer that matches the sample's pH and ionic strength. Do not allow the sorbent bed to dry out before sample loading.
Step 2: Sample Loading
- Purpose: To pass the prepared sample through the sorbent, allowing the target analytes to be retained.
- Procedure: Adjust the sample pH if necessary to suppress ionization for reversed-phase extraction (typically pH 2-7 depending on analytes). Load the sample at a controlled flow rate of 5-10 mL/min to maximize the contact time and avoid analyte breakthrough, which occurs when the sorbent's capacity is exceeded [21].
Step 3: Interference Washing
- Purpose: To remove weakly adsorbed matrix components without displacing the target analytes.
- Procedure: Wash the sorbent with 2-5 mL of a weak solvent, typically water or a water/organic mixture (e.g., 5-20% methanol in water). This step elutes highly polar interferences while retaining the analytes of interest.
Step 4: Analyte Elution
- Purpose: To release the concentrated, cleaned-up analytes from the sorbent in a small volume of solvent.
- Procedure: Pass 1-5 mL of a strong organic solvent (e.g., methanol, acetonitrile, or ethyl acetate) through the cartridge. Collect the eluate in a clean vial. The elution solvent is chosen based on the polarity of the analytes and its compatibility with the subsequent chromatographic analysis (e.g., LC-MS) [20] [21].
Step 5: Post-Processing and Analysis
- Purpose: To prepare the extract for instrumental analysis.
- Procedure: The eluate may be evaporated to near-dryness under a gentle stream of nitrogen and reconstituted in a solvent compatible with the chromatographic system. The final extract is then analyzed by GC or LC-MS.
Quantitative Comparison of SPE Configurations
The choice of SPE format significantly impacts factors such as sample throughput, required volume, and potential for automation. The following table compares the common configurations.
Table 2: Comparison of Common SPE Configurations and Their Applications
| Parameter | Cartridge | Disk | Pipette-Tip (PT-SPE) | Solid-Phase Microextraction (SPME) |
|---|---|---|---|---|
| Sorbent Weight | 4–30 mg [2] | 4–200 mg [2] | 4–400 µg [2] | N/A (coated fiber) |
| Typical Sample Volume | 500 µL – 50 mL [2] | Up to 1 L [2] | 0.5 – 1 mL [2] | Small (headspace or immersion) |
| Key Benefits | Easy to use, wide range of sorbents [2] | Large cross-sectional area, fast flow rates, good for large volumes [2] | Simplicity, shorter extraction time, small elution volume, amenable to automation [2] | Solvent-free, minimal sample preparation, easily coupled to GC/LC [22] |
| Primary Limitations | Slow flow rates, potential channeling [2] | Can be costly, may require filtration [2] | Limited capacity, not suited for large samples [2] | Lower adsorption capacity, limited effectiveness for some analytes [2] |
| Ideal Application | General-purpose, wide variety of sample matrices [2] | Processing large-volume environmental water samples [20] [2] | Small-volume biological samples, high-throughput proteomics [23] [2] | Rapid, green analysis of volatile/semi-volatile compounds [22] |
The Scientist's Toolkit: Essential Research Reagent Solutions
Selecting the appropriate sorbent and configuration is fundamental to successful method development. The following table catalogs key materials and their functions in SPE.
Table 3: Key Research Reagent Solutions for Solid-Phase Extraction
| Material / Product | Function & Principle of Action | Typical Application Example |
|---|---|---|
| C18-Bonded Silica | Reversed-phase sorbent; retains analytes via hydrophobic (van der Waals) interactions [20] [21]. | Isolation of hydrophobic compounds (e.g., PAHs) from aqueous samples [20]. |
| Polymeric Sorbent (e.g., PS-DVB) | Hydrophobic polymer with higher capacity and better retention of polar compounds than C18; more resistant to pH extremes [20] [2]. | Broad-spectrum extraction of pesticides with varying polarities from water [20]. |
| Mixed-Mode Cation/Anion Exchanger | Combines reversed-phase and ion-exchange mechanisms; allows selective retention based on both hydrophobicity and charge [21]. | Separation of acidic herbicides from their neutral parent compounds [20] [21]. |
| Molecularly Imprinted Polymer (MIP) | Synthetic polymer with pre-determined selectivity for a specific template molecule; offers antibody-like specificity [20]. | Selective enrichment of a specific triazine herbicide from complex soil extracts [20]. |
| Immunosorbent (IS) | Antibody-coated sorbent; provides extreme selectivity via antigen-antibody binding [20]. | Ultra-selective extraction of a single pesticide class (e.g., phenylureas) from groundwater [20]. |
| SOLAµ HRP Spin Plates | Micro-SPE format in a 96-well plate; enables rapid, semi-automated processing of small volumes with minimal solvent [23]. | High-throughput clean-up of protein digests in proteomic studies [23]. |
| ZIPTIP C18 Pipette Tips | Pipette-tip based micro-SPE; used for desalting and concentrating small-volume samples directly in a pipette tip [23]. | Purification of peptide mixtures prior to LC-MS/MS analysis [23]. |
Method Validation and Quality Assurance
Validating an SPE method is critical for demonstrating its reliability for regulatory analysis. A core principle is that the more selective the SPE step, the better the sensitivity achieved in the final analysis [20]. Validation is typically performed by:
- Participation in inter-laboratory exercises where a sample with a known concentration of contaminants is analyzed and the results are compared with those from other laboratories [20].
- Analysis of Certified Reference Materials (CRMs), which have certified values for specific analytes, allowing for the determination of method accuracy and precision [20].
- Establishing key performance metrics including recovery (should be consistent and high), precision (repeatability and reproducibility), and the method's Limit of Detection (LOD) and Limit of Quantification (LOQ) to ensure they meet legislative requirements [20].
The development of solid-phase extraction techniques has been inextricably linked to the demands of environmental legislation. The journey from conventional C18 silica to advanced molecularly selective sorbents like MIPs and immunosorbents underscores a continuous pursuit of greater specificity, sensitivity, and efficiency in trace analysis. As regulatory standards become ever more stringent, the role of robust, validated, and highly selective SPE methodologies will only grow in importance. By understanding the principles, materials, and protocols outlined in this guide, researchers and drug development professionals are equipped to develop analytical methods that not only comply with regulatory demands but also contribute meaningfully to environmental monitoring and public health protection.
The early development of research into solid-phase extraction (SPE) techniques was fundamentally driven by the need to overcome the significant limitations of liquid-liquid extraction (LLE), the dominant sample preparation method for decades. While LLE is based on the partitioning of analytes between two immiscible liquid phases, its application in analytical laboratories presented considerable challenges, including being time-consuming, labor-intensive, and requiring large volumes of organic solvents, which resulted in high costs and environmental concerns [2] [24]. Furthermore, LLE is notoriously inefficient for extracting polar compounds and prone to forming emulsions, which can halt analytical workflows and introduce errors [2] [25]. Within the context of pharmaceutical and environmental research, these drawbacks limited analytical throughput, increased the environmental footprint of laboratories, and compromised the reliability of results for certain analyte classes.
The transition to SPE represented a paradigm shift from a solution-based partitioning process to a sorption mechanism, where analytes are selectively retained on a solid sorbent packed into a cartridge or disk [26]. This core principle offered a new framework for sample preparation, enabling researchers to simultaneously achieve purification and concentration of target analytes with far greater efficiency and less solvent. This guide explores the fundamental principles of SPE that facilitated this transition, provides a quantitative comparison of the techniques, details foundational experimental protocols, and visualizes the key workflows that have become standard in modern analytical laboratories.
Fundamental Principles of Solid-Phase Extraction
SPE's operation is governed by the same principles as liquid chromatography, utilizing a solid stationary phase and a liquid mobile phase [26]. The process involves passing a liquid sample through a sorbent bed, where analytes are selectively retained based on interactions with the sorbent material. Interfering components are washed away, and the purified analytes are then recovered in a small volume of a strong elution solvent [2]. This process effectively separates the analytes from the sample matrix and concentrates them, thereby improving the sensitivity and reliability of subsequent analysis by HPLC, GC, or MS [26].
The historical development of SPE was marked by key advancements in sorbent technology. Initial applications in the 1940s and 1950s used materials like animal charcoal for removing pigments [2]. The technology evolved significantly with the introduction of pre-filled cartridges containing silica-based sorbents in 1977, which standardized the procedure and made it more convenient for laboratories [2]. The subsequent development of disk formats in 1989 and polymeric monoliths in the 1990s offered higher flow rates and better performance for specific applications, further cementing SPE's role as a versatile sample preparation tool [2].
Key Sorbent Phases and Their Applications
The selectivity of SPE is largely determined by the chemistry of the sorbent material. Early research focused on developing a range of sorbents to address different analytical needs.
Table 1: Key SPE Sorbent Types and Their Applications
| Sorbent Type | Mechanism of Action | Typical Applications |
|---|---|---|
| Reversed-Phase (e.g., C18) | Hydrophobic interactions | Retains non-polar analytes from polar matrices (e.g., water) [2] [26] |
| Normal-Phase | Polar interactions (e.g., hydrogen bonding, dipole-dipole) | Retains polar analytes from non-polar matrices [2] [26] |
| Ion Exchange (Cationic/Anionic) | Electrostatic attraction | Retains ionizable compounds based on their charge [2] [26] |
| Mixed-Mode | Combined mechanisms (e.g., reversed-phase and ion exchange) | Offers high selectivity for complex matrices [2] |
Quantitative Comparison: LLE vs. SPE
The transition from LLE to SPE is justified by clear and substantial advantages across multiple operational parameters. The following table summarizes a quantitative comparison between the two techniques, highlighting the core efficiencies gained by adopting SPE.
Table 2: Quantitative Comparison of LLE and SPE
| Parameter | Liquid-Liquid Extraction (LLE) | Solid-Phase Extraction (SPE) |
|---|---|---|
| Typical Solvent Volume | High (often 100s of mL) [24] | Low to moderate (tens of mL) [24] [25] |
| Processing Time | Lengthy (often >30 min per sample) [27] | Rapid (10-15 minutes per sample) [24] |
| Automation Potential | Low [25] | High (amenable to 96-well plates and robotics) [2] [25] |
| Risk of Emulsion Formation | High [2] [25] | Very Low [27] |
| Selectivity | Moderate [25] | High (wide choice of sorbents) [25] |
| Ability to Concentrate Analytes | Limited | Excellent (via large sample load/small elution volume) [26] |
The data demonstrates that SPE provides a more efficient, reproducible, and environmentally friendly approach than LLE [24] [27]. The dramatic reduction in solvent use is a key "green" advantage, reducing costs and environmental impact [25]. Furthermore, the avoidance of emulsions and the high degree of automation possible with SPE make it particularly suitable for high-throughput laboratories, such as those in drug development, where processing hundreds of samples reliably is a routine requirement [27].
Foundational SPE Experimental Protocol
The following section outlines a generic but detailed protocol for performing SPE using a standard cartridge. This protocol can be adapted and optimized for specific analytes and matrices.
Reagent Solutions and Materials
Table 3: Essential Research Reagent Solutions for SPE
| Item | Function / Explanation |
|---|---|
| SPE Cartridge | Disposable device containing the solid sorbent (e.g., C18, Ion Exchange) that selectively retains analytes [26]. |
| Conditioning Solvent | Prepares the sorbent for optimal interaction with the sample; typically a strong solvent like methanol [26]. |
| Equilibration Solvent | Re-equilibrates the sorbent to a solvent compatible with the sample matrix; often water or a buffer [26]. |
| Wash Solvent | Removes weakly retained interferents from the sorbent without eluting the analytes of interest [26]. |
| Elution Solvent | A strong solvent that disrupts the analyte-sorbent interaction to recover the purified analytes [26]. |
Step-by-Step Methodology
- Sample Pre-Treatment: The sample must be prepared to ensure optimal retention of the analytes. This typically involves dilution with water or a buffer (e.g., for serum or urine), adjusting the pH to suppress ionization of the analyte and enhance retention, and removing particulates via filtration or centrifugation to prevent clogging of the SPE bed [26].
- Cartridge Conditioning: Pass 1-2 column volumes of a strong solvent (e.g., methanol) through the cartridge to solvate the sorbent and activate the functional groups. This is followed by 1-2 column volumes of the equilibration solvent (e.g., water or a weak buffer). The sorbent must not be allowed to dry out after this step [26].
- Sample Loading: Apply the pre-treated sample to the top of the cartridge reservoir. A controlled, slow flow rate (e.g., 1-2 mL/min) is critical to maximize the contact time between the analytes and the sorbent, ensuring quantitative retention [26].
- Washing: Pass 1-2 column volumes of a wash solvent through the cartridge. This solvent must be strong enough to remove unwanted matrix interferences but weak enough to leave the target analytes bound. A common wash is a water-organic mixture (e.g., 5% methanol) [26].
- Elution: The purified analytes are recovered by applying 1-2 column volumes of a strong elution solvent. Using two small aliquots of eluent is more efficient than one large volume. The elution solvent is selected to disrupt the specific interactions retaining the analytes (e.g., a strong organic solvent like acetonitrile for reversed-phase SPE) [26]. The collected eluate can now be analyzed directly or concentrated further before analysis.
SPE Workflow Visualization
The following diagram illustrates the logical sequence and decision points in a typical SPE protocol.
Advanced and Alternative Techniques
While cartridge-based SPE is the most common format, the principle of using a solid phase for extraction has evolved into several advanced techniques. Solid-Phase Microextraction (SPME) is a non-exhaustive, solvent-free technique where a coated fiber is exposed to the sample for extraction, ideal for volatile compounds and direct coupling to GC or LC [2]. Magnetic Solid-Phase Extraction (MSPE) uses magnetic or magnetically-modified sorbents dispersed in the sample; after extraction, the sorbent is easily retrieved with a magnet, simplifying the process and avoiding column clogging issues [2].
Beyond SPE, other modern extraction techniques have also been developed on the principle of reducing solvent consumption. Supercritical Fluid Extraction (SFE), which uses supercritical CO₂ as the extraction fluid, is recognized for its high efficiency, low environmental impact, and minimal solvent use [28] [29]. Similarly, Pressurized Liquid Extraction (PLE) uses solvents at high temperatures and pressures to achieve fast and efficient extraction from solid matrices [30] [31]. These techniques, while often used for initial extraction from solid samples, share the common goal of overcoming the drawbacks of traditional, solvent-intensive methods.
The transition from Liquid-Liquid Extraction to Solid-Phase Extraction marks a critical advancement in the history of analytical chemistry. Framed within the broader thesis of early SPE research, this shift was not merely a change in technique but a fundamental improvement driven by the need for greater efficiency, selectivity, and sustainability. SPE successfully addressed the core limitations of LLE—its solvent-intensive nature, operational cumbersome, and inefficiency with polar compounds—by introducing a sorption-based methodology that allows for simultaneous purification and concentration. The quantitative data, foundational protocols, and workflows detailed in this guide underscore why SPE has become the method of choice in research and drug development laboratories worldwide. The continued innovation in sorbent chemistries and formats, building on these early developments, ensures that SPE will remain a cornerstone of efficient sample preparation for the foreseeable future.
Methodological Evolution: Sorbents, Configurations, and Driving Modern Applications
The evolution of solid phase extraction (SPE) represents a critical pursuit in analytical chemistry, driven by the need for more efficient, selective, and robust sample preparation techniques. SPE serves as a fundamental bridge between complex sample matrices and sophisticated analytical instruments, enabling the isolation, purification, and concentration of target analytes from interfering substances [13]. The transition from traditional silica-based sorbents to advanced polymeric and mixed-mode phases marks a significant technological advancement in this field, particularly for pharmaceutical research and environmental analysis where complex samples demand superior clean-up capabilities [2].
This technical guide examines the developmental pathway of SPE sorbents within the context of early extraction technique research, highlighting how material innovations have addressed persistent challenges in analytical sample preparation. The journey from basic silica materials to sophisticated mixed-mode phases demonstrates a continuous refinement of retention mechanisms and selectivity profiles, ultimately expanding the applicability of SPE across diverse scientific domains including drug discovery, bioanalysis, and environmental monitoring [13] [2].
Historical Development and Evolutionary Timeline
The development of SPE technologies has progressed through distinct phases marked by significant material innovations. Initial applications emerged in the 1940s, with animal charcoal serving as one of the first adsorbents for removing pigments from reaction mixtures [2]. The period from 1968 to 1977 represented a formative stage where synthetic polymers like styrene-divinylbenzene resins first appeared in SPE applications [2].
A transformative advancement occurred in October 1977 with the introduction of pre-filled cartridges containing silica sorbents, particularly those with stable covalently bound reverse phases like C18, which dramatically improved convenience and expanded applications into environmental, clinical, and pharmaceutical markets [2]. The 1989 introduction of SPE disks or membranes marked another evolutionary leap, incorporating sorbent materials between Teflon or fiberglass pads to create short, precise extraction formats with improved flow characteristics [2].
The 1990s witnessed the emergence of monolithic polymer stationary phases, with poly(styrene-co-divinylbenzene) (PS-DVB) demonstrating superior recovery rates for non-polar compounds compared to traditional octadecyl silica (ODS) phases [2]. This period also saw the development of multi-functional polymer-based sorbents designed to enhance retention of polar analytes through hydrophilic interactions, addressing a significant limitation of earlier materials [2].
The following diagram illustrates this evolutionary pathway of SPE sorbent technologies:
Fundamental SPE Principles and Sorbent Classifications
Core Mechanism of Solid Phase Extraction
Solid Phase Extraction operates on chromatographic principles utilizing differential affinities between a liquid mobile phase and solid stationary phase [13]. The process exploits various intermolecular interactions including van der Waals forces, hydrogen bonding, dipole-dipole interactions, and ionic interactions to achieve selective separation [13]. In practice, a liquid sample is passed through adsorbent particles that exhibit greater affinity for target analytes than the bulk liquid, followed by selective elution using appropriate solvents [2]. This fundamental mechanism enables significant simplification of complex sample matrices while concentrating target compounds for subsequent analysis.
The standard SPE process comprises four critical stages: (1) Conditioning - preparing the sorbent by passing solvents to activate and wet the surface; (2) Sample Loading - introducing the liquid sample containing analytes of interest; (3) Washing - removing interfering compounds with selective solvents; and (4) Elution - releasing purified analytes using strong solvents [13]. This systematic approach allows researchers to selectively retain target compounds while eliminating matrix interferences that could compromise analytical results.
Sorbent Classification by Retention Mechanism
SPE sorbents are categorized primarily by their retention mechanisms, each designed for specific analyte properties and sample matrices:
Table 1: Classification of SPE Sorbents by Retention Mechanism
| Sorbent Type | Retention Mechanism | Common Phases | Typical Applications |
|---|---|---|---|
| Reversed-Phase | Hydrophobic interactions | C18, C8 bonded silica, polymeric resins | Non-polar to moderately polar compounds from polar matrices (e.g., pharmaceuticals, pesticides) [13] |
| Normal-Phase | Hydrogen bonding, dipole-dipole, π-π interactions | Silica, alumina, cyano, diol | Polar compounds from non-polar matrices (e.g., lipids, carbohydrates) [13] |
| Ion Exchange | Electrostatic interactions | Cation exchange (sulfonate), anion exchange (quaternary ammonium) | Charged compounds (acids, bases) from aqueous matrices [13] |
| Mixed-Mode | Multiple mechanisms (e.g., reversed-phase + ion exchange) | Polymer-based with ion-exchange groups | Analytes with diverse properties (acidic, basic, neutral compounds) in complex matrices [32] |
Mixed-mode sorbents represent a significant advancement by combining multiple retention mechanisms in a single material [13]. These phases typically incorporate a polymeric reversed-phase backbone with ion-exchange functional groups (e.g., sulfonate groups for cation exchange or quaternary ammonium groups for anion exchange) covalently bonded to it [32]. This dual-mechanism approach enables retention of compounds through both hydrophobic and ionic interactions, providing superior selectivity for complex samples containing analytes with diverse chemical properties [32].
Comparative Analysis of Sorbent Materials
Silica-Based Sorbents: Foundations and Limitations
Silica-based sorbents formed the historical foundation of SPE technology, with C18 (octadecyl silica) becoming one of the most widely used materials for reversed-phase applications [2]. These sorbents feature silanol groups that can be derivatized with various functional groups to achieve different selectivity profiles. While silica-based phases offered significant improvements over liquid-liquid extraction, they suffer from several limitations including limited pH stability (typically effective only between pH 2-8) and potential for irreversible adsorption due to residual silanol groups [2].
The introduction of pre-packed silica cartridges in 1977 standardized SPE procedures and made the technology more accessible to analytical laboratories [2]. Early applications demonstrated the effectiveness of silica-based sorbents for cleaning histamine from wine, establishing a precedent for food and environmental analysis [2]. However, the constrained pH stability and inconsistent retention of polar compounds highlighted the need for more robust sorbent materials.
Polymer-Based Sorbents: Enhanced Performance Characteristics
Polymeric sorbents, particularly styrene-divinylbenzene copolymers, addressed several limitations of silica-based materials [2]. These sorbents exhibit superior pH stability across the entire pH range, enabling more aggressive sample pretreatment and washing conditions [32]. Polymer-based phases also demonstrate higher capacity and better retention for polar compounds compared to traditional C18 silica [2].
Research by Huck and colleagues demonstrated that poly(styrene-co-divinylbenzene) achieved an average recovery of 77% for pesticides compared to 69% for ODS phases, confirming the enhanced extraction efficiency of polymeric materials [2]. The development of hydrophilic-modified polymers further improved retention of polar analytes through incorporated functional groups that facilitate hydrogen bonding [2].
Mixed-Mode Sorbents: Strategic Selectivity Advancements
Mixed-mode sorbents represent the current state-of-the-art in SPE technology, combining reversed-phase retention with ion-exchange capabilities in a single material [32]. These phases are particularly valuable for mass spectrometry applications where comprehensive sample clean-up is essential to minimize ion suppression and matrix effects [32].
The strategic advantage of mixed-mode phases lies in their ability to retain compounds through two independent mechanisms simultaneously. For example, mixed-mode cation exchange phases retain basic compounds through both hydrophobic interactions with the polymeric backbone and ionic interactions with negatively charged sulfonate groups [32]. This dual-retention capability enables more aggressive wash steps with strong organic solvents (e.g., 100% methanol or acetonitrile) without eluting target analytes, resulting in superior sample clean-up [32].
Table 2: Comparative Performance Characteristics of SPE Sorbent Materials
| Characteristic | Silica-Based | Polymer-Based | Mixed-Mode |
|---|---|---|---|
| pH Stability | Limited (pH 2-8) | Full pH range | Full pH range [32] |
| Capacity | Moderate | High | High [32] |
| Retention Mechanism | Primarily single mechanism | Primarily hydrophobic | Multiple mechanisms [32] |
| Polar Compound Retention | Limited | Good | Excellent [32] |
| Clean-up Efficiency | Moderate | Good | Excellent [32] |
| Method Development | Straightforward | Moderate | Complex but highly selective |
Experimental Protocols and Method Development
Systematic Method Development for Mixed-Mode SPE
Developing an effective SPE method using mixed-mode sorbents requires a systematic approach that leverages the dual retention mechanisms. The following protocol provides a framework for method development:
Step 1: Compound Characterization
- Determine logP and pKa values for all target analytes [32]
- Identify which compounds can be positively or negatively charged at relevant pH values
- Note stability constraints for pH-sensitive compounds
Step 2: Sorbent Selection
- Select mixed-mode cation exchange if basic (positively chargeable) analytes predominate [32]
- Choose mixed-mode anion exchange for acidic (negatively chargeable) analytes [32]
- Consider the balance between charged and neutral compounds in the panel
Step 3: pH Optimization
- Adjust sample pH to 2 units below the pKa for basic compounds to protonate them [32]
- Adjust sample pH to 2 units above the pKa for acidic compounds to deprotonate them [32]
- Ensure all compounds remain stable at the selected pH
Step 4: Sample Loading and Washing
- Load pretreated samples at the optimized pH [32]
- Perform aqueous wash with solution similar to pretreatment solution [32]
- Implement organic wash with increasing organic percentages to determine optimal stringency [32]
Step 5: Elution and Reconstitution
- Elute with basic organic solvent (e.g., 2% ammonium hydroxide in methanol) for cation exchange [32]
- Elute with acidic organic solvent (e.g., 2-5% formic acid in methanol) for anion exchange [32]
- Dry samples under nitrogen and reconstitute in compatible solvent [32]
Chemometric Optimization Approach
Modern SPE method development increasingly employs chemometric approaches to optimize multiple parameters efficiently. A recent study demonstrated this methodology for pesticide analysis in water samples using Oasis HLB cartridges [33]:
Initial Screening: Apply Full Factorial Design to identify significant factors (sample volume, pH, elution solvent composition) [33]
Response Optimization: Utilize Central Composite Design to determine optimal conditions [33]
Method Validation: Evaluate statistical models and validate significance [33]
This approach identified optimal conditions of 300 mL sample volume adjusted to pH 3.0 with elution using 3 mL of methanol:ethyl acetate (70:30 v/v) [33]. The systematic methodology reduced experimental requirements while ensuring robust performance across 24 multiclass pesticides [33].
Application Protocol for Environmental Water Analysis
A validated protocol for pharmaceutical analysis in environmental waters using homemade silica-based mixed-mode ion-exchange sorbents demonstrates practical implementation:
- Sorbent Preparation: Silica-based mixed-mode sorbents with strong ion-exchange moieties, without graphene additives for optimal performance [34]
- Sample Pretreatment: Adjust water samples to pH 3 using acidification [34]
- SPE Procedure: Load samples, wash with 2 mL methanol, elute with appropriate solvent [34]
- Analysis: Employ LC-MS/MS with appropriate chromatographic separation
This methodology achieved apparent recoveries of 22-68% with method detection limits of 1-28 ng/L for various pharmaceuticals and drugs of abuse in river water, effluent wastewater, and influent wastewater [34].
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful implementation of advanced SPE methodologies requires specific materials and reagents optimized for different applications. The following table details essential components for establishing robust SPE protocols in pharmaceutical and environmental research:
Table 3: Essential Research Reagents and Materials for SPE Applications
| Item | Specifications | Function/Application |
|---|---|---|
| Mixed-Mode Cation Exchange Sorbent | Polymeric reversed-phase with sulfonate groups (e.g., EVOLUTE EXPRESS CX) [32] | Retention of basic compounds through hydrophobic and cation exchange mechanisms |
| Mixed-Mode Anion Exchange Sorbent | Polymeric reversed-phase with quaternary ammonium groups (e.g., EVOLUTE EXPRESS AX) [32] | Retention of acidic compounds through hydrophobic and anion exchange mechanisms |
| Oasis HLB Cartridges | 6 mL, 200 mg, 30 μm [33] | Hydrophilic-lipophilic balanced polymer for broad-spectrum retention |
| Vacuum Manifold | 12-port Visiprep SPE vacuum manifold [33] | Simultaneous processing of multiple samples under controlled flow |
| Elution Solvents | Methanol, acetonitrile, ethyl acetate, with acid/base modifiers [32] [33] | Selective elution of retained analytes from mixed-mode sorbents |
| pH Adjustment Reagents | Formic acid, ammonium hydroxide, acetic acid | Optimization of ionization state for ionic retention |
| Solvent Evaporation System | Nitrogen evaporation system with water bath [33] | Concentration of eluates prior to analysis |
| UHPLC-MS/MS System | Waters Acquity UHPLC-MS/MS TQD with BEH C18 column [33] | High-resolution separation and detection of extracted analytes |
Analytical Pathways and Retention Mechanisms
The effectiveness of mixed-mode SPE stems from the intelligent application of complementary retention mechanisms. The following diagram illustrates the strategic pathway for leveraging these interactions in method development:
The evolution from silica to polymer-based and mixed-mode sorbents represents a paradigm shift in solid phase extraction technology, enabling researchers to address increasingly complex analytical challenges. Mixed-mode phases in particular offer unprecedented selectivity through their dual retention mechanisms, making them indispensable for modern LC-MS applications in pharmaceutical research and environmental monitoring [32].
The continued refinement of SPE sorbents demonstrates how fundamental material science advancements drive progress in analytical chemistry. As researchers face growing demands for sensitivity, selectivity, and throughput in sample preparation, further innovations in sorbent technology will undoubtedly emerge. The strategic integration of mixed-mode principles with emerging formats such as pipette-tip SPE and 96-well plate configurations promises to extend the capabilities of this foundational technique, ensuring its continued relevance in the analytical laboratory of the future [2].
Solid-phase extraction (SPE) is a critical sample preparation technique based on liquid-solid chromatographic separation principles, designed to isolate, concentrate, and purify target compounds from complex sample matrices [35]. This technique has largely replaced many traditional liquid-liquid extraction methods by offering significant advantages, including reduced solvent consumption (by over 60% in some applications), improved sample throughput, more tunable selectivity, easier automation, and avoidance of emulsion formation [36] [37]. In pharmaceutical research and environmental analysis, SPE serves as a fundamental sample preparation step that enhances detection sensitivity and method robustness by removing matrix interferences that could compromise analytical results [38].
The fundamental principle of SPE involves passing a liquid sample through a solid sorbent packed in a cartridge, disk, or well-plate format, where target analytes are selectively retained based on their physical and chemical properties, while unwanted matrix components are washed away [39] [35]. The selectively retained analytes can then be eluted in a purified, concentrated form using an appropriate elution solvent [39]. The four basic steps in any SPE protocol include: (1) conditioning the sorbent to activate the functional groups, (2) loading the sample, (3) washing to remove impurities, and (4) eluting the target compounds [37]. Understanding the three core retention mechanisms—reversed-phase, normal-phase, and ion exchange—is essential for selecting the appropriate SPE methodology for specific research applications in drug development and analytical science.
Normal-Phase SPE Mechanism
Fundamental Principles and Interactions
Normal-phase SPE operates on a polar retention mechanism, where the stationary phase is polar relative to the mobile phase [40] [41]. This mechanism primarily utilizes polar interactions such as hydrogen bonding, dipole-dipole interactions, and π-π interactions between polar functional groups on the analytes and the polar sorbent surface [40]. The strength of these polar forces is greater than non-polar van der Waals forces but weaker than ionic interactions [40]. This mechanism is particularly effective for extracting polar compounds from non-polar or weakly polar sample matrices, as the polar analytes interact more strongly with the polar sorbent than with the non-polar solvent environment [41].
In normal-phase SPE, the polar sorbent features hydrophilic properties with surface functional groups capable of forming hydrogen bonds with appropriate analyte functional groups [40]. Common polar functional groups that interact effectively with normal-phase sorbents include hydroxyl (-OH), amine (-NH₂), and sulfhydryl (-SH) groups [40]. The successful application of normal-phase SPE relies on maintaining a sample matrix environment that does not compete with these polar interactions, which is why weakly polar solvents are ideal for sample dissolution in this mode.
Stationary Phases and Sorbent Materials
Normal-phase SPE utilizes several types of polar sorbent materials, each with specific characteristics and applications:
- Silica gel (underivatized silica): Consists of porous silica with silanol (Si-OH) groups that interact with analytes through hydrogen bonding and dipole interactions [40].
- Alumina (Al₂O₃): Aluminum oxide available in acidic, basic, or neutral forms, providing different selectivity for various compound classes [40].
- Florisil (magnesium silicate): Particularly useful for extracting polar pesticides and other environmental contaminants [40].
- Bonded polar phases: Silica-based sorbents chemically modified with polar functional groups including cyano (CN), amino (NH₂), and diol (2OH) groups [40] [37]. These bonded phases offer more reproducible and predictable retention characteristics compared to underivatized silica.
The selection of specific sorbent depends on the analyte properties and the required selectivity. For instance, amino phases are particularly effective for capturing carbohydrates and sugars, while cyano phases offer intermediate polarity for more selective separations.
Experimental Protocol and Methodological Considerations
A typical normal-phase SPE procedure follows these methodical steps:
- Conditioning: The sorbent is conditioned with 3-5 bed volumes of a non-polar solvent (e.g., hexane or dichloromethane) to solvate the functional groups and prepare the sorbent surface for sample interaction [40] [37].
- Sample Loading: The sample, dissolved in a non-polar or weakly polar solvent (e.g., hexane, dichloromethane, or vegetable oil), is applied to the column [40]. The flow rate should be controlled (approximately 1-2 drops/second) to ensure sufficient interaction time between analytes and sorbent [42].
- Washing: Interfering compounds are removed using a non-polar or slightly polar wash solvent (e.g., hexane or dichloromethane) that has insufficient strength to elute the retained analytes [40].
- Elution: Target compounds are recovered using a polar solvent (e.g., methanol, acetonitrile, or ethyl acetate with increasing polarity modifiers) that disrupts the polar interactions between the analytes and sorbent [40] [41].
A critical consideration in normal-phase SPE is maintaining a solvent environment where the sample solvent is less polar than the wash solvent, which is less polar than the elution solvent [40]. This increasing polarity series ensures that analytes remain retained during loading and washing steps while being efficiently eluted during the final stage. The elution strength of solvents must be carefully calibrated to disrupt the specific polar interactions without requiring excessive volumes.
Applications in Research Context
Normal-phase SPE finds particular utility in pharmaceutical analysis for extracting polar drugs, metabolites, and natural products from non-polar matrices [40]. In environmental monitoring, it effectively isolates polar pesticides and contaminants from organic extracts [40]. The technique is also valuable in lipidomics for fractionating lipid classes based on their polarity differences and in natural product research for purifying bioactive compounds from crude extracts [35].
Figure 1: Normal-Phase SPE Workflow and Mechanism. This diagram illustrates the four-step procedure and fundamental interactions in normal-phase solid-phase extraction, highlighting the polar stationary phases and increasing solvent polarity required for effective elution.
Reversed-Phase SPE Mechanism
Fundamental Principles and Interactions
Reversed-phase SPE operates on a hydrophobic retention mechanism, where the stationary phase is non-polar relative to the mobile phase [40]. This mechanism primarily relies on non-polar van der Waals forces between hydrophobic regions of the analyte molecules and the hydrophobic functional groups bonded to the sorbent surface [40] [41]. Also known as hydrophobic or non-polar interaction, this mechanism involves temporary, electric interactions created by fleeting dipole moments in molecules without a net dipole [41]. These interactions, while individually weak, collectively provide strong retention when the sorbent has sufficient hydrophobic surface area and the sample matrix is polar enough to promote the hydrophobic effect.
The hydrophobic effect plays a crucial role in reversed-phase SPE, where polar molecules in the solvent environment effectively repel non-polar analytes, thereby strengthening the interaction between analytes and the hydrophobic sorbent [41]. This mechanism is particularly effective for retaining non-polar to moderately polar compounds from polar aqueous or semi-aqueous matrices, making it one of the most widely used SPE mechanisms in bioanalytical and environmental applications.
Stationary Phases and Sorbent Materials
Reversed-phase SPE employs several classes of hydrophobic sorbents with varying selectivity and retention characteristics:
- C18 (Octadecyl): The most common reversed-phase sorbent, featuring an 18-carbon hydrocarbon chain bonded to silica, providing strong retention for non-polar compounds [40] [37].
- C8 (Octyl): An 8-carbon chain bonded phase offering slightly less retention than C18, often used for more moderately hydrophobic compounds [37].
- C6, C4, C2: Shorter alkyl chains providing progressively weaker hydrophobic retention, suitable for more polar analytes or when milder elution conditions are required [41].
- Cyclohexyl: A cyclic hydrocarbon phase offering different selectivity compared to linear alkyl chains [41].
- Polymeric sorbents (e.g., PS-DVB): Hydrophobic polystyrene-divinylbenzene polymers that often provide higher capacity and better pH stability compared to silica-based sorbents [37].
- Hydrophilic-Lipophilic Balanced (HLB) sorbents: Copolymer sorbents containing both hydrophobic and hydrophilic monomers that can retain a wide range of acidic, basic, and neutral compounds [38].
The selection of sorbent involves considering the hydrophobicity of the target analytes, with longer alkyl chains generally providing stronger retention for highly non-polar compounds.
Experimental Protocol and Methodological Considerations
A standardized reversed-phase SPE procedure typically follows this sequence:
- Conditioning: The sorbent is conditioned with 3-5 bed volumes of a water-miscible organic solvent (e.g., methanol or acetonitrile) to solvate the hydrophobic chains, followed by 3-5 bed volumes of water or aqueous buffer to create a compatible environment for sample loading [38] [37].
- Sample Loading: The sample, dissolved in a polar solvent (typically water or a buffered aqueous solution), is applied to the column [40]. Maintaining a controlled flow rate (1-2 drops/second) ensures optimal retention [42].
- Washing: Polar interferences are removed using a predominantly aqueous wash solution (typically water or 5-20% organic solvent in water) that is insufficient to elute the retained hydrophobic analytes [40].
- Elution: Target compounds are recovered using an organic solvent (e.g., methanol, acetonitrile, tetrahydrofuran, or isopropanol) that disrupts the hydrophobic interactions [40] [41].
In reversed-phase SPE, the solvent polarity decreases sequentially from sample solvent (most polar) to wash solvent (moderately polar) to elution solvent (least polar) [40]. This decreasing polarity series ensures that the hydrophobic interactions remain intact during loading and washing, while being effectively disrupted during elution. For strongly retained analytes, solvent mixtures such as dichloromethane:ethyl acetate (1:1) may be required for complete elution [40].
Applications in Research Context
Reversed-phase SPE is extensively used in pharmaceutical research for extracting drugs and metabolites from biological fluids such as plasma, serum, and urine [35] [38]. In environmental analysis, it effectively concentrates non-polar to moderately polar pollutants (pesticides, hydrocarbons, industrial chemicals) from water samples [35]. The technique also finds applications in food safety testing for isolating contaminants from aqueous food extracts, clinical toxicology for drug screening, and proteomics for peptide cleanup and concentration [35] [38].
Figure 2: Reversed-Phase SPE Workflow and Mechanism. This diagram illustrates the four-step procedure and fundamental interactions in reversed-phase solid-phase extraction, highlighting the hydrophobic stationary phases and decreasing solvent polarity required for effective elution.
Ion-Exchange SPE Mechanism
Fundamental Principles and Interactions
Ion-exchange SPE operates on an electrostatic retention mechanism, where charged analytes are retained through ionic interactions with oppositely charged functional groups on the sorbent surface [43] [39]. This mechanism requires that both the analyte and the sorbent functional groups be in their ionized form, which is controlled through precise pH adjustment of the sample matrix [42]. For effective retention, the sample pH must be adjusted to ensure the analytes are charged—for basic compounds, the pH should be at least two units below the pKa, while for acidic compounds, the pH should be at least two units above the pKa [42].
Ion-exchange sorbents are classified based on the type of ionic group bonded to the surface and the pH range over which they maintain their ionic functionality [43] [41]. The kinetic exchange processes in ion-exchange SPE are considerably slower than in reversed-phase or normal-phase mechanisms, necessitating controlled flow rates (approximately 1 drop/second) to allow sufficient interaction time between analytes and sorbent functional groups [42].
Stationary Phases and Sorbent Materials
Ion-exchange SPE sorbents are available in four primary categories:
- Strong Cation Exchange (SCX): Contains strong acid functional groups (typically aliphatic sulfonic acid, -SO₃⁻) that remain negatively charged across the entire pH range, suitable for retaining positively charged bases [43] [39].
- Weak Cation Exchange (WCX): Features weak acid functional groups (typically carboxylic acids, -COO⁻) that are negatively charged at higher pH (above approximately 5) but neutral at low pH [43] [39].
- Strong Anion Exchange (SAX): Contains strong base functional groups (typically quaternary ammonium, -N(R)₃⁺) that maintain a positive charge across the entire pH range, suitable for retaining negatively charged acids [43] [39].
- Weak Anion Exchange (WAX): Features weak base functional groups (primary, secondary, or tertiary amines, -NH₂, -NHR, -NR₂) that are positively charged at low pH but neutral at high pH (above approximately 9) [43] [39].
The distinction between strong and weak exchangers is crucial for method development. Strong exchangers are preferred when consistent retention is needed across a wide pH range, while weak exchangers offer the advantage of being neutralized for easier elution under specific pH conditions.
Experimental Protocol and Methodological Considerations
A comprehensive ion-exchange SPE procedure involves these critical steps:
- Sample Pretreatment: The sample is diluted with an appropriate buffer to ensure the ionic strength is less than 0.1M and the pH is adjusted to ionize the target analytes [42]. For basic compounds, dilution with 10-25 mM buffer (e.g., potassium phosphate) at pH 3-6; for acidic compounds, dilution with 10-50 mM buffer at pH 7-9 [42].
- Conditioning/Equilibration: The sorbent is conditioned with 1-2 bed volumes of methanol or acetonitrile, then equilibrated with a buffer similar in pH and ionic strength to the sample pretreatment buffer [42]. This step ensures the sorbent is in the appropriate ionic form for retention.
- Sample Loading: The pretreated sample is applied at a controlled, reduced flow rate of approximately 1-2 drops/second to allow sufficient time for electrostatic interactions [42].
- Washing: A buffer of appropriate pH and ionic strength is used to remove polar interferences without displacing the retained analytes [43]. More hydrophobic interferences can be removed using up to 100% methanol diluted in the sample pretreatment buffer [42].
- Elution: Three primary strategies can be employed for eluting analytes from ion-exchange sorbents [43]:
- pH manipulation: Adjusting the pH to neutralize the charge on either the analyte or sorbent functional group (e.g., using 2-5% ammonium hydroxide in methanol for basic compounds or 2-5% acetic acid in methanol for acidic compounds) [42].
- High ionic strength buffer: Using a buffer with high salt concentration (>1M) to disrupt the electrostatic interactions [43] [42].
- Competitive displacement: Employing a counterion with higher affinity for the sorbent surface to displace the analytes [43].
The counterion selectivity series must be considered for method development. For cation exchangers, the selectivity follows Ca²⁺ > Mg²⁺ > K⁺ > NH₄⁺ > Na⁺ > H⁺ > Li⁺, while for anion exchangers, it follows benzene sulphonate > citrate > NO₃⁻ > Cl⁻ > HCO₃⁻ > formate > acetate > F⁻ > OH⁻ [42].
Applications in Research Context
Ion-exchange SPE is particularly valuable in pharmaceutical analysis for isolating acidic or basic drugs and their metabolites from biological matrices [38]. In environmental monitoring, it effectively extracts ionic contaminants such as herbicides, pesticides, and inorganic ions from water samples [35]. The technique also finds specialized applications in biochemical research for purifying proteins, peptides, and nucleic acids, and in clinical diagnostics for extracting specific biomarker classes from complex biological fluids [35] [38].
Comparative Analysis of SPE Mechanisms
Mechanism Selection Guidelines
Selecting the appropriate SPE mechanism requires systematic evaluation of analyte properties and sample matrix characteristics. The following decision framework provides guidance for mechanism selection:
- Analyte Polarity: Non-polar to moderately polar compounds are best suited for reversed-phase SPE; polar compounds with functional groups capable of hydrogen bonding are ideal for normal-phase SPE; ionizable compounds with acidic or basic functional groups are candidates for ion-exchange SPE [40] [41].
- Sample Matrix: Aqueous or polar matrices favor reversed-phase SPE; organic or non-polar matrices favor normal-phase SPE; aqueous matrices with controllable pH and ionic strength suit ion-exchange SPE [40].
- Primary Interaction Required: Hydrophobic interactions indicate reversed-phase; polar interactions (hydrogen bonding, dipole-dipole) indicate normal-phase; electrostatic interactions indicate ion-exchange [40] [41].
- Compound State: Neutral compounds typically suit reversed-phase or normal-phase; ionizable compounds that can be charged through pH control suit ion-exchange [42] [40].
For compounds possessing multiple functional groups, the retention mechanism that provides the best separation from matrix interferences should be selected. For example, 2-naphthylamine contains both hydrophobic and hydrophilic groups—in a weakly polar matrix, normal-phase extraction utilizing polar interactions would be appropriate, while in a polar matrix, reversed-phase extraction utilizing hydrophobic interactions would be preferable [40].
Quantitative Performance Comparison
Table 1: Comparative Analysis of Core SPE Mechanisms
| Parameter | Reversed-Phase | Normal-Phase | Ion-Exchange |
|---|---|---|---|
| Primary Retention Mechanism | Hydrophobic interactions (van der Waals forces) [40] [41] | Polar interactions (hydrogen bonding, dipole-dipole) [40] | Electrostatic (ionic) interactions [43] [39] |
| Stationary Phase Polarity | Non-polar (C18, C8, HLB, polymer) [40] | Polar (silica, CN, NH₂, diol) [40] | Charged functional groups (SCX, WCX, SAX, WAX) [43] [41] |
| Sample Matrix | Polar (water, buffers) [40] | Non-polar (hexane, DCM, oils) [40] | Aqueous with controlled pH and low ionic strength (<0.1M) [42] |
| Elution Solvents | Methanol, acetonitrile, THF, chloroform, ethyl acetate [40] | Methanol, acetonitrile, more polar modifiers [40] | pH-adjusted solvents, high ionic strength buffers, competing ions [43] [42] |
| Optimal Flow Rates | 1-2 drops/second [42] | 1-2 drops/second [42] | <1 mL/min for 100-mg bed [43] |
| Key Applications | Drugs, metabolites, environmental pollutants from aqueous matrices [35] [38] | Polar drugs, pesticides, natural products from organic matrices [40] | Acidic/basic compounds, biomolecules, ionic contaminants [35] [38] |
| Solvent Polarity Trend | Decreases: sample → wash → elution [40] | Increases: sample → wash → elution [40] | Controlled by pH and ionic strength [43] [42] |
Advanced and Mixed-Mode Mechanisms
Beyond the three core mechanisms, advanced SPE approaches have been developed to address complex separation challenges:
Mixed-Mode SPE: Incorporates multiple retention mechanisms within a single sorbent, most commonly combining reversed-phase and ion-exchange properties [38] [41]. These sorbents are particularly valuable for fractionating multiple compound classes from the same sample or for achieving higher selectivity for challenging separations [41]. For example, a mixed-mode sorbent containing both non-polar and cation-exchange media can sequentially elute non-polar compounds with organic solvent followed by basic compounds using pH adjustment [41].
Multimodal SPE: Utilizes either serial connection of multiple SPE cartridges with different mechanisms or a single cartridge containing multiple functionalized sorbents [37]. This approach is beneficial when dealing with samples containing diverse analyte classes or when requiring high specificity for a particular analyte in a complex matrix [37].
Molecular Imprinted Polymers (MIPs): Highly selective sorbents created to contain recognition sites complementary to specific template molecules in terms of size, shape, and functional group orientation [37]. These sorbents operate on a "lock-and-key" principle similar to antibody-antigen interactions, providing exceptional specificity for target compounds [37].
Restricted Access Media (RAM): Specialized sorbents that combine size exclusion with chromatographic retention, typically featuring an outer hydrophilic layer that excludes macromolecules like proteins while allowing smaller analytes to access inner hydrophobic retention sites [37]. These are particularly valuable for direct injection of biological fluids in therapeutic drug monitoring and bioanalysis [37].
Experimental Protocols and Research Applications
Detailed Methodologies from Cited Research
A comprehensive study evaluating SPE reliability for hydrocarbon analysis provides an exemplary protocol for mechanism comparison [36]. This research systematically compared five different sorbents—neutral Si (silica), SiOH (silica gel), Ag-ion (silver-ion), neutral Al (alumina), and Ag-ion mixed with activated silica—against traditional liquid column chromatography (LCC) for separating saturate and aromatic compounds from crude oils and organic-rich source rock extracts [36].
The experimental methodology included these critical steps:
- Sample Preparation: Two organic-rich source rock extracts (ORE-A, ORE-G) and two crude oils (CO-1, CO-2) from the Eagle Ford Formation in Texas were selected to represent immature to peak oil maturity windows, ensuring the presence of detectable biomarkers [36].
- SPE Procedure: Samples were passed through the various SPE cartridges following conditioning with dichloromethane and n-hexane. The saturate fraction was eluted with n-hexane, followed by elution of the aromatic fraction with dichloromethane [36].
- Analysis: Fractions were analyzed by gas chromatography-mass spectrometry (GC-MS) to determine peak resolution, compound concentration, and biomarker ratios for thermal maturity assessment [36].
- Recovery Assessment: An aromatic ortho-terphenyl (OTP) standard was quantified in each sample to evaluate recovery compared to traditional LCC [36].
The results demonstrated significant performance differences between mechanisms. Neutral Si, SiOH, and neutral Al sorbents showed poor separation efficiency, leaving 40-100% of aromatics in the saturate fraction [36]. In contrast, Ag-ion mixed with activated silica provided superior separation, yielding up to 23 times higher aromatic concentration than SiOH and reducing solvent consumption by 63% compared to traditional LCC methods [36].
Research Reagent Solutions and Materials
Table 2: Essential Research Reagents and Materials for SPE Method Development
| Reagent/Material | Function/Application | Representative Examples |
|---|---|---|
| Reversed-Phase Sorbents | Retention of non-polar to moderately polar compounds from polar matrices [40] [37] | C18, C8, C6, cyclohexyl, HLB copolymer [38] [41] |
| Normal-Phase Sorbents | Retention of polar compounds from non-polar matrices [40] | Silica gel, alumina, Florisil, CN-, NH₂-, diol-bonded phases [40] [37] |
| Ion-Exchange Sorbents | Retention of ionizable compounds through electrostatic interactions [43] [41] | SCX, WCX, SAX, WAX [38] [41] |
| Organic Solvents | Conditioning, washing, and elution in various SPE mechanisms [40] | Methanol, acetonitrile, hexane, dichloromethane, ethyl acetate [36] [40] |
| Buffers and pH Adjusters | Control of ionization state for ion-exchange SPE and stability in reversed-phase SPE [42] | Ammonium acetate, phosphate buffers, ammonium hydroxide, acetic acid [42] |
| SPE Device Formats | Accommodation of different sample volumes and throughput requirements [39] [38] | Cartridges (1-100 mL), 96-well plates, disks, µElution plates, pipette tips [39] [38] |
| Vacuum Manifolds | Simultaneous processing of multiple samples with flow control [39] | 24-position cartridge manifolds, 6-disk manifolds, positive pressure systems [39] |
The strategic selection of appropriate SPE mechanisms—reversed-phase, normal-phase, and ion-exchange—fundamentally influences the success of sample preparation in analytical chemistry and drug development research. Each mechanism offers distinct advantages tailored to specific analyte properties and matrix characteristics: reversed-phase excels with non-polar compounds in polar matrices; normal-phase optimizes polar compound extraction from non-polar environments; and ion-exchange provides selective retention for ionizable molecules through electrostatic interactions [40] [41].
Advanced SPE technologies, including mixed-mode sorbents, molecular imprinted polymers, and restricted access media, continue to expand application possibilities for complex sample matrices [37]. The evolution of SPE from a simple cleanup technique to a sophisticated separation tool reflects its critical role in modern analytical workflows, particularly as detection instruments become more sensitive to matrix effects [38]. Through understanding these core mechanisms and their appropriate implementation, researchers can develop more efficient, selective, and robust sample preparation methods that enhance analytical accuracy while reducing solvent consumption and laboratory waste [36].
Solid-phase extraction (SPE) has undergone significant technological evolution since its early experimental applications, transforming from a technique primarily using activated carbon filters into a sophisticated sample preparation mainstay [18]. The core principle of SPE, which involves the partitioning of analytes between a liquid sample and a solid stationary phase, has remained constant, but the configurations through which this is achieved have diversified remarkably to meet evolving analytical demands [2] [39]. The drive for greater efficiency, reduced solvent consumption, higher throughput, and compatibility with complex matrices has propelled innovation from traditional cartridges to disks and, more recently, to miniaturized formats such as pipette-tip SPE (PT-SPE) [2] [44]. This progression reflects a broader trend in analytical science toward miniaturization, automation, and environmentally friendly methods. The historical development of SPE can be traced through several distinct "ages," from the early use of active carbon, through the search for optimal sorbent materials, to the current era of technical refinement and format diversification [18]. This guide provides an in-depth examination of the core SPE configurations—cartridges, disks, and pipette-tips—detailing their operational principles, relative advantages, and specific applications within modern analytical workflows, particularly in pharmaceutical and bioanalytical research.
Core SPE Principles and Historical Context
SPE is a sample preparation process that utilizes a solid phase and a liquid phase to isolate, concentrate, and purify analytes from a complex sample matrix [45]. It was developed as a more efficient alternative to liquid-liquid extraction (LLE), overcoming numerous drawbacks such as incomplete phase separations, the need for large quantities of organic solvents, and the use of expensive, breakable glassware [45]. The fundamental procedure involves loading a sample solution onto the SPE medium, washing away undesired components, and then eluting the desired analytes with a suitable solvent into a collection tube [45].
The first experimental applications of SPE began in the 1940s, with animal charcoal serving as one of the earliest adsorbents for removing pigments from reaction mixtures [2]. A significant milestone occurred in October 1977 with the introduction of pre-packed cartridges containing silica sorbents, which made the procedure more convenient and marked the beginning of a new developmental phase [2]. The 1980s saw the introduction of SPE disks or membranes, creating an extremely short, wide bed geometry that enabled higher flow rates [2]. The continual quest for more efficient, faster, and greener sample preparation has since driven the development of miniaturized and automated formats, including the rise of pipette-tip-based SPE [46].
Table 1: Historical Milestones in SPE Development
| Time Period | Major Development | Key Feature |
|---|---|---|
| 1940s - 1950s | First Experimental Applications [2] [18] | Use of activated carbon and animal charcoal |
| Late 1960s - 1980s | Search for Optimal Materials [18] | Development of new sorbents like bonded silicas |
| 1977 | Introduction of Pre-packed Cartridges [2] | Commercial availability of standardized, convenient formats |
| 1989 | Introduction of SPE Disks [2] | Higher flow rates for large volume samples |
| 1990s - Present | Miniaturization and Automation [2] [44] | Development of PT-SPE, 96-well plates, and online systems |
Traditional and Modern SPE Configurations
SPE Cartridges
The SPE cartridge is the most traditional and common configuration, resembling a medical syringe barrel made of polypropylene or glass, packed with a small amount of sorbent (typically less than 1 gram, though up to 10-gram versions are available) [45] [2]. The sorbent, often based on silica bonded with specific functional groups, is contained between two porous frits made of materials like polyethylene, Teflon, or stainless steel [45] [2]. The particle size of the packing material is generally around 40 microns, larger than in HPLC, and irregularly shaped particles are often used as cost is more central than efficiency in SPE [45].
Cartridges are available with a wide variety of stationary phases, including reversed-phase (C18, C8), normal-phase (silica, cyano, amino), and ion-exchange (cation, anion) sorbents, allowing for separation based on hydrophobicity, polarity, or ionic interactions [45] [39]. Their operation typically involves four key steps, which can be visualized in the following workflow:
Figure 1: The Four Fundamental Steps of a Solid-Phase Extraction Protocol.
SPE Disks
SPE disks were developed to address the limitations of cartridges, particularly for processing large sample volumes [45] [2]. Disks are flat, with a thickness of 1 mm or less and diameters ranging from 4 mm to 96 mm [45]. They resemble filtration membranes but differ in that they have sorbent particles (making up 60-90% of the disk weight) embedded within an expanded PTFE (polytetrafluoroethylene) network or a fiberglass matrix [45] [2].
The primary advantage of the disk format is its high cross-sectional area and short bed length, which creates a small void volume and minimizes flow resistance [2]. This design enables significantly higher flow rates than cartridges. For example, one liter of relatively clean water can pass through a 45 mm disk in approximately 20 minutes, a process that may require one to two hours with a standard cartridge [45]. This makes disks particularly suitable for environmental applications where large volumes of water need to be processed to concentrate trace-level pollutants [2]. A potential limitation is that they can be more prone to plugging with very dirty samples [2].
The Rise of Pipette-Tip Solid-Phase Extraction (PT-SPE)
PT-SPE represents the trend toward miniaturization in sample preparation [2] [44]. This format involves integrating a small amount of sorbent (typically 4-400 µg) into a pipette tip [2]. The extraction is performed by repeatedly aspirating and dispensing the sample solution through the sorbent bed using a pipette controller or syringe, enhancing the contact between the analyte and the sorbent [47].
The primary advantages of PT-SPE are its low consumption of samples and solvents, its simplicity, and its ease of integration into workflows without requiring additional hardware like vacuum manifolds [2] [44]. It is ideally suited for processing small volume samples (0.5-1 mL), such as those encountered in biological and pharmaceutical analysis [2]. A comparative study of commercial PT-SPE (ZIPTIP C18) and a 96-well SPE plate (SOLAµ HRP) for porcine retinal proteomics found that both methods performed equally well in terms of protein identifications and reproducibility, but the plate format offered greater convenience and semi-automation for higher throughput [48].
Recent innovations have focused on improving the design of PT-SPE devices. Traditional designs that pack sorbent between two frits can suffer from sorbent leakage and are incompatible with high-capacity nanosorbents smaller than the frit's pore size [44]. To address this, sorbent packing-free PT-SPE strategies have been developed, where the pipette tip itself is reshaped (e.g., through heating and stretching into slender or self-crimping tubes) and its inner surface functionalized to act as the extraction phase, eliminating the need for packed sorbents and filters altogether [44].
Table 2: Comparison of Key SPE Configurations
| Parameter | SPE Cartridge | SPE Disk | Pipette-Tip (PT-SPE) |
|---|---|---|---|
| Sorbent Mass | 4 mg - 10 g [45] [2] | 4 - 200 mg [2] | 4 - 400 µg [2] |
| Typical Sample Volume | 500 µL - 50 mL [2] | Up to 1 L [2] | 0.5 - 1 mL [2] |
| Format & Dimensions | Syringe barrel (e.g., 1-20 mL) [45] | Flat disk (Ø 4-96 mm) [45] | Modified pipette tip [44] |
| Key Advantages | Wide range of sorbents; easy lab assembly; low cost [2] | High flow rates; small void volume; fast for large samples [45] [2] | Miniaturized; low sample/solvent use; simple operation [2] [44] |
| Common Applications | Wide variety of sample matrices [2] | Environmental water analysis [45] [2] | Biological samples (urine, plasma), proteomics [2] [47] [48] |
Detailed Experimental Protocol: Pipette-Tip SPE for Bioanalysis
The following detailed methodology is adapted from a recent study that developed a self-assembly pipette tip with restricted access mesoporous polypyrrole for the extraction of enalapril from human urine, followed by analysis with capillary electrophoresis (CE-DAD) [47]. This protocol exemplifies a modern, green, and miniaturized SPE approach.
Materials and Reagents
Table 3: Research Reagent Solutions for PT-SPE
| Item | Function / Specification | Example / Source |
|---|---|---|
| Pipette Tips | Substrate for sorbent or functionalized surface | Standard or reshaped plastic tips [44] |
| Sorbent Material | Selective retention of analytes | Mesoporous Polypyrrole (mPPy), C18 silica [47] |
| Sample | Source of analytes | Human urine, adjusted to pH 7.5 [47] |
| Washing Solvent | Removes weakly retained interferences | n-Hexane (250 µL) [47] |
| Elution Solvent | Releases purified analytes for collection | Acetonitrile (750 µL) [47] |
| Buffers | Control pH for optimal extraction/elution | Phosphate buffer (pH 7.5 for sample) [47] |
Step-by-Step Procedure
- Pipette-Tip Preparation: The adsorbent (e.g., 40 mg of mesoporous polypyrrole double-coated with hydrophilic monomers and encapsulated with protein) is packed into the tip or the inner surface of a reshaped tip is chemically functionalized [47] [44].
- Conditioning: The tip is conditioned by aspirating and dispensing an appropriate solvent (e.g., methanol and water) to activate the sorbent surface.
- Sample Loading: Aspirate 750 µL of the prepared sample (e.g., urine at pH 7.5) into the tip and dispense it back to waste. Repeat this process several times to ensure sufficient contact between the analytes and the sorbent [47].
- Washing: Aspirate 250 µL of a washing solvent (e.g., n-hexane) into the tip and dispense to waste to remove undesired matrix components and interferences [47].
- Elution: Aspirate 750 µL of elution solvent (e.g., acetonitrile) into the tip and dispense it into a clean collection vial. This fraction contains the purified analytes and is ready for direct analysis or further concentration [47].
- Analysis: The eluent is injected into an analytical instrument such as a Capillary Electrophoresis system with a Diode Array Detector (CE-DAD) or an LC-MS for separation and quantification [47].
The entire process and the configuration of a standard PT-SPE device can be summarized in the following diagram:
Figure 2: Configuration and Operational Workflow of a Standard Pipette-Tip SPE Device.
The landscape of SPE configurations has expanded significantly from its origins with activated carbon, offering scientists a toolkit tailored to diverse analytical challenges. Cartridges remain the versatile workhorse for a broad range of applications, while disks provide an efficient solution for processing large sample volumes. The rise of pipette-tip SPE marks a clear trajectory toward miniaturization, offering unmatched efficiency for small-volume biological samples and demonstrating a commitment to green chemistry through reduced solvent consumption. The ongoing innovation in sorbent materials, device design, and integration with analytical instrumentation ensures that SPE will continue to be a critical and evolving technique for researchers and drug development professionals seeking robust, reproducible, and efficient sample preparation.
The late 1980s marked a pivotal moment in the evolution of sample preparation techniques with the commercial introduction of Solid Phase Extraction (SPE) disks in 1989 [2]. This innovation addressed significant limitations of conventional SPE cartridges, particularly for environmental and bioanalytical applications requiring processing of large volume or particulate-rich samples. Whereas traditional cartridge-based SPE utilized packed beds of sorbent material, the disk format incorporated sorbent particles within an inert, web-like matrix [2], creating a revolutionary platform that combined the enrichment capabilities of SPE with the filtration characteristics of a membrane. This technological advancement emerged within the broader context of SPE's historical development, which had evolved from early uses of activated carbon in the 1940s to the introduction of pre-filled cartridges in 1978 [49]. The disk format represented the next logical step in this progression, specifically engineered to overcome the flow rate restrictions and plugging issues that hampered cartridges when processing challenging sample matrices [2]. For researchers handling large volume environmental waters or complex biological fluids, SPE disks delivered practical improvements in throughput and reliability that fundamentally expanded the application boundaries of solid-phase extraction techniques.
Technical Limitations of Pre-1989 SPE Cartridges
Prior to the introduction of SPE disks, the sample preparation landscape was dominated by cartridge-based SPE systems that presented several operational constraints, particularly for large-scale or matrix-rich applications.
Flow Rate and Throughput Limitations: Traditional cartridges featured a partially small cross-section that resulted in sluggish flow rates, dramatically increasing processing time for large sample volumes [2]. The packed bed design created significant flow resistance, forcing analysts to choose between prolonged processing times or potential analyte breakthrough from excessive pressure.
Plugging and Channeling Issues: The granular sorbent beds in cartridges were highly susceptible to plugging when confronted with particulate-rich samples, such as surface waters, wastewater, or biological homogenates [2]. This problem was exacerbated by the tendency for channeling to occur within the packed bed, where samples would create preferential pathways through the sorbent, resulting in incomplete utilization of the sorbent bed and variable recovery rates [2].
Volume Handling Constraints: Cartridge designs featured a tremendous void volume within the packed bed, requiring careful optimization of elution solvents to achieve adequate concentration factors [2]. This limitation was particularly problematic for trace analysis where high concentration factors were necessary to achieve detectable analyte levels.
Table 1: Primary Limitations of Pre-Disk SPE Cartridges
| Limitation Category | Specific Technical Challenges | Impact on Analytical Workflow |
|---|---|---|
| Flow Dynamics | Sluggish flow rates; Small cross-sectional area | Extended processing time; Limited sample throughput |
| Matrix Compatibility | Plugging with particulate-rich samples; Channeling effects | Frequent cartridge failure; Variable recovery rates |
| Volume Handling | Large void volumes; Inefficient elution | Reduced concentration factors; Compromised sensitivity |
| Practical Constraints | Labor-intensive processing; Costly for large samples | Increased analysis cost; Limited application scope |
SPE Disk Technology: Design and Operational Advantages
The SPE disk architecture fundamentally reimagined the physical configuration of extraction media by embedding sorbent particles within an inert fibrous or porous matrix [2], rather than packing them loosely into a column. This integrated design delivered transformative practical advantages that addressed the core limitations of cartridge-based systems.
Structural Innovation and Performance Benefits
The disk format incorporated approximately the same mass of sorbent material as conventional cartridges (typically 4-200 mg) but distributed it within a thin, wide membrane [2]. This geometrical innovation created a significantly larger cross-sectional area relative to the bed mass, which directly translated to two key benefits: higher applicable flow rates without channeling and reduced pressure requirements for processing viscous samples [2]. The embedded sorbent design also minimized the void volume issues that plagued cartridge elution, enabling more efficient analyte recovery with smaller solvent volumes. Perhaps most importantly for environmental applications, the integrated matrix provided filtration capability that allowed direct processing of particulate-containing samples without pre-filtration steps [2], streamlining the workflow and reducing contamination risks.
Comparative Technical Specifications
The performance differences between disk and cartridge formats manifested in quantifiable metrics that directly impacted analytical efficiency, particularly for large volume samples.
Table 2: Performance Comparison: SPE Disks vs. Traditional Cartridges
| Performance Parameter | SPE Disks | SPE Cartridges | Practical Implications |
|---|---|---|---|
| Cross-Sectional Area | Large | Small | Higher flow rates possible with disks |
| Flow Rates | Fast | Sluggish | Significantly reduced processing time for disks |
| Bed Mass | 4-200 mg | 4-30 mg | Comparable capacity in different formats |
| Void Volume | Smaller | Larger | More efficient elution with disks |
| Particulate Tolerance | High (integrated filtration) | Low (prone to plugging) | Disks handle turbid samples directly |
| Applicable Sample Volume | Up to 1L | 500 μL - 50 mL | Disks superior for large volumes |
| Elution Volume | Small | Larger | Better concentration factors with disks |
Application Domains: Revolutionizing Large Volume and Complex Matrix Analysis
The introduction of SPE disks found immediate application in fields where sample volume or matrix complexity had previously limited analytical efficiency, particularly in environmental monitoring and biological analysis.
Environmental Water Analysis
SPE disks demonstrated exceptional utility for monitoring trace organic pollutants in environmental waters, where analysts routinely needed to process liter-sized samples to achieve detectable concentration levels [50] [2]. The disk format enabled rapid extraction of diverse compound classes including halogenated hydrocarbons, chlorinated pesticides, polychlorinated biphenyls (PCBs), polycyclic aromatic hydrocarbons (PAHs), and phthalic esters from river water, lakes, and other surface waters [50]. The high flow rates possible with disks (up to 40 mL/min in some optimized systems) dramatically reduced processing time [51], with one study demonstrating that a 0.5L sample that previously required 50 minutes could be processed in just 12 minutes - a 76% reduction in preparation time [51]. This throughput improvement was particularly valuable for regulatory compliance monitoring where laboratories needed to process numerous samples within tight deadlines.
Biological and Pharmaceutical Applications
In the pharmaceutical and clinical domains, SPE disks offered advantages for processing biological fluids including plasma, serum, and urine [2]. The integrated filtration capability allowed direct handling of minimally processed samples, reducing preparation time and potential analyte loss. While 96-well plates eventually became dominant for high-throughput bioanalysis, disks found niche applications where larger sample volumes or particularly challenging matrices required their unique combination of filtration and extraction [2].
Practical Implementation: Methodology and Workflow
The effective utilization of SPE disks follows a systematic workflow that leverages their unique physical properties while maintaining the fundamental principles of solid-phase extraction.
Standardized Experimental Protocol
The following methodology outlines a generalized protocol for implementing SPE disk extraction for large volume water samples, adaptable to specific analytical requirements:
Disk Conditioning: Pre-wet the disk with 5-10 mL of methanol followed by 5-10 mL of reagent water to activate the sorbent surface. Maintain a small solvent layer above the disk to prevent drying [6].
Sample Loading: Transfer the sample to the extraction apparatus. For large volumes (>500 mL), apply positive pressure or vacuum to maintain flow rates of 10-100 mL/min, depending on disk diameter and sorbent mass [51]. Monitor flow to ensure consistent processing.
Interference Removal: After sample loading, rinse with 5-20 mL of a mild solvent (typically 5-20% methanol in water) to remove weakly retained matrix components. Adjust pH and solvent strength based on analyte properties [6].
Disk Drying: Apply vacuum or positive pressure for 5-20 minutes to remove residual water. This step is particularly important when switching to water-immiscible elution solvents [51].
Analyte Elution: Pass 5-15 mL of appropriate elution solvent (e.g., methanol, acetonitrile, or optimized mixtures) through the disk. Collect eluate in a calibrated vessel. Often, two smaller aliquots are more efficient than one large volume [6].
Sample Concentration (Optional): Gently evaporate the eluate under nitrogen stream and reconstitute in smaller volume compatible with subsequent analysis to enhance sensitivity [13].
Research Reagent Solutions for SPE Disk Applications
Successful implementation of SPE disk technology requires appropriate selection of supporting reagents and materials tailored to specific analytical challenges.
Table 3: Essential Research Reagents for SPE Disk Applications
| Reagent/Material | Function/Purpose | Application Notes |
|---|---|---|
| C18 Embedded Disks | Reversed-phase extraction of non-polar analytes | Most common format; ideal for PAHs, PCBs, pesticides from water [13] |
| Hydrophilic-Lipophilic Balanced (HLB) Disks | Mixed-mode retention for polar and non-polar compounds | Superior for pharmaceuticals, pesticides with wide polarity range [51] |
| Ion Exchange Disks | Selective extraction of ionic compounds | Targeted extraction of acidic/basic compounds under pH control [13] |
| Methanol (HPLC Grade) | Disk conditioning and analyte elution | Primary solvent for reversed-phase elution; compatible with HPLC analysis [6] |
| Reagent Water | Sample dilution and wash solutions | Essential for maintaining proper solvent strength during sample loading [6] |
| In-Line Sand Filters | Particulate removal for challenging matrices | Prevents disk clogging with turbid environmental samples [51] |
| pH Adjustment Reagents | Optimize ionization state for target analytes | Buffers, acids, bases to control retention/elution of ionizable compounds [6] |
Impact and Contemporary Relevance
The introduction of SPE disks represented more than merely a new commercial product; it fundamentally expanded the capabilities of solid-phase extraction and influenced subsequent developments in sample preparation technology. By enabling efficient processing of large volumes (up to 1L or more) and tolerant handling of particulate-rich matrices [2], disks opened new application areas that were impractical with cartridge-based systems. The technology's legacy is evident in contemporary sample preparation approaches, including the 96-well plate formats that dominate high-throughput bioanalysis [2] and the continued specialization of SPE media for challenging applications. Modern environmental monitoring protocols for emerging contaminants, including pharmaceuticals, per- and polyfluoroalkyl substances (PFAS), and pesticide residues [13], continue to leverage the fundamental advantages of disk-based extraction, particularly when processing the large sample volumes necessary to achieve low method detection limits. The disk platform also provided the conceptual foundation for subsequent innovations in solid-phase microextraction (SPME) [2] and related techniques that further miniaturized and streamlined sample preparation. For today's researcher, understanding the capabilities and implementation requirements of SPE disks remains essential when designing methods for trace analysis in complex matrices, particularly in environmental and food safety applications where large sample volumes are prerequisite for achieving necessary sensitivity.
The development of solid-phase extraction (SPE) represents a pivotal chapter in the history of analytical chemistry. The first experimental applications of SPE began approximately five decades ago with the use of activated carbon filters for trace organic analysis in water [49] [18]. This technique emerged from the fundamental recognition that solid surfaces could bind organic molecules through various affinity mechanisms, a principle observed in chromatographic techniques dating back to Tswett at the turn of the twentieth century [18]. The 1970s marked a significant transition with the introduction of pre-packed cartridges containing silica-based sorbents, notably the 1977 commercial introduction of the Sep-Pak cartridge, which repurposed liquid chromatography technology for sample preparation [2] [19]. This innovation initiated the shift from liquid-liquid extraction to more efficient, solvent-minimized solid-phase techniques, establishing SPE as a cornerstone of modern analytical methodology.
The latest evolutionary step involves the development of monolithic sorbents, which offer transformative advantages over traditional particle-packed systems. These continuous porous structures circumvent inherent limitations of particulate beds, including high backpressure and flow resistance, while enabling unprecedented control over selectivity and permeability [52] [53]. This technical guide examines how advanced monolithic materials and functionalization strategies are addressing core challenges in analytical throughput and selectivity within contemporary research environments.
Fundamental Principles and Historical Trajectory
Core Principles of Solid-Phase Extraction
SPE operates on the fundamental principle of distributing analytes between a liquid sample phase and a solid sorbent phase [2]. The process involves multiple stages: sorbent conditioning, sample application, interference removal, and analyte elution. Traditional particle-based SPE utilizes beds of granular material packed into cartridges, where separation occurs through differential affinity based on chemical interactions between the analyte and functionalized sorbent surfaces [2]. The technique provides significant advantages over liquid-liquid extraction, including reduced organic solvent consumption, elimination of emulsion formation, and greater procedural simplicity [2].
The Trajectory to Monolithic Architectures
The development of SPE materials has progressed through several distinct phases:
- The Age of Active Carbon (1950s-1960s): Initial applications utilized activated carbon for concentrating organic contaminants from water, though recovery was often complicated by strong, non-specific adsorption [18].
- The Search for Optimal Materials (1970s-1980s): Research expanded to include XAD resins, bonded silicas, and polymeric sorbents, seeking improved specificity and recovery [18].
- Technical Refinement (1980s-1990s): Standardization of cartridge formats and automation compatibility became focal points [18].
- The Monolithic Era (1990s-Present): The introduction of continuous porous polymer networks addressed fundamental limitations of particulate systems [2].
Monolithic sorbents represent a paradigm shift from packed particle technology. Their continuous porous structure typically comprises macropores (1-10 μm) that facilitate convective mass transfer at low backpressure, and mesopores (10-50 nm) that provide high surface area for analyte interaction [52]. This dual-scale porosity enables rapid binding kinetics and high flow rates without the channeling effects that can plague packed beds [53].
Table 1: Comparative Performance Characteristics of SPE Formats
| Parameter | Particle-Based SPE | Monolithic SPE |
|---|---|---|
| Backpressure | High, especially with small particles | Low due to flow-through pores |
| Flow Rates | Limited by pressure buildup | High, suitable for viscous samples |
| Mass Transfer | Diffusion-limited | Convection-enhanced |
| Selectivity | Dependent on particle chemistry | Tunable through functionalization |
| Manufacturing | Well-established packing processes | In-situ polymerization |
| Scale-down Potential | Limited by packing consistency | Excellent for microformats |
Advanced Monolithic Materials and Functionalization Strategies
Monolithic Substrate Compositions
Modern monolithic sorbents are fabricated from both organic and inorganic precursors, each offering distinct advantages:
- Organic Polymer Monoliths: Typically synthesized from methacrylate polymers (butyl-, lauryl-, octadecyl-, or 2-hydroxyethyl methacrylate) via free-radical polymerization within confined molds [53]. These materials provide hydrophobic character suitable for reversed-phase SPE applications, with demonstrated high capacity, porosity, and permeability [53].
- Silica-Based Monoliths: Fabricated through sol-gel processes, creating siloxane networks with hierarchical pore structures [54]. These substrates offer superior mechanical stability and surface functionality for further modification.
- Organic-Inorganic Hybrids: Combine the flexibility of organic polymers with the mechanical strength of silica, enabling tailored properties for specific applications [52].
Selectivity-Enhancing Functionalization Approaches
Surface functionalization transforms inert monolithic scaffolds into selective extraction media through several advanced strategies:
Biomimetic Recognition Elements
- Aptamer-Functionalized Monoliths: Synthetic oligonucleotides with specific three-dimensional structures provide exceptional molecular recognition capabilities. Aptamers offer advantages over antibodies, including ease of synthesis, rapid regeneration, and efficient target capture [55]. For example, gold nanoparticles (AuNPs) have been employed to increase aptamer coverage density to 3,636 pmol μl−1, significantly enhancing extraction yield for target analytes like zearalenone [55].
- Antibody and Peptide Modification: Biomolecules with high affinity for targeted analytes can be covalently grafted onto monolithic surfaces. Pore size and specific surface area must be carefully controlled to facilitate effective grafting while minimizing non-specific interactions [52].
Molecular Imprinting
Molecularly Imprinted Polymers (MIPs) create synthetic recognition sites through template-directed synthesis [52]. The process involves:
- Complexation of template molecules with functional monomers via non-covalent interactions
- Polymerization with cross-linking agents around the template
- Template removal to create cavities complementary in size, shape, and functional group positioning [52]
MIP-monolith hybrids enable selective extraction that eliminates matrix effects, particularly valuable in LC-MS applications where interference can compromise analysis [52].
Nanomaterial Integration
- Carbon Nanotubes and Graphene Oxide: Provide ultra-high specific surface area and mechanical strength. Graphene oxide composites with silica nanoparticles (GO-SiO₂ NPs) have demonstrated enhanced separation efficiency for complex samples [55].
- Metal-Organic Frameworks (MOFs) and Covalent Organic Frameworks (COFs): Porous crystalline materials that significantly increase specific surface area, particularly valuable in miniaturized formats where available surface area is limited [52].
- Metal Nanoparticles: Functional nanoparticles like gold and iron oxide provide modification platforms for further functionalization while enhancing mechanical properties [55].
Supramolecular Recognition Systems
Macrocyclic compounds like crown ethers enable metal ion recognition through host-guest interactions. These systems demonstrate exceptional selectivity for specific ions, such as lead (Pb²⁺), even in complex matrices with competing ions [53].
Table 2: Functionalization Strategies for Monolithic Sorbents
| Functionalization Approach | Key Materials | Selectivity Mechanism | Representative Applications |
|---|---|---|---|
| Biomimetic Aptamers | Synthetic oligonucleotides | 3D structural complementarity | Mycotoxins, proteins |
| Molecular Imprinting | Cross-linked polymers | Shape-specific cavities | Pharmaceuticals, biomarkers |
| Nanomaterial Composites | Graphene, MOFs, CNTs | Enhanced surface area | Trace contaminant enrichment |
| Supramolecular Receptors | Crown ethers, cyclodextrins | Host-guest chemistry | Metal ions, chiral compounds |
| Biomolecule Grafting | Antibodies, peptides | Affinity interactions | Biomarkers, pathogens |
Experimental Protocols and Performance Analysis
Protocol: Monolithic SPE for Selective Lead Separation
Recent research demonstrates the efficacy of monolithic SPE for selective metal ion separation [53]. The following protocol details the optimization of a crown ether-functionalized monolithic column for lead extraction:
Materials and Instrumentation
- Monolithic Columns: Supramolecule-equipped monoliths (AnaLig Pb-02) in appropriate housing [53]
- Standards and Reagents: Pb standard solution (1000 mg L⁻¹), buffer solutions (pH 3-10), ethylenediaminetetraacetic acid (EDTA) eluent [53]
- Instrumentation: ICP-OES or ICP-MS for detection, pH meter, vacuum manifold system [53]
Optimization Parameters
- Solution pH: Vary pH from 3.0 to 10.0 using appropriate buffer systems
- Flow Rate: Test range from 0.5 to 5.0 mL/min under vacuum
- Eluent Selection: Evaluate EDTA concentration (0.01-0.1 M) and volume
- Interference Studies: Assess matrix effects with competing ions (Li, Na, Mg, K, Ca, Sr, Ba, Fe)
Procedure
- Conditioning: Pre-wet monolith with 5 mL methanol, then 5 mL pH-optimized buffer
- Sample Loading: Load aqueous sample at determined optimal flow rate
- Washing: Rinse with 5-10 mL optimized buffer to remove interferents
- Elution: Treat with 5-10 mL EDTA solution to recover bound Pb²⁺
- Analysis: Quantify eluate via ICP-OES/MS
- Regeneration: Recondition column for subsequent uses [53]
Performance Comparison: Monolithic vs. Particle-Based SPE
Direct comparison of identically functionalized monolithic and particle-packed SPE columns reveals distinct performance advantages:
Quantitative analysis demonstrates that monolithic columns achieve equivalent or superior recovery rates (approximately 77% for non-polar compounds) compared to particle-based columns (approximately 69% for octadecyl silica phases) while significantly reducing processing time [2]. The enhanced permeability of monolithic structures enables flow rates 5-10 times higher than particle-packed systems at equivalent backpressure [53].
Applications Across Research Domains
Biomedical and Pharmaceutical Analysis
Monolithic sorbents functionalized with biomimetic recognition elements enable selective extraction of biomarkers, pharmaceuticals, and metabolites from complex biological matrices [52] [55]. Specific applications include:
- Therapeutic Drug Monitoring: Molecularly imprinted monoliths selectively extract target pharmaceuticals from plasma and urine, eliminating matrix effects in LC-MS analysis [52].
- Biomarker Discovery: Aptamer-functionalized monolithic microextraction devices concentrate low-abundance proteins and metabolites from limited-volume biological samples [55].
- Proteomic Applications: Monoliths with immobilized enzymes or affinity ligands (Protein A/G) enable efficient proteolytic digestion and antibody purification in integrated processing workflows [52].
Environmental Monitoring
The low backpressure and high permeability of monolithic sorbents make them ideal for processing large-volume environmental samples:
- Water Quality Assessment: Online SPE-LC systems with monolithic extraction columns continuously monitor trace organic contaminants (pesticides, pharmaceuticals, endocrine disruptors) in water with minimal solvent consumption [52].
- Trace Metal Analysis: Supramolecule-equipped monolithic columns selectively preconcentrate heavy metals (Pb, Cd, Hg) from complex aqueous matrices prior to ICP-MS detection [53].
Food Safety and Natural Products
- Mycotoxin Detection: Affinity monoliths with aptamer or MIP functionalities extract and concentrate mycotoxins (ochratoxin A, zearalenone) from food extracts, achieving detection limits in the sub-ppb range [55].
- Active Compound Analysis: Monolithic SPE clean-up of natural product extracts removes interfering pigments and matrix components prior to quantitative analysis of active constituents [55].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents and Materials for Monolithic Sorbent Research
| Material/Reagent | Function | Application Examples |
|---|---|---|
| Methacrylate Monomers | Monolithic polymer backbone | Base matrix for organic polymer monoliths |
| Cross-linking Agents | Create porous polymer network | Controlling macropore/mesopore ratio |
| Aptamer Sequences | Molecular recognition elements | Selective extraction of small molecules |
| Molecular Templates | Create specific binding cavities | MIP synthesis for target analytes |
| Silica Nanoparticles | Enhance surface area | Composite monolith fabrication |
| Gold Nanoparticles | Aptamer immobilization platforms | Increased biomolecule density |
| Crown Ether Derivatives | Metal ion recognition | Selective heavy metal extraction |
| Ionic Liquid Monomers | Stationary phase modification | Enhanced thermal stability |
Future Perspectives and Research Directions
The evolution of monolithic sorbents continues with several emerging trends shaping future development:
- Multifunctional Composites: Integration of multiple functionality types (e.g., aptamer-MIP hybrids) creates sorbents with orthogonal recognition mechanisms for enhanced selectivity [55].
- Miniaturization and Chip Integration: Monolithic synthesis within microfluidic devices enables lab-on-a-chip analytical systems with integrated sample preparation, separation, and detection [52].
- Green Sorbent Development: Increasing emphasis on bio-based materials and sustainable synthesis routes aligns with green analytical chemistry principles [56].
- Advanced Manufacturing Techniques: 3D printing of monolithic structures with spatially controlled functionality represents the next frontier in sorbent design and customization [52].
- High-Throughput Automation: Monolithic 96-well plate formats and online SPE-LC systems accelerate analytical workflows in pharmaceutical and clinical applications [2].
Monolithic sorbents have fundamentally transformed the solid-phase extraction landscape, bridging five decades of SPE evolution with innovative solutions to persistent challenges in analytical science. Their unique combination of permeability, selectivity, and format flexibility positions these advanced materials as enabling tools for next-generation analytical methodologies across research domains.
The early development of solid phase extraction (SPE) techniques marked a pivotal advancement in analytical chemistry, enabling researchers to isolate and concentrate target analytes from complex matrices. As a cornerstone sample preparation method, SPE research has evolved to address the challenges of detecting trace-level compounds in environmental, pharmaceutical, and clinical samples. The core principle involves selectively retaining analytes on a sorbent material while removing interfering matrix components, thereby significantly enhancing the sensitivity and specificity of subsequent analytical determinations [57]. This technical guide explores how these foundational SPE techniques underpin modern analytical workflows across three critical application fields, with particular emphasis on current methodologies, experimental protocols, and computational approaches that have built upon these early developments.
Analytical Fundamentals: Instrumentation and Techniques
Core Analytical Instrumentation
The identification and quantification of trace contaminants and biomarkers rely heavily on advanced hyphenated techniques that separate complex mixtures and provide sensitive detection.
Table 1: Core Analytical Techniques for Target Applications
| Application Field | Primary Analytical Technique | Key Advantages | Typical Sensitivity Range |
|---|---|---|---|
| Environmental Pollutants | Liquid Chromatography-Tandem Mass Spectrometry (LC-MS/MS) [57] | High selectivity for polar compounds, multi-residue capability | ng/L to µg/L |
| Pharmaceutical Compounds | Liquid Chromatography-Mass Spectrometry (LC-MS) [58] | Broad applicability, structural information, high throughput | pg/mL to ng/mL |
| Clinical Diagnostics | Ultra Performance Liquid Chromatography-MS/MS (UPLC-MS/MS) [58] | Rapid analysis, high resolution, minimal sample volume | pg/mL |
The Centrality of Sample Preparation: Solid Phase Extraction
SPE remains the pre-concentration technique of choice for complex environmental, pharmaceutical, and biological samples [57]. Its evolution has been characterized by the development of novel sorbents (e.g., mixed-mode, molecularly imprinted polymers) that improve selectivity and recovery rates. The general SPE workflow involves four key steps: 1) Condition the sorbent bed, 2) Load the sample matrix, 3) Wash away interfering components, and 4) Elute the purified analytes. The choice of sorbent chemistry (e.g., C18 for reversed-phase, silica for normal-phase, ion-exchange resins) is critical and depends on the physicochemical properties of the target analytes [57].
Application Field 1: Environmental Pollutants
Scope and Challenges
Environmental analysis focuses on detecting emerging contaminants (ECs), notably pharmaceuticals and personal care products (PPCPs), in aqueous and solid matrices like wastewater, surface water, sediments, and soils [57]. These compounds, including antibiotics, analgesics, hormones, and UV filters, are typically present at trace concentrations (ng/L) and exhibit "pseudo-persistent" characteristics due to continuous introduction into ecosystems [57]. Major challenges include the complexity of environmental matrices and the need to detect ultra-low analyte concentrations amidst significant background interference.
Detailed Experimental Protocol: SPE-LC-MS/MS for PPCPs in Water
Sample Collection and Preservation:
- Collect water samples (1 L) in amber glass bottles to prevent photodegradation.
- Acidify to pH ~2 with hydrochloric acid or add preservatives (e.g., sodium azide) to inhibit microbial activity.
- Store at 4°C and process within 24-48 hours [57].
Solid Phase Extraction Procedure:
- SPE Cartridge: Use a mixed-mode reversed-phase/cation exchange sorbent (e.g., Oasis MCX, 60 mg/3 mL).
- Conditioning: Sequentially pass 3-5 mL methanol and 3-5 mL acidified water (pH 2) through the cartridge.
- Sample Loading: Load the water sample at a controlled flow rate of 5-10 mL/minute.
- Washing: Rinse with 2-3 mL of a mild acid solution (e.g., 2% formic acid in water) to remove interferences.
- Drying: Apply vacuum or positive pressure for 10-15 minutes to dry the sorbent.
- Elution: Pass 4-6 mL of a mixture of methanol and ammonium hydroxide (98:2, v/v) to elute the target analytes.
- Concentration: Evaporate the eluate to near dryness under a gentle nitrogen stream at 40°C and reconstitute in 100-200 µL of initial mobile phase for LC-MS/MS analysis [57].
Instrumental Analysis via LC-MS/MS:
- Chromatography: Reverse-phase C18 column (100 mm x 2.1 mm, 1.8 µm); Mobile phase A: 0.1% formic acid in water; B: 0.1% formic acid in acetonitrile; Gradient elution.
- Mass Spectrometry: Electrospray ionization (ESI) in positive/negative switching mode; Multiple Reaction Monitoring (MRM) for quantitation; Use isotopically labeled internal standards for each analyte to correct for matrix effects [58].
Application Field 2: Pharmaceutical Compounds
Scope and Challenges
Pharmaceutical analysis encompasses quality control of drug products, bioavailability studies, and monitoring stability and degradation products. The primary challenges include ensuring method specificity for the active pharmaceutical ingredient (API) amidst excipients and quantifying multiple metabolites with diverse chemical properties.
Detailed Experimental Protocol: SPE for Plasma Drug Concentrations
Sample Pretreatment:
- Thaw frozen plasma samples and vortex mix.
- Aliquot 100 µL of plasma into a microcentrifuge tube.
- Add 300 µL of acetonitrile containing internal standards for protein precipitation.
- Vortex vigorously for 30 seconds and centrifuge at 14,000 x g for 10 minutes [58].
SPE Procedure for Clean-up:
- Use a 96-well SPE plate (e.g., Oasis HLB, 30 µm) for high-throughput processing.
- Condition wells with 1 mL methanol followed by 1 mL water.
- Load the supernatant from the protein precipitation step.
- Wash with 1 mL of 5% methanol in water.
- Elute analytes with 1 mL of methanol.
- Evaporate eluates and reconstitute in 150 µL of mobile phase for LC-MS analysis [58].
Instrumental Analysis:
- Chromatography: HSS T3 column (2.1 x 100 mm, 1.8 µm); Mobile phase: acetonitrile and ammonium formate buffer; Gradient elution over 8 minutes.
- Mass Spectrometry: ESI positive mode; MRM transitions optimized for each pharmaceutical compound; Quadrupole mass analyzer for high sensitivity quantitation [58].
Application Field 3: Clinical Diagnostics
Scope and Challenges
Clinical diagnostics involves quantifying biomarkers, hormones, and drugs in biological fluids (serum, urine) for therapeutic drug monitoring and disease diagnosis. Key challenges include dealing with complex biological matrices and achieving the required sensitivity for low-abundance biomarkers.
Detailed Experimental Protocol: SPE for Serum Biomarkers
Sample Preparation:
- Allow serum samples to clot and centrifuge at 2,000 x g for 10 minutes.
- Aliquot 200 µL of serum and dilute with 400 µL of 100 mM ammonium acetate buffer (pH 4.5) [58].
SPE Procedure:
- Use a C18 SPE micro-column (10 mg/1 mL).
- Condition with 1 mL methanol and 1 mL ammonium acetate buffer (pH 4.5).
- Load the diluted serum sample.
- Wash with 1 mL of 5% methanol in water.
- Elute with 0.5 mL of 80:20 methanol:water with 0.1% formic acid.
- Inject directly into the LC-MS/MS system [58].
Instrumental Analysis:
- Chromatography: BEH C18 column (2.1 x 50 mm, 1.7 µm); Mobile phase: methanol and 0.1% formic acid in water; Fast gradient (3-4 minutes).
- Mass Spectrometry: ESI positive mode; High-resolution mass spectrometry (HRMS) on a Q-TOF or Orbitrap platform for accurate mass measurement; Data-independent acquisition (DIA) for untargeted biomarker discovery [58].
Advanced Topics: Computational and In Silico Approaches
(Q)SAR Models for Environmental Fate Prediction
Computational toxicology tools are increasingly important for predicting the environmental fate of chemicals, especially with regulatory bans on animal testing for cosmetics.
Table 2: Recommended (Q)SAR Models for Cosmetic Ingredient Assessment
| Environmental Fate Parameter | Recommended (Q)SAR Model | Software Platform | Primary Application |
|---|---|---|---|
| Persistence / Biodegradation | Ready Biodegradability IRFMN [59] | VEGA | Classification of ready biodegradability |
| BIOWIN [59] | EPISUITE | Predicting ultimate biodegradation | |
| Bioaccumulation (Log Kow) | ALogP [59] | VEGA | Lipophilicity estimation |
| KOWWIN [59] | EPISUITE | Octanol-water partition coefficient | |
| Bioaccumulation (BCF) | Arnot-Gobas Model [59] | VEGA | Bioconcentration factor in fish |
| KNN-Read Across [59] | VEGA | BCF prediction using similarity | |
| Mobility (Log Koc) | OPERA v.2.0 [59] | VEGA | Soil adsorption coefficient |
| KOCWIN [59] | EPISUITE | Soil/water partitioning |
These in silico tools help researchers prioritize chemicals for further testing and fill data gaps in environmental risk assessments, aligning with the 3Rs principle (Replacement, Reduction, Refinement) in toxicology [59].
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Research Reagents and Materials for SPE-Based Analysis
| Reagent/Material | Function | Example Applications |
|---|---|---|
| Mixed-mode SPE sorbents (e.g., Oasis MCX, WCX) | Simultaneous retention via reversed-phase and ion-exchange mechanisms; superior clean-up for complex matrices. | Extraction of acidic/ basic pharmaceuticals from wastewater and plasma [57]. |
| Isotopically Labeled Internal Standards (e.g., ¹³C, ²H) | Correct for analyte loss during sample preparation and matrix effects during ionization in MS. | Quantitation of opioids in plasma, PPCPs in environmental waters [58]. |
| Enzymatic Digestion Reagents (e.g., β-glucuronidase) | Hydrolyze conjugated metabolites back to the parent compound for accurate total concentration measurement. | Analysis of glucuronidated drug metabolites in urine. |
| Matrix Modification Reagents (e.g., EDTA, Formic Acid) | Preserve analyte stability, prevent degradation, and optimize retention on SPE sorbents by adjusting pH/ionic strength. | Chelating metals in water samples; acidifying to prevent dissociation of acidic drugs [57]. |
| High-purity Elution Solvents (e.g., LC-MS Grade MeOH, ACN) | Efficiently elute retained analytes from SPE without introducing interferences that affect MS detection. | Final elution step in SPE for all application fields prior to LC-MS analysis [58]. |
Visualized Workflows and Signaling Pathways
Analytical Workflow for Environmental and Biological Samples
Environmental Pathway and Bioanalytical Impact of PPCPs
Troubleshooting and Optimization: Solving Common SPE Challenges in the Lab
Solid-phase extraction (SPE) serves as a cornerstone technique in modern analytical sample preparation, enabling the purification, concentration, and isolation of analytes from complex matrices. Its fundamental principle involves the selective partitioning of analytes between a liquid sample and a solid stationary phase [2]. Despite its widespread adoption across pharmaceutical, environmental, and biological applications, method development and optimization persistently challenge researchers, with poor analyte recovery representing a predominant obstacle that compromises analytical accuracy, sensitivity, and reproducibility [60] [61].
This technical guide frames poor recovery within the context of early SPE development research, where understanding fundamental interactions between analyte, sorbent, and solvent is paramount. Recovery failures manifest as analyte loss during three critical phases: loading, washing, and elution. Systematically investigating these fractions is not merely a troubleshooting exercise but a fundamental research activity that reveals the underlying physicochemical interactions governing extraction efficiency [62]. For drug development professionals, optimizing these parameters is essential for developing robust, reliable analytical methods that accurately quantify compounds in complex matrices such as plasma, urine, and tissue homogenates [63] [6].
Systematic Diagnosis of Recovery Losses
Before implementing corrective measures, researchers must first identify the specific stage where analyte loss occurs. The following diagnostic workflow provides a systematic approach for pinpointing the failure point, incorporating fractional analysis as a core investigative methodology [60].
Diagnostic Workflow for Recovery Failures
The diagram below outlines a step-by-step diagnostic protocol to isolate the cause of poor recovery in SPE methods.
Experimental Protocol for Fraction Analysis
Implementing the diagnostic workflow requires a standardized experimental approach to collect and analyze fractions from each SPE step [60]:
- Standard Process Execution: Perform the SPE method exactly as developed, using a spiked sample with known analyte concentration.
- Fraction Collection: Separately collect the liquid that flows through the sorbent during the loading step (load fraction), the solvent from the wash step (wash fraction), and the solvent from the elution step (elution fraction).
- Fraction Analysis: Analyze each collected fraction using the intended analytical method (e.g., LC-MS, GC-MS) to determine where the analyte is detected.
- Data Interpretation: The presence of a significant analyte peak in the load fraction indicates poor retention during loading. Detection in the wash fraction shows the wash solvent is too strong. A weak or absent peak in the elution fraction confirms inadequate elution.
Analyzing and Addressing Losses in Specific Fractions
Loading Phase: Incomplete Analyte Retention
Analyte detection in the loading flow-through fraction indicates failure to be retained by the sorbent. The table below summarizes common causes and research-driven solutions.
Table 1: Troubleshooting Analyte Loss During the Loading Phase
| Cause of Loss | Underlying Principle | Research-Grounded Solution |
|---|---|---|
| Incompatible Sample Solvent | Sample solvent strength competes with/overcomes analyte-sorbent interactions [61]. | Dilute sample with water or a weak solvent; use solvent exchange to aqueous buffer for reversed-phase SPE [6]. |
| Incorrect Sample pH | Analyte exists in uncharged state, minimizing ionic interactions with ion-exchange sorbents [62]. | Adjust sample pH to ensure analyte is ionized: for acidic analytes, set pH ≥ pKa + 2; for basic analytes, set pH ≤ pKa - 2 [62] [61]. |
| Sorbent Overload | Sample mass or interference concentration exceeds sorbent capacity (~1-5% of sorbent mass) [62] [6]. | Reduce sample load mass; dilute sample; increase sorbent bed mass [6]. |
| Sorbent Drying Post-Conditioning | Dry sorbent bed reduces accessibility of functional ligands and creates flow channels [6]. | Do not let sorbent run dry after conditioning; maintain ~1mm solvent above the top frit [6]. |
| Protein-Bound Analyte | In biological samples, analytes bound to proteins may pass through the sorbent unretained [60]. | Employ protein precipitation before SPE; adjust pH to disrupt binding; use ultrafiltration [60] [61]. |
Wash Phase: Premature Analyte Elution
Detection of the analyte in the wash fraction signifies that the wash solvent is too strong, prematurely eluting the target compound before the final elution step.
Table 2: Troubleshooting Analyte Loss During the Wash Phase
| Cause of Loss | Underlying Principle | Research-Grounded Solution |
|---|---|---|
| Excessive Organic Solvent in Wash | High organic modifier percentage in wash buffer increases solvent strength in reversed-phase SPE, displacing analytes [61]. | Start with a mild wash (e.g., 5-20% methanol or acetonitrile in water) and optimize; ensure solvent strength remains weak [61]. |
| Incorrect Wash pH | pH adjustment during wash neutralizes analyte or sorbent charge, breaking ionic interactions in ion-exchange or mixed-mode SPE [62]. | Optimize wash buffer pH to maintain analyte charge while removing interferences. |
| Insufficient Selectivity | Wash solvent lacks specificity to remove matrix interferences without affecting the analyte [60]. | For non-polar interferences, use a small volume of a non-polar, water-immiscible solvent (e.g., hexane, ethyl acetate) [60]. |
Elution Phase: Incomplete Analyte Recovery
The absence or low level of analyte in the elution fraction indicates inadequate disruption of analyte-sorbent interactions.
Table 3: Troubleshooting Analyte Loss During the Elution Phase
| Cause of Loss | Underlying Principle | Research-Grounded Solution |
|---|---|---|
| Weak Elution Solvent Strength | Solvent lacks sufficient strength to overcome primary retention forces (e.g., van der Waals, ionic) [62] [61]. | Use a stronger solvent (e.g., methanol, acetonitrile, isopropanol). For mixed-mode, add acid/base (ion suppression) or salt (ionic disruption) [62] [61]. |
| Insufficient Elution Volume | The volume is too small to displace all retained analyte from the sorbent bed [6]. | Use 1-3 cartridge void volumes; elute with two small aliquots rather than one large one [6]. |
| Secondary Interactions | Analyte strongly interacts with residual silanols on silica-based sorbents or other active sites [60]. | Add modifiers to elution solvent (e.g., 0.1-1% TFA or formic acid for basic analytes; ammonium hydroxide for acidic analytes) [61]. |
| Strong/Irreversible Binding | The analyte is too strongly retained by the selected sorbent chemistry [60]. | Switch to a sorbent with weaker retention (e.g., C8 instead of C18, or a weak ion-exchange sorbent) [60] [61]. |
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful SPE method development relies on a foundational set of high-quality reagents and materials. The following table details key solutions for research and troubleshooting.
Table 4: Essential Research Reagents and Materials for SPE Method Development
| Item | Function & Application in Research |
|---|---|
| C18 Sorbent | Reversed-phase sorbent for extracting non-polar analytes from polar matrices; a versatile starting point for method scouting [62] [61]. |
| Mixed-Mode Sorbent (e.g., MCX, MAX) | Combines reversed-phase and ion-exchange mechanisms; provides superior selectivity for ionizable compounds in complex matrices like biological fluids [62] [61]. |
| HLB (Hydrophilic-Lipophilic Balance) Sorbent | Water-wettable polymeric sorbent for a broad spectrum of analytes; eliminates need for conditioning and prevents channeling [61]. |
| Strong Elution Solvents (MeOH, ACN) | Disrupt hydrophobic interactions in reversed-phase SPE; essential for complete analyte recovery [62] [61]. |
| pH Modifiers (e.g., FA, TFA, NH₄OH) | Acidic (FA, TFA) or basic (NH₄OH) modifiers are critical for controlling ionization in ion-exchange protocols and breaking secondary interactions [62] [61]. |
| Buffers (e.g., Phosphate, Acetate) | Used to precisely control sample pH during loading and washing to optimize retention and selectivity, particularly in ion-exchange SPE [62] [6]. |
| Non-Polar Wash Solvents (e.g., Hexane) | Selectively remove non-polar matrix interferences (e.g., lipids) without eluting polar analytes during reversed-phase SPE [60]. |
Advanced Considerations: Mechanistic Insights and Future Directions
Understanding SPE as a dynamic interplay of intermolecular forces rather than a simple procedure is key to advanced troubleshooting. The retention mechanism is a combination of hydrophobic, polar, and ionic interactions that must be selectively manipulated across the different SPE steps [62]. For instance, in mixed-mode SPE, both hydrophobic and ionic interactions must be sequentially broken for efficient elution, often requiring elution solvents that contain both an organic modifier and a pH modifier or salt [62].
The choice of sorbent is the foundational decision. The flowchart below outlines the sorbent selection logic based on analyte and matrix properties, a critical step in early method development.
Future directions in SPE research focus on sustainability and efficiency, aligning with the broader principles of green analytical chemistry. This includes automating SPE to save time, reduce solvent consumption, and improve reproducibility [64], developing miniaturized techniques like µ-SPE to drastically reduce solvent and sorbent usage [2], and creating new high-capacity and selective sorbents like molecularly imprinted polymers (MIPs) to improve recovery and clean-up from complex matrices [2]. A critical awareness of the "rebound effect" is also necessary, where a greener method (e.g., cheaper, less solvent) might lead to over-testing, negating its environmental benefits [64].
Systematic analysis of losses in loading, wash, and elution fractions transforms SPE from a black-box sample preparation technique into a rational, predictable component of the analytical method. This approach, grounded in the principles of early development research, empowers scientists to diagnose issues mechanistically and implement targeted solutions. As the field evolves toward more sustainable and efficient practices, a deep understanding of these core principles will remain essential for developing robust, reliable, and green analytical methods that accelerate drug development and scientific discovery.
In the context of early development research for solid phase extraction (SPE) techniques, reproducibility is not merely a desirable attribute but a fundamental requirement for generating reliable, scientifically valid data. SPE serves as a crucial sample preparation technique to isolate, purify, and concentrate analytes from complex matrices prior to chromatographic analysis [2] [13]. The growing emphasis on green analytical chemistry has accelerated the development of miniaturized SPE techniques, making efficient method development more critical than ever [65]. Within this framework, three technical parameters emerge as pivotal control points: conditioning, flow rates, and drying. These seemingly routine steps, when improperly executed, become primary sources of variability that can compromise analytical results, lead to erroneous conclusions, and hinder research progress. This technical guide examines the scientific principles behind these critical parameters and provides detailed methodologies to ensure reproducible SPE outcomes in early development research.
The Solid Phase Extraction Workflow: A Foundation for Understanding
Solid phase extraction operates on the fundamental principles of chromatography, utilizing differential affinities of compounds between a liquid mobile phase and a solid stationary phase [13]. The process exploits various intermolecular interactions—including van der Waals forces, hydrogen bonding, dipole-dipole interactions, and ionic interactions—to achieve separation [62] [13].
A typical SPE process consists of multiple interconnected steps where the output of each directly influences the success of subsequent stages. Understanding this workflow is essential for identifying critical control points where reproducibility can be ensured or lost.
The following diagram illustrates the standard SPE workflow and highlights where our three critical parameters—conditioning, flow rates, and drying—exert their influence:
Figure 1: SPE Workflow with Critical Control Points for Reproducibility
Conditioning: Establishing the Foundation for Reproducible Interactions
The Scientific Principle of Conditioning
Conditioning represents the first critical step in the SPE process, with the primary purpose of activating the sorbent bed and preparing it for optimal interaction with the sample [66] [13]. The fundamental scientific principle involves wetting the sorbent to expose accessible interaction sites and creating a stable chemical environment compatible with the sample matrix. Without proper conditioning, the sorbent may exhibit inconsistent retention characteristics due to insufficient wetting of hydrophobic surfaces or inappropriate pH and ionic conditions for the target analytes [62]. In reversed-phase SPE, for instance, which is commonly used for extracting non-polar to moderately polar compounds from polar matrices, the conditioning step typically employs a strong organic solvent followed by water or buffer to create the ideal environment for hydrophobic interactions [66] [13].
Detailed Conditioning Protocol
To ensure reproducible conditioning across experiments, follow this detailed protocol:
Solvent Selection: Pass 1–2 column volumes of a strong organic solvent (typically methanol or acetonitrile) through the sorbent bed [66]. This solvent serves to solvate the hydrophobic ligands on the sorbent surface (e.g., C18 chains) and displace any air trapped within the porous structure.
Aqueous Compatibility: Without allowing the sorbent to run dry, pass 1–2 column volumes of water or aqueous buffer (pH-matched to your sample) through the device [66]. This step creates an environment compatible with your aqueous sample, preventing premature precipitation of analytes and ensuring smooth sample flow.
Critical Consideration - Avoid Drying: The transition between conditioning and sample loading must occur without the sorbent bed drying out [66]. Drying at this stage causes the hydrophobic ligands to collapse, reducing the effective surface area and creating flow channels that bypass interaction sites. This inevitably leads to inconsistent analyte recovery and poor reproducibility.
Equilibration (When Required): For ion-exchange or mixed-mode mechanisms, an additional equilibration step with a buffer that matches the sample pH and ionic strength may be necessary to establish the proper ionic form of the sorbent [66].
Table 1: Conditioning Parameters by SPE Mechanism
| SPE Mechanism | Primary Conditioning Solvent | Secondary Conditioning Solvent | Critical Considerations |
|---|---|---|---|
| Reversed-Phase | Methanol (1-2 column volumes) | Water or aqueous buffer (1-2 column volumes) | Avoid drying; ensure complete wetting of hydrophobic surface [66] [13] |
| Normal-Phase | Non-polar solvent (e.g., hexane) | Typically not required | Keep environment anhydrous; control humidity [13] |
| Cation Exchange | Methanol, then water | Acidic buffer (pH 2-3 units below analyte pKa) | Ensure sorbent is in correct ionic form [62] |
| Anion Exchange | Methanol, then water | Basic buffer (pH 2-3 units above analyte pKa) | Ensure sorbent is in correct ionic form [62] |
| Mixed-Mode | Methanol (1-2 column volumes) | Water or aqueous buffer (1-2 column volumes) | May require additional buffer equilibration [62] |
Flow Rates: Controlling Interaction Time for Consistent Retention
The Impact of Flow Rate on Extraction Efficiency
Flow rate represents one of the most frequently overlooked yet critically important parameters for ensuring reproducible SPE results. The rate at which samples and solvents pass through the sorbent bed directly controls the interaction time between analytes and the stationary phase [66]. Excessive flow rates introduce two primary mechanisms of failure:
Insufficient Interaction Time: Analytes in the sample solution require adequate contact time with the sorbent particles to diffuse to the active sites and form stable interactions (e.g., hydrophobic, ionic) [66].
Channeling Effects: High flow rates can create preferential pathways through the sorbent bed, causing significant portions of the sample to bypass interaction sites entirely [2]. This effect is particularly pronounced in traditional loose-packed cartridges.
Optimizing Flow Rates for Reproducibility
The following table provides recommended flow rates for different SPE steps and configurations:
Table 2: Recommended Flow Rates for Reproducible SPE Performance
| SPE Step | Recommended Flow Rate | Scientific Rationale | Impact of Deviation |
|---|---|---|---|
| Sample Loading | 0.5–1 mL/min [66] | Maximizes analyte-sorbent interaction time for efficient retention | High rate: Breakthrough and low recoveryLow rate: Prolonged processing time |
| Washing | 1–2 mL/min | Sufficient to remove interferents while retaining analytes | High rate: Potential analyte lossLow rate: Incomplete cleanup |
| Elution | 0.5–1 mL/min | Ensures complete displacement of analytes from sorbent | High rate: Incomplete elutionLow rate: Excessive band broadening |
| Cartridge Format | 1–5 mL/min (depending on bed mass) | Balanced to avoid channeling in loose-packed beds [2] | High rate: Channeling and variable recovery |
| Disk Format | 10–50 mL/min [2] | Higher rates possible due to large cross-sectional area and short bed height | Less sensitive to flow rate variations |
Modern SPE technologies are addressing flow rate challenges through innovative sorbent formats. For instance, structured porous plastics with immobilized active media create an interconnected network of evenly distributed pores that enhance flow-through characteristics and provide more reproducible extractions with reduced variability (< 4% RSD) [67].
Drying: Managing Residual Water for Effective Elution
The Critical Role of Drying in SPE
The drying step, implemented after washing and before elution, serves the crucial function of removing residual water from the sorbent bed [66]. While seemingly straightforward, this step requires precise control as both insufficient and excessive drying can compromise method reproducibility. The primary scientific objective is to eliminate water that would otherwise:
- Dilute the elution solvent, reducing its strength
- Create immiscible barriers that prevent contact between elution solvent and analytes
- Cause freezing during subsequent concentration steps if using cryogenic methods
Comprehensive Drying Methodology
Implement the following drying protocol to ensure reproducible results:
Application of Positive Pressure or Vacuum: Pass air or inert gas (e.g., nitrogen) through the sorbent bed for 5-10 minutes under positive pressure, or apply a vacuum for 1-5 minutes [66]. The optimal duration depends on the sorbent mass and porosity.
Moisture Monitoring: For critical applications, incorporate a visual moisture indicator (such as anhydrous copper sulfate) between the sorbent bed and vacuum source to quantitatively assess the completeness of water removal.
Drying Time Optimization: Determine the optimal drying time experimentally by measuring analyte recovery as a function of drying duration. Plot recovery versus drying time to identify the plateau region where maximum recovery is achieved without degradation.
Problem-Shooting: If dealing with difficult-to-remove water, consider a "drying rinse" with a small volume (100-200 µL) of ethanol or isopropanol after the wash step. These solvents form azeotropes with water and facilitate its removal under milder conditions.
The consequences of improper drying are significant. Incomplete drying leads to diluted eluents and reduced elution strength, while excessive drying of certain sorbents can cause irreversible adsorption of target analytes or degradation of oxygen-sensitive compounds.
Integrated Experimental Protocol for Reproducibility Assessment
To systematically evaluate the impact of conditioning, flow rates, and drying on SPE reproducibility, implement the following experimental protocol:
Materials and Equipment
Table 3: Essential Research Reagent Solutions for Reproducible SPE
| Reagent/Equipment | Function/Application | Technical Specifications |
|---|---|---|
| C18 SPE Cartridges | Reversed-phase extraction for non-polar analytes | 100 mg/1 mL or 500 mg/3 mL bed mass [2] |
| Methanol (HPLC Grade) | Conditioning solvent for reversed-phase SPE | Low UV absorbance, high purity |
| Type 1 Water | Aqueous component for conditioning and washing | 18.2 MΩ·cm resistivity |
| Ammonium Acetate Buffer | pH control for ionizable analytes | 10-100 mM, pH adjusted with acetic acid/ammonia |
| Nitrogen Evaporator | Post-elution sample concentration | Temperature control (30-40°C) [66] |
| Vacuum Manifold | Flow rate control for multiple samples | Adjustable vacuum source with pressure gauge |
| Internal Standard Solution | Monitoring extraction efficiency | Structurally similar analog or stable-isotope labeled compound |
Experimental Procedure
System Suitability Test: Prepare a standard solution containing target analytes at known concentrations (typically 1-10 µg/mL in a compatible solvent).
Conditioning Variability Study:
- Group 1: Condition with methanol followed by water (standard protocol)
- Group 2: Condition with methanol only (incomplete conditioning)
- Group 3: Condition with methanol, then allow sorbent to dry before sample loading
Flow Rate Optimization:
- Load samples at 0.5, 1.0, 2.0, and 5.0 mL/min using a calibrated vacuum manifold or positive pressure system
- Collect flow-through fractions and analyze for breakthrough
Drying Time Profiling:
- Apply drying times of 0, 1, 2, 5, and 10 minutes after the wash step
- Elute with appropriate solvent and quantify recovery
Analysis and Calculation:
- Analyze all fractions (flow-through, wash, eluate) by HPLC-UV or LC-MS/MS
- Calculate recovery for each condition: (Amount in eluate / Total amount) × 100
- Determine reproducibility across replicates (n=5) using %RSD
Advanced Applications in Early Development Research
The principles of reproducible SPE find particular importance in early drug development, where standardized specifications for drug substances and products are still evolving [68]. In this context, robust SPE methods ensure that impurity profiles and degradation products are accurately monitored, supporting the establishment of appropriate acceptance criteria for early-phase materials [68]. The growing field of miniaturized SPE techniques—including solid-phase microextraction (SPME), stir-bar sorptive extraction (SBSE), and microextraction by packed sorbent (MEPS)—further emphasizes the critical need for precise control of conditioning, flow rates, and drying, as these systems typically handle smaller sample volumes where minor variations have magnified effects [65].
Conditioning, flow rates, and drying represent interconnected technical parameters that collectively determine the reproducibility of solid phase extraction. By understanding the scientific principles behind each step and implementing the detailed protocols outlined in this guide, researchers can significantly improve the reliability of their SPE methods. In early development research, where methods are established and refined, this reproducibility is not merely convenient but essential for generating valid, trustworthy data that advances scientific understanding. As SPE technologies continue to evolve toward miniaturization and automation, the fundamental importance of controlling these parameters remains constant, forming the foundation of reproducible sample preparation across diverse scientific disciplines.
In the early development of solid phase extraction (SPE) techniques, achieving selective cleanup presented a fundamental challenge for analytical chemists. Co-eluting interferences—unwanted matrix components that elute at the same time as target analytes—can severely compromise analytical accuracy, sensitivity, and reproducibility. These interfering substances, which range from phospholipids in biological samples to fatty acids in food matrices, compete with analytes during ionization in mass spectrometry, causing signal suppression or enhancement known as matrix effects [69] [70]. The persistence of these effects even after extensive sample preparation underscores the critical need for strategic cleanup approaches.
The evolution of SPE from its initial applications in the 1940s through its commercialization with pre-packed cartridges in 1977 represented significant milestones in sample preparation technology [2]. Early SPE methodologies primarily relied on simple reversed-phase or normal-phase mechanisms, but as analytical challenges grew more complex with the analysis of trace-level compounds in complex matrices, researchers recognized the limitations of these conventional approaches. This recognition drove the development of increasingly selective sorbents and multimodal extraction strategies capable of addressing the stubborn problem of co-elution [71].
Within the context of liquid chromatography-mass spectrometry (LC-MS), co-eluting interferences pose particularly detrimental effects. These compounds alter the ionization efficiency of target analytes in the ion source, leading to quantification inaccuracies that can jeopardize method validation parameters including accuracy, precision, and linearity [69]. The challenge is especially pronounced in complex matrices such as biological fluids, environmental samples, and food products, where thousands of potential interferents coexist with analytes of interest. Overcoming these challenges requires a systematic approach to method development that combines modern sorbent chemistry with rigorous evaluation techniques.
Understanding Matrix Effects and Co-Eluting Interferences
Origins and Impact of Matrix Effects
Matrix effects represent a significant challenge in modern bioanalysis, particularly in LC-MS applications. These effects occur when co-eluting compounds from the sample matrix alter the ionization efficiency of target analytes, leading to either ion suppression or less frequently, ion enhancement [69]. The mechanisms differ between ionization techniques. In electrospray ionization (ESI), which occurs in the liquid phase, interference species can affect droplet formation or charge transfer processes. In atmospheric pressure chemical ionization (APCI), where ionization occurs in the gas phase, different mechanisms prevail, often making APCI less prone to certain matrix effects [69].
The sources of co-eluting interferences vary significantly across different sample matrices:
- In biological samples like plasma and oral fluids, proteins, amino acids, and phospholipids represent major sources of interference [69]
- In environmental and food samples, lipids, humic acids, and pigments commonly co-extract with target analytes [72] [73]
- Residual impurities from solvents, sorbents, or sample containers can also contribute to the interference profile [74]
The extent of matrix effects is notoriously variable and unpredictable, depending on specific interactions between analytes and interfering compounds. Notably, the same analyte can exhibit different MS responses in different matrices, while the same matrix can affect different analytes in divergent ways [69]. This variability poses significant challenges during method validation, negatively affecting crucial parameters including reproducibility, linearity, selectivity, accuracy, and sensitivity [69].
Evaluating Matrix Effects: Diagnostic Approaches
Before implementing cleanup strategies, researchers must first assess the presence and extent of matrix effects. Three primary evaluation techniques have emerged, each providing complementary information (Table 1).
Table 1: Methods for Evaluating Matrix Effects in LC-MS
| Method Name | Description | Type of Assessment | Key Limitations |
|---|---|---|---|
| Post-Column Infusion | Continuous infusion of analyte during chromatography of blank matrix extract | Qualitative identification of suppression/enhancement regions | Does not provide quantitative data; labor-intensive for multi-analyte methods [69] |
| Post-Extraction Spike | Comparison of analyte response in standard solution versus blank matrix spiked post-extraction | Quantitative assessment at specific concentration | Requires blank matrix; single concentration evaluation [69] |
| Slope Ratio Analysis | Comparison of calibration curves in solvent versus matrix across concentration range | Semi-quantitative screening across concentration range | Only semi-quantitative results [69] |
The post-column infusion method, first described by Bonfiglio et al., provides a qualitative assessment of matrix effects across the entire chromatographic run [69]. This approach involves injecting a blank matrix extract while continuously infusing the analyte of interest post-column. Regions of ion suppression or enhancement appear as decreases or increases in the baseline signal, respectively, allowing identification of problematic retention time windows. This method is particularly valuable during method development as it helps optimize chromatographic conditions to separate analytes from interference-rich regions.
For quantitative assessment, the post-extraction spike method compares the analytical response of an analyte in pure solution to its response when spiked into a blank matrix extract after extraction [69]. The ratio of these responses quantifies the matrix effect at a specific concentration. Building on this approach, slope ratio analysis extends the evaluation across a range of concentrations by comparing the slopes of matrix-matched and solvent-based calibration curves [69]. This provides a more comprehensive understanding of concentration-dependent matrix effects.
Strategic Approaches to Selective Cleanup
Sorbent Selection and Mechanism Optimization
The foundation of effective cleanup lies in selecting appropriate sorbent chemistries and optimizing their interaction mechanisms. SPE sorbents function through two primary mechanisms: polarity-based interactions and ion-exchange mechanisms [75].
For polarity-based separations, the choice between normal-phase and reversed-phase modes is critical. Normal-phase SPE employs a polar sorbent (such as silica, alumina, or Florisil) with a non-polar mobile phase to retain polar constituents while eluting non-polar components [75]. Conversely, reversed-phase SPE uses a non-polar sorbent (C18 or C8) with a polar mobile phase to retain non-polar analytes while eluting polar matrix components [75]. The "like-dissolves-like" principle guides these interactions—compounds with similar polarity to the stationary phase exhibit stronger retention.
When analytes exist in a charged state or can be converted to one by adjusting pH, ion-exchange mechanisms offer powerful selectivity. Ion-exchange SPE relies on electrostatic attraction between charged analytes and oppositely charged sorbent surfaces [75]. The strategic pairing of "strong" and "weak" species is crucial: weak ion-exchange sorbents are recommended for strong ionic species (always charged), while strong ion-exchange sorbents pair best with weak ionic species (pH-dependent charge) [75]. This matching ensures sufficient retention without making elution unnecessarily difficult.
Table 2: Comparison of SPE Sorbent Performance for Different Applications
| Sorbent Type | Mechanism | Optimal Applications | Cleanup Efficiency | Recovery Performance |
|---|---|---|---|---|
| Florisil | Unbounded normal phase (polar) | Organochlorine pesticides, PCBs in food samples [73] | High removal of fatty acid interferences [73] | >70% for most compounds [73] |
| Alumina | Unbounded normal phase (polar) | POPs, PAHs in fatty matrices [72] [73] | Effective for low-fat food samples [73] | >78% recovery in herring [73] |
| Aminopropyl (NH2) | Bonded normal phase | Multi-residue analysis in complex matrices [73] | High efficiency for fatty acid removal [73] | Comparable to Florisil and Alumina [73] |
| EMR-Lipid | Novel reversed-phase | Lipid removal from fatty fish samples [72] | Superior lipid removal for POPs and PAHs [72] | 59-120% for POPs and PAHs [72] |
| C18 | Reversed-phase | General purpose reversed-phase applications | Limited interference removal [73] | Variable depending on matrix |
| Z-Sep | Mixed-mode | Fatty matrices | Moderate lipid removal [72] | Comparable to EMR-Lipid for some analytes [72] |
Advanced Sorbent Chemistries for Challenging Separations
For particularly challenging separation problems, advanced sorbent chemistries offer enhanced selectivity:
Mixed-mode sorbents incorporate multiple retention mechanisms, typically combining reversed-phase and ion-exchange properties, within a single cartridge [71]. These sorbents are particularly valuable for pharmaceutical applications where drugs of abuse and medications must be isolated from biological fluids [71]. The multimodal retention mechanism enables more selective cleanup than single-mechanism sorbents.
Molecularly imprinted polymers (MIPs) represent perhaps the most selective SPE sorbents available. These polymers are synthesized in the presence of a template molecule (the target analyte), creating specific recognition sites complementary in size, shape, and functional group orientation to the analyte [69] [71]. While offering exceptional selectivity, MIP technology is not yet commercially available for all applications and requires specialized synthesis for each analyte class [69].
Restricted access media (RAM) sorbents address the challenge of biological fluid analysis directly. These dual-mode sorbents feature an outer hydrophilic layer that excludes proteins and large molecules, combined with an inner hydrophobic surface that retains small analyte molecules [71]. This architecture allows for the direct injection of biological fluids like plasma or serum, with macromolecular matrix components eluting unretained while small molecule analytes are retained for subsequent analysis.
Experimental Protocols for Selective Cleanup
Sorbent Evaluation and Selection Protocol
Objective: Systematically evaluate different SPE sorbents for their ability to remove co-eluting interferences while maintaining adequate analyte recovery.
Materials:
- Selection of SPE sorbents (C18, Florisil, Alumina, NH2, EMR-Lipid, Z-Sep, mixed-mode)
- Blank matrix (appropriate to sample type)
- Analytical standards for target analytes
- Appropriate solvents for conditioning, washing, and elution
- LC-MS/MS system for analysis
Procedure:
- Prepare matrix-matched quality control samples by spiking blank matrix with target analytes at relevant concentrations.
- For each sorbent to be evaluated, condition with appropriate solvent (typically 3-5 mL methanol for reversed-phase, followed by 3-5 mL water or buffer).
- Load samples onto conditioned sorbents using controlled flow rates (typically 1-5 mL/min).
- Wash with optimized solvent volumes (typically 1-3 mL) to remove interferences while retaining analytes.
- Elute analytes with strong solvent (typically 1-5 mL) into collection tubes.
- Evaporate and reconstitute eluents if necessary for compatibility with analytical instrumentation.
- Analyze all samples alongside non-extracted standards to determine recovery and matrix effects.
Evaluation Criteria:
- Absolute recovery: Compare analyte responses in extracted samples to non-extracted standards at equivalent concentrations.
- Matrix effects: Use post-extraction spike method to calculate matrix factor (MF).
- Process efficiency: Combine recovery and matrix effects to assess overall method performance.
- Cleanup efficiency: Monitor specific matrix interferences (e.g., phospholipids in biological samples, lipids in food samples).
This systematic evaluation enables evidence-based sorbent selection, balancing cleanup efficiency with analyte recovery. Studies have demonstrated that sorbents like EMR-Lipid provide superior cleanup for fatty fish samples, with recoveries ranging from 59-120% for persistent organic pollutants and polycyclic aromatic hydrocarbons [72].
Troubleshooting Excessive Recoveries Protocol
Objective: Identify and resolve issues with recoveries exceeding 100%, which often indicate co-eluting interferences rather than true extraction efficiency [74].
Materials:
- SPE extracts showing elevated recoveries
- Alternative analytical column or separation conditions
- Different detection techniques (e.g., DAD alongside MS)
- Blank matrix samples
Diagnostic Procedure:
- Confirm co-eluting interferences:
- Analyze extracts using orthogonal separation techniques
- Employ different detection methods (e.g., diode array detection) to reveal co-eluting peaks
- Compare blank matrix extracts to identify interference sources
Modify wash steps:
- Increase selectivity of wash solvents to remove interferences while retaining analytes
- Implement sequential washing with solvents of increasing strength
- For ionizable compounds, adjust pH to convert analytes to non-ionic forms during washing
Evaluate sorbent selectivity:
- Test alternative sorbents with different retention mechanisms
- Consider ion-exchange sorbents for ionizable compounds
- Implement mixed-mode sorbents for combined retention mechanisms
Assess internal standard performance:
- Verify recovery and stability of internal standards
- Consider switching to structural analog or stable isotope-labeled internal standards
- Use external standardization if internal standard performance is inconsistent
Validate calculations:
- Re-integrate all chromatographic peaks with careful baseline placement
- Verify standard curve linearity and calibration accuracy
- Confirm dilution factors and calculation formulas
This systematic troubleshooting approach addresses the most common causes of excessive recoveries, with co-eluting interferences representing the predominant issue [74].
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Essential Research Reagents for Selective Cleanup
| Reagent/Sorbent | Function | Application Notes |
|---|---|---|
| EMR-Lipid Sorbent | Selective removal of lipid components from fatty matrices | Particularly effective for fish tissue (10% lipid content); superior to Z-Sep for POPs and PAHs [72] |
| Florisil | Unbounded normal-phase sorbent for polar interferences | Effective for organochlorine pesticides and PCBs in low-fat food; provides >70% recovery for most compounds [73] |
| Alumina | Unbounded normal-phase sorbent with adjustable activity | Effective clean-up for low-fat food samples; comparable performance to Florisil and NH2 [73] |
| C18 Bonded Silica | Reversed-phase sorbent for non-polar analytes | Limited interference removal capability; best for simple matrices [73] |
| Mixed-mode Sorbents | Combined reversed-phase and ion-exchange mechanisms | Ideal for pharmaceutical compounds in biological matrices; enhanced selectivity [71] |
| Stable Isotope-Labeled Internal Standards | Correction for matrix effects and recovery variations | Essential for accurate quantification; corrects for both extraction efficiency and ionization effects [69] [76] |
| Molecularly Imprinted Polymers | Highly selective sorbents with template-specific recognition | Exceptional selectivity but limited commercial availability; promising for future applications [69] [71] |
Integrated Cleanup Strategies and Workflow Optimization
Successful cleanup requires integrating multiple strategies tailored to specific analytical challenges. The following diagram illustrates a systematic approach to developing selective cleanup methods:
Systematic Cleanup Development Workflow
Beyond sorbent selection, chromatographic optimization represents a powerful tool for addressing co-elution challenges. Adjusting column chemistry, mobile phase composition, gradient profile, and temperature can achieve separation of analytes from interferences that persist after sample preparation [70]. Employing microflow LC-MS/MS setups has demonstrated up to sixfold sensitivity improvements by optimizing chromatographic flow rates and sample clean-up, thereby minimizing matrix interferences [70].
When interference elimination proves incomplete, calibration strategies can compensate for residual matrix effects. When blank matrices are available, matrix-matched calibration standards provide effective compensation [69]. For situations where blank matrices are unavailable, stable isotope-labeled internal standards represent the gold standard for correction, as they experience nearly identical matrix effects as their native counterparts while being distinguishable mass spectrometrically [69] [76].
Emerging Trends and Future Perspectives
The field of selective cleanup continues to evolve with several promising trends emerging. Mathematical cleanup approaches using multivariate curve resolution with alternating least squares (MCR-ALS) have shown potential for eliminating interferences from extraction solvents, including ionic liquids used in dispersive liquid-liquid microextraction [77]. This chemometric approach enables the mathematical separation of analyte signals from co-eluting interferences by exploiting spectral and chromatographic profile differences [77].
Mixed-mode and multilayer SPE configurations offer enhanced selectivity for complex applications. The serial connection of different SPE cartridges or the use of single cartridges with multiple functional groups enables retention based on multiple mechanisms simultaneously [71]. For example, layered sorbent techniques with amino sorbent over C18 have successfully removed humic acids while retaining pesticides in environmental water samples [71].
Miniaturized extraction formats including pipette-tip SPE (PT-SPE) and solid-phase microextraction (SPME) are gaining traction for their ability to reduce solvent consumption while maintaining effective cleanup [2]. These approaches align with green chemistry principles while offering practical benefits for limited sample volumes.
As analytical challenges continue to evolve toward more complex matrices and lower detection limits, the development of selective cleanup strategies remains a dynamic research area. The integration of advanced sorbent chemistries with optimized chromatographic separations and mathematical correction approaches provides a powerful toolkit for overcoming the persistent challenge of co-eluting interferences in modern bioanalysis.
In the early development of solid phase extraction (SPE) techniques, the optimization of flow rates represents a critical challenge that sits at the intersection of analytical efficiency and data integrity. SPE serves as a fundamental sample preparation technique used to isolate and concentrate analytes from complex matrices, thereby improving the sensitivity and accuracy of downstream analytical methods such as liquid chromatography and mass spectrometry [6]. The flow rate at which samples and solvents pass through the SPE sorbent bed directly influences the kinetic processes of adsorption and desorption, ultimately determining the success of the extraction procedure.
This technical guide examines the fundamental principles governing flow rate optimization in SPE methodologies, with particular emphasis on the delicate balance between processing efficiency and the prevention of analyte breakthrough. Breakthrough occurs when the flow rate exceeds the kinetic capacity of the sorbent to retain target compounds, resulting in premature elution and substantial analyte loss [78] [79]. For researchers and drug development professionals, understanding these dynamics is essential for developing robust, reproducible SPE methods that maintain analytical integrity while maximizing throughput in pharmaceutical applications.
Fundamental Principles of Flow Rate in SPE
The Chromatographic Foundation of SPE
Solid phase extraction operates on chromatographic principles where a liquid mobile phase (samples and solvents) passes through a solid stationary phase (sorbent), facilitating interactions that selectively retain target analytes while removing interfering matrix components [79]. The flow rate directly controls the residence time—the duration that analytes remain in contact with the sorbent material—which in turn governs the efficiency of these interactions.
In SPE systems, two primary interaction mechanisms dictate flow rate considerations: polarity-based interactions (including reversed-phase and normal-phase mechanisms) and ion-exchange interactions [79]. Each mechanism exhibits distinct kinetic properties and consequently different flow rate dependencies:
- Polarity-based interactions follow the "like dissolves like" principle, where nonpolar analytes interact with nonpolar sorbents (reversed-phase) or polar analytes with polar sorbents (normal-phase) [79].
- Ion-exchange interactions operate on the "opposites attract" principle, where charged analytes interact with oppositely charged sorbent surfaces [79].
The flow rate must be optimized to ensure sufficient contact time for these interactions to occur effectively, while simultaneously maintaining practical processing times for laboratory workflows.
The Breakthrough Phenomenon
Analyte breakthrough represents the most significant risk associated with excessive flow rates in SPE. This phenomenon occurs when the volumetric flow rate exceeds the sorbent's kinetic capacity to retain target compounds, resulting in incomplete retention and consequent analyte loss during the loading phase [78] [79].
The relationship between flow rate and breakthrough is inversely proportional—as flow rates increase, the likelihood of breakthrough escalates substantially. This relationship is particularly pronounced for ion exchange sorbents, where interaction kinetics are generally slower than with reversed-phase materials [78]. As noted in SPE optimization guidelines, "If the flow rate is too fast, the force from the vacuum or positive pressure source could be greater than the retention effect exerted by the sorbent (this is especially true for ion exchange sorbents)" [78].
The following diagram illustrates the sequential relationship between flow rate control and its consequences throughout the SPE workflow:
Experimental Protocols for Flow Rate Optimization
Breakthrough Curve Methodology
The most direct approach for evaluating flow rate efficacy involves constructing breakthrough curves through systematic experimentation. This methodology determines the maximum flow rate that can be applied before significant analyte loss occurs.
PROTOCOL: Breakthrough Curve Analysis
Sample Preparation: Prepare a standard solution containing target analytes at known concentrations in a matrix that mimics the actual sample. For biological matrices such as plasma or serum, dilute with an equal volume of appropriate buffer [6].
SPE Cartridge Conditioning: Condition the SPE cartridge with an organic solvent (typically methanol or acetonitrile), followed by an aqueous or buffer solution with similar pH to the sample. Ensure the sorbent does not dry completely before sample loading [78] [6].
Sample Loading at Varied Flow Rates: Divide the standard solution into equal aliquots and load each onto separately conditioned SPE cartridges using different, precisely controlled flow rates. Recommended testing range: 0.5-3 mL/min for standard cartridge formats [80]. Collect the effluent (the liquid passing through the cartridge) during loading.
Analyte Measurement: Analyze the effluent for target analyte content using appropriate analytical methods (e.g., HPLC-UV, LC-MS). This quantifies the percentage of analyte that breaks through at each flow rate.
Data Interpretation: Plot analyte recovery (%) against flow rate (mL/min). The optimal flow rate falls within the plateau region where recovery remains consistently high before declining due to breakthrough.
Calculation of Breakthrough Capacity:
Where Cinitial is the initial analyte concentration and Ceffluent is the concentration in the effluent.
Response Surface Methodology (RSM) with Box-Behnken Design
For comprehensive optimization of multiple interdependent parameters, Response Surface Methodology (RSM) provides a statistically rigorous approach. This method is particularly valuable when flow rate interactions with other variables (pH, bed height, concentration) must be considered simultaneously [80].
PROTOCOL: RSM Optimization
Factor Identification: Identify critical factors affecting SPE performance through preliminary screening. Key factors typically include flow rate, bed height, and inlet analyte concentration [80].
Experimental Design: Implement a Box-Behnken design with flow rate as one of the primary factors. This design efficiently explores multiple factor interactions with reduced experimental runs compared to full factorial designs.
Model Development: Conduct experiments according to the design matrix and fit response data (e.g., recovery percentage) to a quadratic model:
Where Y represents the response variable, β represents regression coefficients, and X represents coded factor levels.
Design Space Establishment: Use response surface plots to visualize the relationship between flow rate and other factors. Identify the operational design space where method performance meets predefined criteria (e.g., >90% recovery).
Method Verification: Validate optimized conditions by conducting confirmatory experiments and assessing method robustness.
Quantitative Data on Flow Rate Effects
Flow Rate Ranges by SPE Mechanism
Table 1: Recommended Flow Rate Ranges by Sorbent Mechanism
| Sorbent Mechanism | Optimal Flow Rate Range | Critical Considerations | Maximum Tolerance |
|---|---|---|---|
| Reversed-Phase | 1-3 mL/min [6] | Neutral pH enhances non-polar retention; adjust pH to neutralize analyte charge [78] | ≤5 mL/min for most applications |
| Ion Exchange | 0.5-2 mL/min [78] [80] | Slower kinetics require longer residence times; especially sensitive to flow rate [78] | ≤3 mL/min with strong ion pairing |
| Mixed-Mode | 1-2 mL/min [81] | Balance between hydrophobic and ion-exchange interactions | ≤4 mL/min for well-retained analytes |
| Molecularly Imprinted Polymers | 0.5-1.5 mL/min [82] | Highly specific interactions but sensitive to flow conditions | ≤2 mL/min to maintain specificity |
Experimental Data from SPE Optimization Studies
Table 2: Experimental Flow Rate Optimization Data from Published Studies
| Study Focus | Sorbent Type | Optimal Flow Rate | Recovery at Optimal Rate | Recovery at Suboptimal Rate | Key Findings |
|---|---|---|---|---|---|
| Triclosan Adsorption | Alumina Pillared Clays | 0.5 cm³/min [80] | >98% | ~80% at 3 cm³/min | Lower flow rates significantly improved adsorption capacity in fixed-bed columns |
| Basic Drug Extraction | Strong Cation Exchange | 1-2 mL/min [81] | 98% | ~60% with rapid loading | 70% methanol wash possible without analyte loss at controlled flow rates |
| Molecular Imprinted SPE | MIPs | ~1 mL/min [82] | >95% | Substantial decrease above 2 mL/min | Flow rate critical for achieving sorption equilibrium in specific binding |
| SPE-HPLC-UV/ELSD | C18 | 1 mL/min [83] | >95% for 9 compounds | NR | Identified as statistically significant factor (p<0.05) via Plackett-Burman design |
Advanced Optimization Strategies
Quality by Design (QbD) Approach
Implementing Quality by Design principles provides a systematic framework for flow rate optimization that aligns with regulatory expectations in pharmaceutical development. The QbD approach involves:
Risk Assessment: Identify flow rate as a critical method parameter through initial risk assessment tools such as Fishbone diagrams or Failure Mode Effects Analysis [83].
Design of Experiments (DoE): Employ statistical experimental designs (e.g., Plackett-Burman for screening, Box-Behnken for optimization) to characterize flow rate interactions with other method parameters [80] [83].
Design Space Establishment: Define the multidimensional combination of input variables (including flow rate) that provides assured quality. Operating within this design space provides method robustness despite minor operational variations [83].
Control Strategy: Implement appropriate controls for maintaining flow rates within established optimal ranges, particularly when transitioning methods across different equipment or platforms.
Mechanistic Workflow for Flow Rate Optimization
The following diagram illustrates a comprehensive, mechanistic approach to flow rate optimization, integrating both breakthrough testing and statistical optimization methods:
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Research Reagents and Materials for SPE Flow Rate Studies
| Item Category | Specific Examples | Function in Flow Rate Optimization |
|---|---|---|
| SPE Sorbents | Strata-X-C (strong cation exchange) [81], Bond Elut C18 [78], Molecularly Imprinted Polymers [82] | Varying interaction mechanisms and kinetics for evaluating flow rate dependencies |
| Solvent Systems | Methanol, Acetonitrile [78], Ammonium Hydroxide (2-5%) [81], Buffer Solutions (pH-specific) [6] | Control elution strength and maintain optimal pH conditions during flow rate studies |
| SPE Formats | 1mL, 3mL, 6mL cartridges [6], 96-well plates [6] [81] | Different geometries affecting flow dynamics and optimal flow rates |
| Analytical Standards | Atenolol, Toliprolol, Bunitrolol, Bupranolol [81], Triclosan [80] | Model compounds with varying physicochemical properties for breakthrough studies |
| Automation Equipment | Automated SPE systems [81], Andrew+ Pipetting Robot [84] | Precise flow rate control and reproducible method implementation |
The optimization of flow rates in solid phase extraction represents a fundamental methodological consideration that directly influences the success of subsequent analytical measurements. Through systematic approaches including breakthrough testing and statistical optimization, researchers can identify flow conditions that balance analytical efficiency with quantitative recovery. The implementation of Quality by Design principles further enhances method robustness, ensuring reliability across different instruments and operators. As SPE technologies continue to evolve with advancements in sorbent materials and automation capabilities, the fundamental importance of flow rate optimization remains constant—serving as a critical control parameter in the development of robust, reproducible analytical methods for pharmaceutical research and drug development.
Solid-phase extraction (SPE) has undergone a remarkable transformation since its early development, evolving from simple pigment-removal techniques using animal charcoal in the 1940s to today's sophisticated, selective sorbents [2]. The first experimental applications for organic trace analysis in water samples began in the 1950s, but the technique gained significant scientific recognition during the 1970s with the introduction of synthetic polymers like styrenedivinylbenzene resins [18] [2]. A pivotal moment occurred in October 1977 with the commercial introduction of pre-filled cartridges containing silica sorbents, which made the procedure more convenient and began a new phase of development focused on standardized methodologies [2]. The subsequent development of stable, covalently bound reverse-phase sorbents, particularly C18, opened applications in environmental, clinical, and pharmaceutical markets, establishing SPE as a fundamental sample preparation technique [2].
This guide examines the historical principles and modern practices for selecting sorbents and elution solvents, providing researchers with a practical framework for developing robust SPE methods within drug development and analytical research contexts.
Fundamental Principles of SPE
Solid-phase extraction is a sample preparation technique that purifies and concentrates analytes from a liquid sample by passing it through a solid sorbent material [6]. SPE operates on the same basic principles as liquid chromatography, utilizing mechanisms of polarity and ion exchange to separate analytes from matrix interferences [85]. The fundamental process involves distributing analytes between a liquid sample medium and a solid adsorbent to which the analytes have greater affinity, followed by elution with an appropriate solvent [2].
The typical SPE sequence consists of four essential steps, as illustrated in the workflow below and described in detail thereafter [71] [6].
Conditioning and Equilibration
Conditioning prepares the sorbent by solvating the bonded phase to ensure proper interaction with the sample. It typically involves passing a solvent with similar characteristics to the sample through the sorbent. A critical consideration is to prevent the sorbent from drying after conditioning, as this can significantly reduce retention efficiency [6].
Sample Application
The prepared sample is loaded onto the conditioned sorbent at a controlled flow rate, typically around 1 mL/minute, to maximize analyte retention. Higher flow rates can lead to inconsistent extractions and analyte breakthrough [6].
Washing
This step removes interfering compounds by applying a solvent strong enough to elute impurities but weak enough to leave the target analytes bound to the sorbent. The wash solvent is selected according to the phase mechanism and analyte properties [6].
Elution
The final step disrupts the analyte-sorbent interaction using a strong solvent that selectively recovers the analytes in a small volume. Using two small aliquots rather than one large volume provides better recovery efficiency [6].
Sorbent Selection: Mechanisms and Materials
Choosing the appropriate sorbent is the most critical decision in SPE method development. Sorbents are selected based on the chemical properties of the target analytes and the sample matrix, with primary separation mechanisms falling into three categories: reversed-phase, normal-phase, and ion-exchange [85] [71].
Reversed-Phase Sorbents
Reversed-phase sorbents retain analytes based on hydrophobic interactions and are characterized by a nonpolar stationary phase paired with a polar mobile phase (typically water or buffers) [85]. The "like-dissolves-like" principle applies, where nonpolar analytes have stronger interactions with the nonpolar stationary phase [85].
Table 1: Common Reversed-Phase Sorbents
| Sorbent Type | Characteristics | Applications | Historical Context |
|---|---|---|---|
| C18 (Octadecyl) | High hydrophobicity, strong retention of non-polar compounds | Pharmaceuticals, environmental pollutants | One of the earliest bonded phases; became standard for reversed-phase applications [2] |
| C8 (Octyl) | Moderate hydrophobicity, slightly less retentive than C18 | Mid-polarity analytes, larger molecules | Developed as an alternative to C18 for less hydrophobic compounds |
| Hydrophilic-Lipophilic Balance (HLB) | Balanced wettability, retains both polar and non-polar compounds | Acidic, basic, and neutral compounds simultaneously | Advanced copolymer development (e.g., Oasis HLB) simplified methods for diverse analytes [86] [87] |
| Polymer-based (PS-DVB) | High capacity, stable across pH range 1-14 | Broad-spectrum extraction, harsh pH conditions | Styrenedivinylbenzene polymers were among first synthetic sorbents [2] |
Normal-Phase Sorbents
Normal-phase sorbents employ a polar stationary phase with a nonpolar mobile phase to retain polar compounds from nonpolar matrices [85]. This approach was historically the "normal" mode before reversed-phase became more prevalent.
Table 2: Normal-Phase Sorbents
| Sorbent Type | Characteristics | Applications |
|---|---|---|
| Silica (unbonded) | Polar surface with silanol groups, hydrophilic | Alcohols, carbohydrates, pharmaceuticals from organic solvents |
| Diol | Mildly polar, hydrogen bonding capabilities | Polar compounds requiring milder retention than silica |
| Amino (NH₂) | Basic character, weak anion exchanger | Carbohydrates, organic acids, anionic compounds |
| Cyano (CN) | Moderate polarity, dual functionality | Versatile for both normal-phase and reversed-phase applications |
Ion-Exchange Sorbents
Ion-exchange sorbents separate compounds based on ionic interactions between charged analytes and oppositely charged sorbents, operating on the principle that "opposites attract" [85]. The effectiveness depends on the sample pH relative to the analyte's pKa to ensure proper ionization [85].
Table 3: Ion-Exchange Sorbents
| Sorbent Type | Characteristics | Analyte Properties | Applications |
|---|---|---|---|
| Strong Cation Exchange (SCX) | Contains sulfonic acid groups (SO₃⁻), permanently charged | Positively charged analytes (cations), pKa > 2 units above sample pH | Basic drugs, peptides, catecholamines |
| Weak Cation Exchange (WCX) | Contains carboxylic acid groups (COO⁻), pH-dependent charge | Strong cations (permanently charged) | Better for strong cations due to easier elution |
| Strong Anion Exchange (SAX) | Contains quaternary ammonium groups (NR₃⁺), permanently charged | Negatively charged analytes (anions), pKa < 2 units below sample pH | Organic acids, nucleotides, acidic drugs |
| Weak Anion Exchange (WAX) | Contains primary/secondary/tertiary amine groups, pH-dependent charge | Strong anions (permanently charged) | Better for strong anions due to easier elution |
Mixed-Mode Sorbents
Mixed-mode sorbents incorporate multiple retention mechanisms (typically reversed-phase and ion-exchange) in a single cartridge, providing enhanced selectivity for complex samples [71]. These can be configured as either a single sorbent with multiple functional groups or as layered sorbents within one cartridge [71]. For example, a C8 and phenyl sulfonic acid combination can simultaneously retain drugs via hydrophobic interactions while capturing cationic species through ion exchange [71].
Elution Solvent Selection
Elution solvent selection depends on effectively disrupting the specific interactions retaining analytes on the sorbent. The key principles include:
- Solvent strength: Must be sufficient to overcome analyte-sorbent interactions
- Selectivity: Should elute target analytes while leaving interferents retained
- Volume: Smaller volumes produce more concentrated extracts [6]
Table 4: Elution Solvents by Sorbent Type
| Sorbent Mechanism | Analyte Characteristics | Recommended Elution Solvents | Historical Optimization Evidence |
|---|---|---|---|
| Reversed-Phase | Non-polar to moderately polar | Methanol, acetonitrile, tetrahydrofuran, ethyl acetate, dichloromethane | Methanol provided 67-94% recovery for efavirenz/levonorgestrel in wastewater [86] |
| Normal-Phase | Polar | Polar solvents with increasing strength: hexane → ethyl acetate → acetone → methanol → water | Method development focused on polarity matching |
| Cation Exchange | Basic/positively charged | Methanol with 2-5% acetic acid, ammonia/methanol, ammonium acetate/methanol, hydrochloric acid | pH adjustment to neutralize analyte charge (2 pH units below pKa for weak cations) [85] |
| Anion Exchange | Acidic/negatively charged | Methanol with 2-5% formic acid, acetic acid/methanol, ammonium hydroxide | pH adjustment to neutralize analyte charge (2 pH units above pKa for weak acids) [85] |
| Mixed-Mode | Complex properties | Multi-step elution: organic solvent for hydrophobic disruption followed by pH-adjusted solvent for ionic disruption | Sequential methanol then methanol with ammonia for basic drugs [71] |
Advanced Sorbent Chemistries
Modern SPE has evolved beyond traditional sorbents to include highly selective materials for specialized applications:
Restricted Access Media (RAM)
RAM sorbents combine size exclusion with chemical interaction to directly inject biological fluids like plasma and serum [71]. These dual-mode phases feature an outer hydrophilic layer that excludes proteins and an inner hydrophobic surface that retains small molecules, enabling automated analysis of biological samples without protein precipitation steps [71].
Molecularly Imprinted Polymers (MIPs)
MIPs are synthetic polymers with predefined selectivity for specific target molecules, created by polymerizing functional monomers around a template molecule [71]. After template removal, complementary binding sites remain that exhibit high affinity for the target analyte, approaching antibody-like specificity for challenging applications requiring exceptional selectivity [71].
Solvent-Assisted Dispersive SPE (SA-DSPE)
A modern approach where sorbent material is dispersed throughout the sample as a fine suspension using a solvent, significantly increasing the contact surface area between the sorbent and analytes [88]. This technique has been applied successfully for determining anionic surfactants in water samples, demonstrating the continued innovation in SPE formats [88].
Practical Application and Experimental Optimization
Case Study: Simultaneous Extraction of Pharmaceuticals from Wastewater
Research on extracting efavirenz (antiretroviral) and levonorgestrel (contraceptive) from wastewater demonstrates systematic SPE optimization [86]:
- Sorbent Selection: Hydrophilic-lipophilic balance (HLB) cartridge (60 mg/3 mL) was selected for its ability to retain both polar and non-polar compounds simultaneously [86]
- Parameter Optimization: One-factor-at-a-time approach varying pH, solvent concentration, and elution volume
- Optimal Conditions: pH 2, 100% methanol as elution solvent, 4 mL elution volume
- Performance Outcomes: Recoveries of 67-83% for efavirenz and 70-95% for levonorgestrel with correlation coefficients >0.98 [86]
This case exemplifies the "like-dissolves-like" principle, where methanol effectively disrupted the hydrophobic interactions between the pharmaceutical compounds and the HLB sorbent [86] [85].
The Scientist's Toolkit: Essential Research Reagents
Table 5: Essential SPE Laboratory Materials
| Reagent/Equipment | Function/Application | Technical Specifications |
|---|---|---|
| HLB Cartridges | Broad-spectrum extraction of acidic, basic, and neutral compounds | 60 mg/3 mL; compatible with pH 1-13 [86] |
| C18 Sorbents | Reversed-phase extraction of non-polar compounds | Various sizes (1mL to 150mL cartridges); end-capped to reduce silanol effects [85] [6] |
| Mixed-Mode MCX | Simultaneous hydrophobic and cation-exchange retention | 1-500 mg sorbent weights; ideal for basic compounds [87] |
| Methanol (HPLC grade) | Versatile elution solvent for reversed-phase and ion-exchange | 100% for strong elution; variously diluted for washing [86] |
| Acetonitrile | Alternative strong elution solvent | Different selectivity than methanol; useful for method development |
| Ammonium Hydroxide | pH adjustment for elution of basic compounds | Typically 2-5% in methanol for ion-exchange elution [85] |
| Formic Acid | pH adjustment for elution of acidic compounds | Typically 2-5% in methanol for ion-exchange elution [85] |
| Vacuum Manifold | Processing multiple samples simultaneously | Compatible with various cartridge sizes (1-150mL) [6] |
Method Development and Troubleshooting
The following decision framework illustrates a systematic approach to SPE method development:
When evaluating SPE protocol success, measure three key parameters [87]:
- Percentage Recovery: The percentage of analyte recovered from the sample, with values of 70-120% typically considered acceptable for validated methods
- Matrix Effect: The impact of co-extracted substances on analyte detection, particularly important in LC-MS applications
- Mass Balance: Accounting for total analyte throughout the extraction process to identify irreversible binding or degradation
The evolution of solid-phase extraction from its early beginnings with activated carbon filters to today's advanced selective sorbents demonstrates how fundamental principles of polarity and ion exchange have been refined into powerful separation tools [18] [2]. Successful SPE method development relies on understanding these historical principles while applying systematic optimization of sorbent-eluent combinations specific to the analytical challenge. As SPE continues to evolve with new materials and formats, the core principles of selective retention and efficient elution remain the foundation for effective sample preparation in drug development and environmental analysis.
In the early development of solid phase extraction (SPE) techniques, sample pre-treatment and pre-filtration are not merely preliminary steps; they are foundational to the success of downstream analysis. Complex matrices—from environmental waters and plant material to biological fluids—are laden with interferents that can compromise analytical accuracy. This guide explores the critical role of managing these matrices, providing researchers with validated protocols and strategic insights to enhance recovery, precision, and sensitivity in their SPE workflows.
Solid Phase Extraction is a pivotal sample preparation technique designed to isolate and concentrate analytes from complex matrices, thereby enhancing the sensitivity and selectivity of subsequent analytical methods like liquid chromatography-mass spectrometry (LC-MS) [89]. Its efficiency, however, is heavily dependent on the initial condition of the sample. Complex matrices introduce three primary challenges:
- Matrix Effects: Co-extracted compounds from the sample matrix can suppress or enhance the ionization of target analytes in mass spectrometers, leading to inaccurate quantification [89] [90].
- Column Fouling: Particulate matter and macromolecules (e.g., proteins, phospholipids) can clog SPE sorbent pores and HPLC columns, increasing backpressure and causing analytical downtime [91].
- Reduced Recovery and Selectivity: Interfering compounds may compete with analytes for binding sites on the SPE sorbent, leading to poor recovery and low purity of the final extract [89] [92].
Sample pre-treatment and pre-filtration serve as the first line of defense against these challenges. By removing particulates and mitigating matrix effects early in the workflow, these steps ensure the robustness of the SPE protocol and the reliability of the generated data.
Core Principles and Strategic Workflows
A well-designed sample preparation strategy is built on understanding the interactions between the sample matrix, the analytes of interest, and the SPE sorbent. The following workflow provides a logical sequence for managing complex matrices, from initial assessment to final extraction.
This workflow underscores that pre-treatment is not one-size-fits-all. The chosen method must align with the sample's physical state and chemical composition. For instance, centrifugal pre-filtration is highly effective for clarifying biological samples like plasma [90], while pH adjustment is a critical pre-treatment for ionizable analytes in water samples to ensure optimal retention during SPE [93].
Experimental Protocols and Validation Data
Translating strategic principles into actionable, validated methods is key to success. The following protocols, drawn from recent research, illustrate the application of these principles across different sample types.
Protocol 1: Analysis of Organic UV-Filters in Surface Water
This protocol demonstrates a robust SPE method for trace-level environmental contaminants, highlighting the importance of sorbent selection and pH control [93].
- Sample Matrix: Surface freshwater.
- Target Analytes: Avobenzone (AVO), iscotrizinol (DBT), octinoxate (EHMC), ethylhexyl triazone (EHT), octocrylene (OC).
- Pre-treatment and Pre-filtration:
- Sample Collection: Collect water samples in clean, amber glass containers.
- Pre-filtration: Filter samples through 0.45 µm glass fiber filters to remove suspended particulate matter. This step prevents clogging of the subsequent SPE cartridges.
- pH Adjustment: Adjust the pH of the filtered water sample to 7.0 using a dilute acid or base. This is a critical step as the mobile phase pH was identified as one of the most influential factors on chromatographic separation.
- SPE Procedure:
- Sorbent: Use Phenomenex Strata SI-1 Silica (500 mg/3 mL) cartridges.
- Conditioning: Condition the cartridge with 6 mL of methanol followed by 6 mL of ultrapure water.
- Loading: Load the pre-treated water sample (e.g., 100-500 mL) onto the cartridge at a controlled flow rate of 5-10 mL/min.
- Washing: Wash the cartridge with 3 mL of a 5% methanol solution in water to remove weakly retained interferents.
- Elution: Elute the target UV-filters with 2 x 4 mL of methanol. Combine the eluents.
- Concentration: Gently evaporate the eluent to dryness under a stream of nitrogen and reconstitute the residue in 1 mL of methanol for UHPLC-MS/MS analysis.
- Validation Data: The method was rigorously validated, yielding the following performance metrics [93]:
Table 1: Validation Metrics for UV-Filter SPE Method in Water
| Analyte | Accuracy (%) | Recovery (%) | LOD (ng/L) | LOQ (ng/L) |
|---|---|---|---|---|
| Avobenzone (AVO) | 85.9 - 114.2 | 65.1 - 70.3 | 1.05 | 3.18 |
| Is cotrizinol (DBT) | 92.3 - 110.1 | 45.2 - 58.7 | 0.76 | 2.29 |
| Octinoxate (EHMC) | 88.5 - 123.4 | 68.9 - 73.4 | 2.15 | 6.52 |
| Ethylhexyl Triazone (EHT) | 90.4 - 118.6 | 48.5 - 55.1 | 3.16 | 9.58 |
| Octocrylene (OC) | 94.7 - 115.8 | 62.4 - 69.8 | 1.89 | 5.72 |
Protocol 2: Determination of Pharmaceuticals in Vegetables Using MISPE
This protocol showcases a selective extraction technique for complex solid matrices, where pre-treatment involves a comprehensive extraction and centrifugation process [92].
- Sample Matrix: Lettuce, carrot, cucumber, green pepper.
- Target Analytes: Fenoprofen, naproxen, diclofenac, ibuprofen, gemfibrozil.
- Pre-treatment and Pre-filtration:
- Homogenization: Chop and homogenize the vegetable samples using a blender.
- Solid-Liquid Extraction: Weigh 2 g of homogenized sample into a centrifuge tube. Add 10 mL of acetonitrile and vortex vigorously for 2 minutes.
- Centrifugation: Centrifuge the mixture at 4000 rpm for 10 minutes to separate the solid plant debris from the liquid extract.
- Pre-filtration: The supernatant is carefully decanted and may be passed through a filter paper to remove any remaining fine particles.
- SPE Procedure (MISPE):
- Sorbent: Use a molecularly imprinted polymer (MIP) SPE cartridge tailored for the specific pharmaceuticals (e.g., Strata-X cartridges).
- Conditioning: Condition with 5 mL of methanol and 5 mL of ultrapure water.
- Loading: Load the clarified vegetable extract onto the cartridge.
- Washing: Wash with 2-3 mL of a water-methanol (90:10, v/v) solution to remove matrix interferents.
- Elution: Elute the analytes with 2 x 2.5 mL of methanol. The MIP sorbent provides high selectivity, reducing co-extraction of plant pigments and other interferences.
- Concentration: Evaporate the eluent to dryness and reconstitute in the HPLC mobile phase for analysis.
- Validation Data: The optimized MISPE method demonstrated excellent performance, enabling a comprehensive health risk assessment [92]:
Table 2: Validation Metrics for MISPE of Pharmaceuticals in Vegetables
| Analyte | Average Recovery (%) | RSD (%) | Maximum Concentration Found (mg kg⁻¹) |
|---|---|---|---|
| Fenoprofen | 95 - 103 | 0.9 - 4.1 | 6.44 (Pepper) |
| Naproxen | 88 - 96 | 2.5 - 5.8 | 2.15 (Lettuce) |
| Diclofenac | 45 - 65 | 3.2 - 8.5 | 1.98 (Cucumber) |
| Ibuprofen | 75 - 92 | 4.1 - 9.7 | 3.21 (Carrot) |
| Gemfibrozil | 80 - 95 | 2.8 - 13.0 | 1.76 (Lettuce) |
The Scientist's Toolkit: Essential Research Reagent Solutions
Selecting the appropriate materials is critical for developing a robust SPE method. The following table details key reagents and their functions in the sample preparation workflow.
Table 3: Essential Materials for SPE in Complex Matrices
| Item | Function & Importance | Example Applications |
|---|---|---|
| Silica-based SPE Sorbents (e.g., Phenomenex SI-1): | General-purpose sorbent for polar and mid-polar analytes; the backbone of many classic SPE methods. | Isolation of organic UV-filters from water [93]. |
| Molecularly Imprinted Polymers (MIPs): | Provides high selectivity by containing synthetic cavities complementary to the target analyte, reducing matrix effects. | Selective extraction of pharmaceuticals from vegetable extracts [92]. |
| C18 Sorbents: | Reversed-phase sorbent for non-polar to moderately polar compounds; one of the most widely used sorbents. | Determination of estrogens in milk [94]; flavonoids in plants [95]. |
| 0.45 µm Syringe Filters (Nylon, PTFE): | Pre-filtration to remove particulate matter that could clog SPE cartridges or HPLC systems. | Standard pre-treatment for water samples and clarified biological/plant extracts [93] [91]. |
| Centrifuges: | Separates solids from liquids in complex matrices like biological fluids or plant homogenates. | Pre-treatment of rat plasma, urine, and feces [90]. |
| Ultrasonic Bath: | Facilitates the dissolution and extraction of analytes from solid samples or drug products. | Solubilization of active ingredients from tablet formulations [91]. |
In the context of early SPE research and modern analytical science, the adage "garbage in, garbage out" holds profound significance. As demonstrated by the cited protocols, meticulous attention to sample pre-treatment and pre-filtration is not an optional refinement but a non-negotiable component of method development. These steps directly enable the high sensitivity, accuracy, and reproducibility required in regulatory analysis, drug development, and environmental monitoring. By adopting a strategic, matrix-aware approach to sample preparation, researchers can unlock the full potential of solid phase extraction, transforming complex, noisy samples into clean, analyzable data.
Validation and Comparative Analysis: Benchmarking SPE Performance and Selectivity
The pursuit of efficient and reliable sample preparation methods has been a driving force in analytical chemistry, particularly within pharmaceutical and environmental research. For decades, liquid-liquid extraction (LLE) stood as the predominant technique for isolating analytes from complex matrices. However, the emergence and development of solid-phase extraction (SPE) since the 1970s marked a paradigm shift in extraction science, offering a modern alternative that addresses many of LLE's inherent limitations [18]. This transition reflects a broader trend in analytical methodology toward techniques that prioritize selectivity, efficiency, and environmental sustainability. The historical context of this evolution is crucial for understanding the current applications and capabilities of each method. SPE has progressed from early applications using activated carbon in the 1950s to the sophisticated, commercially available cartridges and disks that are standard in modern laboratories [2] [18]. This whitepaper provides a direct technical comparison between SPE and LLE, with a specific focus on their selectivity and solvent consumption, to guide researchers in selecting the optimal technique for their analytical challenges.
Working Principles and Historical Trajectories
Liquid-Liquid Extraction (LLE): A Classical Partitioning Method
LLE is a classical separation technique that relies on the differential solubility of analytes between two immiscible liquid phases, typically an aqueous phase (e.g., a sample matrix) and an organic solvent phase (e.g., ethyl acetate or dichloromethane) [96] [25]. The fundamental mechanism is a partition equilibrium, where analytes distribute themselves between the two phases based on their relative solubilities [96]. When vigorously mixed, the analytes migrate from the phase where they are less soluble to the phase where they are more soluble. The two phases are then allowed to separate, and the phase containing the enriched analytes is collected for further processing and analysis. Despite its long history of use, LLE faces several operational challenges, including the potential for emulsion formation, incomplete phase separation, and the requirement for large volumes of solvent, which is both costly and environmentally burdensome [96] [2].
Solid Phase Extraction (SPE): A Modern Sorbent-Based Approach
SPE operates on a fundamentally different principle. Instead of liquid-liquid partitioning, SPE utilizes a solid sorbent material (e.g., silica- or polymer-based) packed in a cartridge, disk, or 96-well plate to selectively retain target analytes from a liquid sample [96] [6]. The process is based on selective adsorption and desorption, where the chemical properties of the sorbent are chosen to have a high affinity for the target compounds [96]. The typical SPE procedure involves several critical steps designed to purify and concentrate the analytes, which are visually summarized in the workflow diagram below.
The historical development of SPE has been characterized by continuous innovation in sorbent technology and format configurations. Its first experimental applications began in the 1950s with activated carbon for analyzing organic traces in water [18]. A significant milestone occurred in 1977 with the introduction of pre-filled cartridges containing silica sorbents, which made the procedure more convenient and accessible [2]. The subsequent introduction of stable reversed-phase sorbents, such as C18-bonded silica, dramatically expanded SPE's applications in pharmaceutical, clinical, and environmental markets [2] [18]. The 1989 introduction of SPE disks, which contain sorbent enmeshed in a web of PTFE or glass fibers, enabled the processing of large sample volumes at higher flow rates without channeling [2] [18]. More recently, the development of 96-well SPE plates in the early 2000s created a world-wide standard for high-throughput bioanalysis, enabling the simultaneous processing of dozens of samples and seamlessly integrating with automation [97]. This evolution from simple cartridges to diverse formats has cemented SPE's role as a versatile and powerful sample preparation tool.
Direct Comparison: Selectivity and Solvent Consumption
The core differences between SPE and LLE become most apparent when comparing their selectivity and environmental impact, particularly regarding solvent use. The following table summarizes the key performance indicators that influence method selection.
Table 1: Quantitative Comparison of SPE and LLE Performance Characteristics
| Performance Factor | Solid Phase Extraction (SPE) | Liquid-Liquid Extraction (LLE) |
|---|---|---|
| Selectivity | High (driven by sorbent chemistry) [25] | Moderate (driven by solvent polarity) [25] |
| Solvent Consumption | Low to moderate [96] [25] | High (often 10x more than SPE) [96] |
| Reproducibility | High [96] | Variable (due to emulsions and phase separation issues) [96] |
| Automation Compatibility | Excellent (96-well formats, robots) [96] [97] | Poor (manual shaking and separation) [96] |
| Labor Time | Shorter, especially when automated [96] | Labor-intensive and manual [96] [98] |
| Environmental Impact | Lower solvent waste, greener [96] | Higher solvent disposal burden [96] |
Analysis of Selectivity
Selectivity refers to the ability of an extraction technique to isolate target analytes from a complex sample matrix while removing interfering compounds.
SPE Selectivity: SPE offers superior selectivity because it can be engineered at multiple levels. First, the sorbent chemistry (e.g., reversed-phase, ion-exchange, normal phase, mixed-mode) can be selected to match the physicochemical properties of the target analyte [6] [2]. Second, selectivity is further refined during the wash and elution steps. A carefully optimized wash solvent removes weakly bound interferences without displacing the analytes. Finally, a strong elution solvent is applied to recover the purified analytes [6]. This multi-stage process provides a high degree of cleanup, resulting in cleaner extracts and a better signal-to-noise ratio in downstream chromatographic analysis [96].
LLE Selectivity: The selectivity in LLE is primarily governed by a single factor: the polarity of the organic solvent used [25]. While effective for separating non-polar from polar compounds (e.g., extracting hydrophobic drugs from plasma), LLE offers limited ability to separate analytes with similar polarities. It is less effective for extracting polar compounds and provides fewer opportunities to remove interferences that co-extract with the targets due to similar solubility properties [2]. This often results in dirtier extracts and a higher potential for matrix effects in the analysis.
Analysis of Solvent Consumption
Solvent consumption is a critical consideration for cost, safety, and environmental sustainability.
LLE Solvent Use: LLE is notoriously solvent-intensive. It typically requires large volumes of organic solvent, often 10 times or more than the volume used in a comparable SPE protocol [96]. Furthermore, the process is often repeated multiple times to achieve high recovery, compounding solvent waste. This high consumption leads to a significant environmental disposal burden and increased operational costs [96] [25].
SPE Solvent Use: SPE is far more economical and environmentally friendly in its solvent use. The technique requires only small volumes of solvent for column conditioning, washing, and elution [96] [6]. The trend toward miniaturized formats, such as 96-well plates with sorbent bed weights as low as 2 mg, further reduces solvent consumption to the microliter scale [6] [2]. This reduction in solvent waste aligns with the principles of green chemistry and makes SPE a more sustainable choice for modern laboratories [96].
Detailed Methodologies and Experimental Protocols
Detailed Protocol for Solid Phase Extraction
The following steps outline a generalized SPE method using a cartridge format, which can be adapted based on sorbent chemistry and analyte properties [6].
- Sample Pre-Treatment: The sample is prepared to ensure optimal retention of the analytes. This may involve dilution, pH adjustment, or the addition of a buffer to ensure the analytes are in the correct ionic form. Particulates should be removed by filtration or centrifugation to prevent clogging of the sorbent bed [6].
- Column Conditioning: The SPE cartridge is prepared by passing 2-3 column volumes of a solvent that wet the sorbent and activate the functional groups. For reversed-phase sorbents, methanol or acetonitrile is typically used, followed by an equal volume of water or buffer to create a compatible environment for the aqueous sample.
- Column Equilibration: The sorbent is equilibrated with a solvent that matches the sample matrix (e.g., water or a buffer). It is critical not to let the sorbent run dry after this step, as channeling can occur, leading to poor and inconsistent recovery [6].
- Sample Application: The pretreated sample is loaded onto the cartridge. A controlled, slow flow rate (e.g., 1-5 mL/min) is recommended to ensure adequate time for the analytes to interact with and be retained by the sorbent [6].
- Wash: Interfering compounds that are weakly retained are selectively removed by applying 1-2 column volumes of a wash solvent. This solvent is strong enough to remove impurities but not so strong as to elute the target analytes. A common example is a water-methanol mixture for reversed-phase SPE.
- Elution: The target analytes are recovered by applying a strong solvent that disrupts the analyte-sorbent interactions. Using the smallest effective volume of a strong elution solvent (e.g., methanol with an acid for reversed-phase cation exchange) in two small aliquots, rather than one large volume, maximizes the concentration of the final extract [6].
Detailed Protocol for Liquid-Liquid Extraction
A standard LLE protocol, often performed in a separatory funnel, involves the following steps [96] [25]:
- Sample Preparation: The aqueous sample (e.g., plasma, urine, or water) may be buffered to a specific pH to ensure analytes are in their non-ionized form, facilitating their partitioning into the organic solvent.
- Extraction: The sample is combined with a volume of a water-immiscible organic solvent (e.g., ethyl acetate, dichloromethane, or hexane) in a separatory funnel. The ratio of sample to solvent volume can vary widely but is often in the range of 1:1 to 1:3.
- Mixing: The mixture is shaken vigorously for several minutes to maximize the surface area between the two immiscible phases, allowing the analytes to partition into the organic solvent based on their solubility.
- Phase Separation: The mixture is allowed to stand undisturbed until the two phases fully separate. This can be time-consuming if stable emulsions form at the interface.
- Collection: The lower phase (denser solvent) is carefully drained through the stopcock, and the upper phase (less dense solvent) is decanted or collected via pipette. The process (steps 2-5) is often repeated 2-3 times with fresh solvent to maximize recovery.
- Concentration: The combined organic extracts may be dried with an anhydrous salt (e.g., sodium sulfate) and then concentrated to a small volume using a nitrogen blow-down evaporator or rotary evaporator before analysis [99].
The Scientist's Toolkit: Essential Research Reagent Solutions
Selecting the appropriate materials is fundamental to developing a successful extraction method. The following table catalogs key reagents and their functions for both SPE and LLE.
Table 2: Essential Research Reagents for SPE and LLE Protocols
| Category | Reagent/Material | Primary Function in Extraction |
|---|---|---|
| SPE Sorbents | C18 (Octadecyl silica) [6] [2] | Reversed-phase extraction of non-polar to moderately polar analytes. |
| Ion-Exchange Sorbents [6] [2] | Selective retention of ionic compounds based on charge. | |
| Mixed-Mode Sorbents [2] | Combine reversed-phase and ion-exchange mechanisms for high selectivity. | |
| SPE Formats | Cartridges (1mL - 150mL) [6] | Versatile format for processing small to moderate sample volumes. |
| 96-Well Plates [6] [97] | High-throughput processing for bioanalysis, amenable to automation. | |
| LLE Solvents | Ethyl Acetate [100] | Medium-polarity solvent for extracting a wide range of semi-polar compounds. |
| Dichloromethane [100] | Dense, non-polar solvent; effective but has significant toxicity concerns. | |
| Hexane [6] [100] | Non-polar solvent for extracting lipids and very hydrophobic compounds. | |
| MTBE (Methyl tert-butyl ether) | Low-toxicity alternative to chlorinated solvents for LLE. | |
| Additives | Formic Acid / Ammonium Hydroxide [6] | pH modifiers to suppress or enhance analyte ionization for better retention/partitioning. |
| Buffers (e.g., Phosphate, Acetate) [6] | Maintain a stable pH during the extraction process. |
The choice between SPE and LLE is ultimately dictated by the sample matrix, the analytical goals, and available laboratory resources. The following decision diagram synthesizes the key selection criteria to guide researchers.
Application-Based Selection
- Prefer SPE when working with complex matrices (e.g., biological fluids, food homogenates, environmental water) that require effective cleanup [96]. It is the method of choice for achieving high reproducibility, lower detection limits, and when scalability or automation is essential [96] [25]. SPE is also strongly indicated for high-throughput laboratories processing many samples and for applications where reducing environmental impact is a priority [96].
- Consider LLE when processing very large sample volumes for the enrichment of non-polar or semi-polar analytes from relatively simple matrices [96] [25]. It remains a viable technique in legacy methods that have not been updated or in situations where the formation of emulsions is not a concern and the laboratory lacks access to SPE equipment or automation [96].
In conclusion, while both SPE and LLE are foundational techniques in analytical chemistry, SPE has established itself as the more modern, selective, and environmentally friendly option for most contemporary applications. Its development from a specialized technique to an analytical mainstay reflects the field's ongoing pursuit of efficiency and precision. For drug development professionals and researchers, leveraging the selectivity and low solvent consumption of SPE can lead to more robust, reproducible, and sustainable analytical methods, ultimately enhancing the quality and reliability of scientific data.
Solid-phase extraction (SPE) is a critical sample preparation technique in liquid chromatography-mass spectrometry (LC-MS)-based proteomic workflows [101]. Its primary role is to purify and concentrate peptide samples by removing interfering substances and ionic detergents, thereby enhancing peptide ionization and overall analytical performance [101]. The selection of an appropriate SPE method significantly influences the speed, performance, reproducibility, and automation potential of high-throughput proteomic data acquisition.
This case study fits within the broader context of early development research on SPE techniques, focusing on the comparative evaluation of different SPE phases and formats. As proteomics continues to bridge the gap between genomics and clinical manifestations, capturing dynamic protein events through reliable sample preparation becomes increasingly crucial for understanding cellular functions and disease mechanisms [102].
Methodology
Experimental Design
This comparative analysis utilized porcine retinal tissues, which provide an excellent model for studying human eye diseases, including glaucoma [101]. The study design incorporated:
- Protein Extraction: Two distinct protein fractions were prepared using different extraction buffers: 0.1% dodecyl-ß-maltoside (DDM) for cytoplasm-derived and membrane-associated proteins, and 1% trifluoroacetic acid (TFA) for nucleus-derived proteins, including histones and mitochondrial proteins [101].
- Sample Processing: Each protein fraction was subjected to in-gel trypsin digestion followed by purification using two different SPE-based workflows.
- Technical Replicates: Six technical replicates for each protein fraction were processed to ensure statistical reliability (N = 3 per SPE method) [101].
- LC-MS Analysis: Purified peptides were analyzed using LC-MS/MS to evaluate the performance of both SPE methods.
SPE Methods Compared
The study evaluated two commercial SPE-based sample preparation protocols:
- ZIPTIP C18 Pipette Tips (Merck Millipore): A traditional manual purification method using C18 sorbent material [101].
- SOLAμ HRP SPE Spin Plates (Thermo Fisher Scientific): A more modern, semi-automated platform utilizing hydrophilic resin-based particles (HRP) in a spin plate format designed for higher throughput processing [101].
Data Analysis
The analytical performance was assessed based on:
- Qualitative Analysis: Number of proteins and peptides identified with a false discovery rate (FDR) < 1% [101].
- Quantitative Analysis: Recovery rates of 25 predefined glaucoma-related retinal protein markers.
- Statistical Analysis: Student's t-test for protein/peptide identification counts and Mann-Whitney U-test for protein ion score distributions [101].
- Reproducibility: Assessment of technical variability across replicates.
- Throughput: Evaluation of processing time and convenience for each method.
Table 1: Key Research Reagent Solutions and Materials
| Item | Function in Protocol |
|---|---|
| Porcine Retinal Tissues | Biological source for protein extraction; excellent model for human eye diseases [101]. |
| Dodecyl-ß-maltoside (DDM) | Detergent for extracting cytoplasm-derived and membrane-associated proteins [101]. |
| Trifluoroacetic Acid (TFA) | Acidic solution for extracting nucleus-derived proteins, including histones [101]. |
| Trypsin | Enzyme for digesting extracted proteins into peptides for MS analysis [101]. |
| ZIPTIP C18 Pipette Tips | Traditional SPE format with C18 sorbent for peptide desalting and purification [101]. |
| SOLAμ HRP SPE Spin Plates | Semi-automated SPE platform with hydrophilic resin particles for higher throughput purification [101]. |
| Nanoflow LC-MS System | Analytical instrument for separating, detecting, and identifying purified peptides [103]. |
Diagram 1: Experimental workflow for comparative SPE performance analysis.
Results and Discussion
Qualitative Performance: Protein and Peptide Identification
Both SPE methods demonstrated comparable effectiveness in identifying proteins and peptides from complex retinal tissue samples. The following table summarizes the identification results for both protein fractions across the two SPE methods:
Table 2: Protein and Peptide Identification Results by SPE Method and Extraction Buffer
| Sample Fraction | SPE Method | Proteins Identified | Peptides Identified |
|---|---|---|---|
| DDM | ZIPTIP C18 | 550 ± 70 | 1,512 ± 199 |
| DDM | SOLAμ HRP | 513 ± 55 | 1,347 ± 180 |
| TFA | ZIPTIP C18 | 305 ± 48 | 806 ± 144 |
| TFA | SOLAμ HRP | 300 ± 33 | 722 ± 87 |
Statistical analysis revealed no significant differences (P > 0.05) in the number of proteins or peptides identified between the two SPE methods for either protein fraction [101]. Venn diagram analysis further demonstrated a substantial overlap in protein identifications, with an average overlap of 65 ± 2% for the DDM fraction and 69 ± 4% for the TFA fraction between both SPE-based methods [101].
The distribution of protein ion scores and peptide molecular weights showed similar patterns between both methods, indicating no significant bias in the detection of high-confidence proteins or specific peptide classes [101].
Quantitative Performance: Recovery of Target Protein Markers
The quantitative analysis focused on 25 glaucoma-related retinal protein markers to assess potential differences in protein recovery between the two SPE methods. The results demonstrated remarkable consistency:
- Only one protein marker, MECP2, showed a statistically significant (P = 0.02) higher abundance in ZIPTIP-purified replicates compared to SOLAμ-treated samples [101].
- This isolated difference was not confirmed in a subsequent verification experiment using recombinant MECP2 (P = 0.24), suggesting it was not a systematic methodological bias [101].
- For the remaining 24 protein markers, no significant differences (P > 0.05) in protein recovery were observed between the two SPE purification methods [101].
Throughput and Practical Considerations
While analytical performance was equivalent, significant practical differences emerged:
- The SOLAμ HRP SPE spin plate workflow offered substantially improved analysis speed and semi-automation capabilities compared to the manual ZIPTIP C18 method [101].
- The spin plate format enabled more convenient processing of multiple samples simultaneously, reducing hands-on time and increasing throughput.
- The potential for semi-automation made the SOLAμ platform better suited for high-throughput proteomic study designs requiring minimal manual intervention.
Diagram 2: Key performance findings from SPE method comparison.
This comparative case study demonstrates that both ZIPTIP C18 pipette tips and SOLAμ HRP SPE spin plates perform equally well in terms of analytical capabilities for proteomic analysis of retinal tissues. Both methods showed comparable protein and peptide identification rates, similar protein ion score distributions, and equivalent recovery of target protein markers.
The critical differentiator lies in operational efficiency. The SOLAμ HRP SPE spin plate method provides significantly improved analysis speed and semi-automation capabilities without compromising data quality. This advantage makes it particularly suitable for high-throughput proteomic workflows where processing efficiency and reduced manual intervention are priorities.
These findings contribute valuable insights to the early development of SPE techniques, highlighting that while traditional and modern SPE phases can deliver equivalent analytical results, format innovations substantially impact practical implementation. Future SPE development should continue to focus on automation compatibility, throughput enhancement, and user convenience while maintaining robust analytical performance.
In the early development of solid phase extraction (SPE) techniques, the evaluation of three key metrics—recovery, reproducibility, and analysis speed—forms the cornerstone of robust analytical method development. SPE serves as "the silent chromatography," employing the same principles as liquid chromatography but without an in-line detector, making the optimization of these metrics crucial for success [104]. This technical guide provides a structured framework for researchers and drug development professionals to systematically develop, troubleshoot, and validate SPE methods, ensuring data quality and efficiency from initial discovery through development phases.
Core SPE Principles and Metric Interrelationships
Fundamental Separation Mechanisms
SPE sorbents operate through several primary retention mechanisms, each with distinct applications and optimization requirements [62]:
- Nonpolar Phases (e.g., C18, C8): Retain analytes via van der Waals forces; ideal for hydrophobic compounds from polar matrices like water.
- Polar Phases (e.g., silica, Florisil): Utilize dipole-dipole or hydrogen bonding interactions; suited for polar analytes from nonpolar matrices.
- Ion Exchange Phases: Employ electrostatic interactions for charged analytes, available in strong/weak anion or cation exchange varieties.
- Mixed-Mode Phases: Combine multiple mechanisms (typically reversed-phase and ion exchange) for highly selective extractions from complex matrices.
Interdependence of Key Metrics
The relationship between recovery, reproducibility, and analysis speed involves critical trade-offs. For instance, higher recoveries often require more thorough (and time-consuming) washing and elution steps, potentially impacting analysis speed. Similarly, optimal reproducibility demands严格控制 flow rates and conditioning, which may limit maximum processing speed. Understanding these interrelationships enables researchers to strategically balance method parameters based on their specific analytical objectives.
Quantitative Evaluation of SPE Performance
Recovery: Measurement and Optimization
Recovery, expressed as the percentage of analyte successfully extracted from the sample matrix, is the fundamental measure of extraction efficiency [105].
Table 1: SPE Sorbent Capacity Guidelines by Chemistry
| Sorbent Type | Capacity Guideline | Example (100 mg sorbent) | Key Influencing Factors |
|---|---|---|---|
| Silica-Based | ≤5% of sorbent mass | Can retain ~5 mg of compound | Surface area, end-capping, ligand density |
| Polymer-Based | ~3x silica capacity | Can retain ~15 mg of compound | Hydrophilic-lipophilic balance, surface area |
| Ion Exchange | 0.25-1 mmol/g | 1g sorbent adsorbs 1 mmol monovalent charged substance | pH, ionic strength, counter-ion selection |
Methodology for recovery calculation:
When recoveries exceed 100%, this typically indicates methodological issues requiring investigation [74]:
- Co-eluting interferences: Matrix components eluting with target analytes
- System contaminants: Impurities in solvents or sorbents
- Internal standard compromise: Inaccurate reference points for quantification
- Calculation errors: Incorrect integration or dilution factor accounting
Reproducibility: Ensuring Method Robustness
Reproducibility in SPE encompasses both precision (inter- and intra-assay variability) and method robustness. Key controlling parameters include:
Table 2: Factors Influencing SPE Reproducibility
| Parameter | Impact on Reproducibility | Optimization Strategy |
|---|---|---|
| Flow Rate | High flow rates cause channeling and breakthrough; low rates increase processing time | Maintain 1 mL/min for cartridges; use consistent vacuum/pressure [6] |
| Sorbent Conditioning | Inconsistent conditioning causes poor retention and variable recovery | Prevent column drying; use adequate solvent volumes [78] |
| Sample Pretreatment | Variable pH or ionic strength alters retention mechanics | Adjust sample pH to neutralize charges for reversed-phase; ensure charge for ion exchange [78] |
| Wash Stringency | Incomplete interference removal or analyte loss | Balance between cleanliness (strong wash) and recovery (weak wash) [78] |
Analysis Speed: Throughput Optimization
Analysis speed encompasses total sample processing time, influenced by several factors [105]:
- Sorbent particle size: Smaller particles increase surface area but may reduce flow rates
- Cartridge format selection: Match cartridge size to sample volume (Table 3)
- Flow rate optimization: Balance between efficient mass transfer and practical processing time
- Automation potential: 96-well plates enable higher throughput versus cartridges
Table 3: SPE Cartridge Selection Guidelines by Sample Volume
| Cartridge Volume | Typical Sorbent Mass | Sample Size Range | Minimum Elution Volume |
|---|---|---|---|
| 1 mL | 50-100 mg | 2.5-10 mg | 100-200 µL |
| 3 mL | 500 mg | 25-100 mg | 1-3 mL |
| 6 mL | 500-1000 mg | 25-100 mg | 2-6 mL |
| 12 mL | 2000 mg | 100-200 mg | 10-12 mL |
Experimental Protocols for Metric Evaluation
Comprehensive SPE Workflow
The following experimental workflow ensures systematic evaluation of all key metrics:
Diagram: Comprehensive SPE Workflow for Method Evaluation
Protocol for Recovery and Reproducibility Assessment
Materials: Appropriate SPE cartridges, sample, internal standards, conditioning solvents (methanol, acetonitrile), aqueous buffer, wash solvents, elution solvents [6].
Procedure:
- Sample Pretreatment: Adjust sample pH to ensure analytes are in appropriate form for retention (neutral for reversed-phase, charged for ion exchange). Dilute if necessary to reduce ionic strength [78].
- Conditioning: Pass 2-3 column volumes of methanol or acetonitrile through sorbent, followed by 2-3 volumes of aqueous solution or buffer matching sample pH. Do not allow sorbent to dry completely [6].
- Equilibration: Pass 2-3 volumes of solvent matching the sample matrix through conditioned sorbent.
- Sample Loading: Apply sample at controlled flow rate (typically 1 mL/min). Collect flow-through for breakthrough analysis if troubleshooting [78].
- Washing: Apply 2-3 column volumes of wash solvent strong enough to remove interferences but weak enough to retain analytes. Collect wash fraction for analysis if troubleshooting recovery issues.
- Elution: Apply 2 aliquots of strong elution solvent, allowing first aliquot to sit in cartridge for 30-60 seconds before applying gentle pressure. Collect eluate for analysis [78].
- Analysis: Quantify analyte in appropriate fractions and calculate recovery. Perform minimum of 6 replicates to assess reproducibility.
Protocol for Analysis Speed Optimization
Materials: SPE cartridges of varying sizes, sorbents with different particle sizes, vacuum manifold or positive pressure system, timer.
Procedure:
- Flow Rate Calibration: Measure actual flow rates at different vacuum/pressure settings using water.
- Breakthrough Testing: Load sample at increasing flow rates while analyzing flow-through to determine maximum rate without significant analyte loss.
- Elution Efficiency: Test different elution solvent volumes and application methods (single vs. multiple aliquots) to determine minimum effective volume and time.
- Comparative Format Assessment: Process identical samples using cartridge and 96-well plate formats, comparing total processing time per sample.
Troubleshooting Common SPE Performance Issues
Low Recovery Scenarios and Solutions
- Problem: Analyte loss during loading.
- Investigation: Analyze flow-through fraction.
- Solution: Optimize sample pH; change sorbent chemistry; reduce flow rate; check sorbent capacity [78].
- Problem: Analyte loss during washing.
- Investigation: Analyze wash fraction.
- Solution: Use weaker wash solvent; adjust wash solvent pH [74].
- Problem: Incomplete elution.
- Investigation: Perform second elution with stronger solvent.
- Solution: Increase elution solvent strength; use multiple aliquots; add modifiers; increase contact time [78].
Poor Reproducibility Scenarios and Solutions
- Problem: Variable recovery between replicates.
- Investigation: Check flow rate consistency; review conditioning procedure.
- Solution: Use calibrated vacuum/pressure; ensure consistent conditioning; implement internal standardization [74].
- Problem: Column drying during processing.
- Investigation: Monitor solvent meniscus above sorbent bed.
- Solution: Do not allow sorbent to dry after conditioning; maintain approximately 1mm of solvent above frit [6].
Matrix Effect Evaluation and Mitigation
Matrix effects, particularly critical in LC-MS applications, occur when co-eluting compounds alter analyte ionization efficiency. Evaluation methodology:
- Post-Infusion Analysis: Infuse analyte standard while injecting extracted blank matrix to detect suppression/enhancement regions.
- Post-Extraction Spiking: Compare analyte response in spiked extracted matrix versus pure solution.
- Mitigation Strategies: Use matrix-matched calibration standards; improve extraction selectivity; implement effective internal standards [106].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 4: Essential Research Reagents and Materials for SPE Method Development
| Item | Function | Selection Criteria |
|---|---|---|
| Reversed-Phase Sorbents (C18, C8, HLB) | Retain hydrophobic analytes from polar matrices | HLB for broad spectrum; C18 for highly non-polar compounds [106] |
| Ion Exchange Sorbents (MCX, MAX, WCX, WAX) | Retain ionizable compounds via electrostatic interactions | Strong exchangers for weak analytes; weak exchangers for strong analytes [62] |
| Mixed-Mode Sorbents | Combine multiple retention mechanisms for cleaner extracts | Ideal for basic/acidic drugs in complex matrices [62] |
| Conditioning Solvents (Methanol, Acetonitrile) | Activate sorbent and wet functional groups | Match solvent strength to application; methanol more polar, acetonitrile less viscous |
| Elution Solvents (Methanol, ACN with modifiers) | Disrupt analyte-sorbent interactions for recovery | Include pH modifiers (e.g., formic acid, ammonia) for ionizable compounds [78] |
| Internal Standards | Normalize for variability in extraction and analysis | Stable isotope-labeled analogs preferred; structurally similar compounds acceptable [74] |
Advanced SPE Methodologies and Emerging Trends
Miniaturized and Automated SPE Formats
Recent advances in SPE technology focus on miniaturization and automation to enhance reproducibility and analysis speed:
- µElution Plates: Designed for low-volume samples, minimizing analyte loss from non-specific binding [106].
- On-Line SPE: Integrates extraction with analytical instrumentation, reducing manual handling and improving reproducibility [107].
- Magnetic SPE (MSPE): Utilizes magnetic sorbents for simplified phase separation, increasing processing speed [107].
Method Validation and Quality Control
For regulatory applications, implement comprehensive validation protocols assessing:
- Accuracy and Precision: Across relevant concentration range (minimum 6 replicates).
- Matrix Effects: Quantify suppression/enhancement in target matrices.
- Stability: Evaluate analyte stability in matrix during storage and processing.
- System Suitability: Establish QC criteria for ongoing method performance verification.
The systematic evaluation of recovery, reproducibility, and analysis speed provides a framework for developing robust SPE methods in early research and development. By understanding the fundamental principles, implementing structured experimental protocols, and utilizing appropriate troubleshooting strategies, researchers can optimize these critical metrics to meet their specific analytical needs. The continued evolution of SPE technologies, particularly in miniaturization and automation, promises further enhancements in method performance and efficiency for pharmaceutical and bioanalytical applications.
The Role of SPE in Method Standardization and Regulatory Compliance (e.g., EPA Methods)
The adoption of Solid-Phase Extraction (SPE) as a standardized sample preparation technique represents a critical advancement in analytical chemistry, particularly for regulatory compliance across environmental, pharmaceutical, and food safety sectors. SPE is a sample preparation technique that utilizes a solid sorbent to selectively isolate, concentrate, and purify target analytes from liquid samples [2]. Its development has fundamentally addressed the need for reliable, reproducible, and efficient sample handling prior to instrumental analysis [18]. The technique operates on principles similar to liquid chromatography, where analytes distribute between a liquid sample and a solid sorbent phase, followed by elution with an appropriate solvent [2].
The standardization of SPE methods by regulatory bodies like the United States Environmental Protection Agency (EPA) has been instrumental in ensuring data quality, comparability, and regulatory enforcement. By providing detailed, validated protocols, these standardized methods transform SPE from a research technique into an indispensable tool for compliance monitoring. This technical guide explores the historical context, regulatory framework, methodological specifics, and future directions of SPE in standardized analytical methods, with particular focus on EPA Method 3535A.
Historical Development and Evolution of SPE
The foundations of SPE were laid over five decades ago, with its development characterized by distinct evolutionary phases focused on sorbent materials and technical configurations.
The Early Ages of SPE Development
The initial experimental applications of SPE, beginning in the 1950s, primarily addressed the challenge of analyzing organic traces in water samples [18]. The first age of SPE was dominated by the use of activated carbon filters for recovering organic compounds from water matrices [18]. While effective for concentration, carbon filters presented significant challenges for quantitative recovery due to strong, often irreversible adsorption of certain analytes [18].
The period from the late 1960s to the early 1980s marked an intensive search for more appropriate sorbent materials [18]. Researchers explored various alternatives, including porous polymer sorbents such as styrenedivinylbenzene, which offered improved performance over carbon for many applications [18]. A significant breakthrough came with the introduction of pre-filled cartridges containing silica-based sorbents in 1977, which dramatically improved convenience and reproducibility [2]. The development of chemically bonded phases, particularly reversed-phase sorbents like C18, further expanded SPE applications into environmental, clinical, and pharmaceutical markets [2] [18].
Technical Configurations and Formats
The evolution of SPE devices has progressed through several formats, each addressing specific application needs:
- Cartridge Formats: The most common disposable format, comprising a polypropylene syringe barrel packed with sorbent between two frits [2]. Sorbent amounts range from milligrams to grams depending on sample volume [2].
- Disk Formats: Introduced in 1989, disks embed sorbent particles within a matrix, providing greater cross-sectional area and faster flow rates, particularly advantageous for large volume environmental samples [2].
- 96-Well Plate Systems: Developed in the 1990s, this format enabled high-throughput bioanalysis, becoming a worldwide standard for pharmaceutical analysis of small molecules [97].
- Pipette-Tip SPE (PT-SPE): Miniaturized formats using very small sorbent amounts (4-400 µg) for biological sample processing with minimal solvent volumes [2].
Figure 1: Historical Development Timeline of SPE Technologies
Regulatory Framework and EPA Method Standardization
EPA Method 3535A: A Case Study in Standardization
EPA Method 3535A, titled "Solid-Phase Extraction (SPE)," is part of the EPA's SW-846 compendium on hazardous waste test methods [108] [109]. This method provides a standardized procedure for isolating target organic analytes from aqueous samples using solid-phase extraction media [109]. The method's primary objective is to ensure consistent sample preparation across different laboratories and analysts, thereby guaranteeing the reliability and comparability of data used for regulatory decision-making.
The method finds specific application within EPA's Selected Analytical Methods for Environmental Remediation and Recovery (SAM), where it is listed for preparation of water samples to be analyzed for numerous compounds including pesticides, pharmaceuticals, explosives, and chemical warfare agents [108]. Specific analytes mentioned include 4-aminopyridine, carfentanil, chlorfenvinphos, diesel range organics, fentanyl, HMTD, RDX, kerosene, nicotine compounds, HMX, PETN, strychnine, and various TNT compounds [108].
The Standardization Advantage
The incorporation of SPE into standardized methods like EPA 3535A provides several critical advantages for regulatory compliance:
- Method Uniformity: All laboratories follow identical procedures, enabling direct comparison of results across different monitoring programs and temporal studies [18].
- Quality Control: Standardized methods include explicit quality control requirements, such as recovery validation through spiking experiments, ensuring data integrity [110].
- Defensible Data: Results generated using approved methods withstand legal and regulatory scrutiny, forming a reliable basis for enforcement actions [18].
- Efficiency Gains: SPE reduces solvent consumption by over 90% compared to traditional liquid-liquid extraction, aligning with green chemistry principles while maintaining analytical performance [2] [110].
Technical Protocols and Methodologies
Fundamental SPE Operational Procedure
The standard SPE procedure comprises four critical steps that must be rigorously controlled to ensure reproducible results:
Figure 2: Core SPE Procedure Workflow
Step 1: Activation and Conditioning The sorbent must be properly activated to ensure optimal analyte retention. This typically involves preconditioning with an organic solvent (e.g., methanol) to wet the stationary phase, followed by equilibration with water or a buffer matching the sample matrix [110]. Proper activation prevents inconsistent retention and improves extraction efficiency [110].
Step 2: Sample Loading The prepared sample solution is passed through the activated sorbent, allowing selective retention of target analytes [110]. Flow rate control is critical during this step, as excessively fast flow rates can lead to incomplete adsorption, while excessively slow rates may cause sample loss due to sorbent drying [110].
Step 3: Washing A carefully chosen weak solvent is applied to remove non-specifically retained interferences while maintaining strong retention of target analytes [110]. For reversed-phase SPE, water or low-concentration organic solvent removes hydrophilic impurities; for ion-exchange SPE, low-ionic-strength buffer removes weakly bound contaminants [110].
Step 4: Elution Target compounds are recovered using a strong solvent that disrupts sorbent-analyte interactions [110]. The elution solvent must maximize analyte solubility while minimizing co-extraction of interferences [71] [110]. The eluted fraction is collected for subsequent analysis.
Sorbent Selection Guide
Choosing the appropriate sorbent chemistry is paramount for method success. The selection depends on analyte properties and matrix characteristics:
Table 1: SPE Sorbent Selection Guide Based on Analyte Properties
| Sorbent Type | Retention Mechanism | Typical Applications | Target Analyte Properties |
|---|---|---|---|
| Reversed-Phase (C18, C8) | Hydrophobic interactions | Environmental pollutants, pharmaceuticals, pesticides | Non-polar to moderately polar compounds [110] |
| Normal-Phase (Silica, CN, NH₂) | Polar interactions (hydrogen bonding, dipole-dipole) | Natural products, food additives, polar pesticides | Polar compounds soluble in non-polar solvents [110] |
| Ion-Exchange (SAX, SCX) | Electrostatic interactions | Antibiotics, organic acids, nucleotides | Charged compounds (acids, bases) [71] [110] |
| Mixed-Mode | Combined hydrophobic and ionic interactions | Drugs of abuse, pharmaceuticals from biological fluids | Compounds with both hydrophobic and ionic character [71] |
| Molecularly Imprinted Polymers (MIP) | Shape-selective recognition | Specific environmental pollutants, biomarkers | Compounds matching template structure [71] |
Experimental Comparison of SPE Methodologies
Recent research has quantitatively compared different SPE approaches for various applications:
Table 2: Performance Comparison of SPE Clean-up Approaches for Antibiotic Analysis in Vegetables [111]
| Parameter | Conventional SPE Clean-up | Dispersive SPE (dSPE) Clean-up |
|---|---|---|
| Analytes | Tetracyclines (TCs) and Sulfonamides (SAs) | Tetracyclines (TCs) and Sulfonamides (SAs) |
| Matrices | Lettuce, onion, tomato, carrot | Lettuce, onion, tomato, carrot |
| Recovery | >70% for most analytes | >70% for most analytes |
| Precision (RSD) | <30% for all matrices | <30% for all matrices |
| Limits of Quantification | 4.4 μg/kg for most TCs; 3.0 μg/kg for most SAs | 0.1-3.7 μg/kg (generally lower) |
| Matrix Effect | Better for TCs | Better for most SAs |
| Application | Regulatory monitoring where established methods exist | High-throughput screening with potentially better sensitivity |
Another study comparing SPE formats for proteomic analysis found that different SPE pipette tips and spin plates showed comparable performance in protein identifications, with no significant differences (P > 0.05) in protein and peptide identifications using Student's t-test [48]. The study highlighted that analysis speed and semi-automation potential varied significantly between formats, emphasizing the importance of selecting appropriate configurations for specific laboratory workflows [48].
Advanced SPE Chemistries and Configurations
Advanced Sorbent Technologies
Modern SPE has evolved beyond conventional phases to include specialized sorbents that address complex analytical challenges:
Restricted Access Media (RAM) RAM sorbents feature dual-mode characteristics with an outer hydrophilic layer that excludes macromolecules like proteins, and an inner hydrophobic surface that retains small analytes [71]. These materials enable direct injection of biological fluids such as plasma or serum, significantly simplifying sample preparation [71]. RAM phases can be used in single-column mode or with multidimensional LC-LC systems for enhanced separation [71].
Molecularly Imprinted Polymers (MIP) MIPs represent the pinnacle of selectivity in SPE, created by polymerizing functional monomers around a template molecule [71]. After template removal, the resulting polymer contains specific recognition sites complementary to the target analyte in size, shape, and functional group orientation [71]. These sorbents provide exceptional specificity and are particularly valuable for analyzing complex matrices where conventional SPE lacks sufficient selectivity [71].
Mixed-Mode Sorbents Mixed-mode sorbents incorporate multiple retention mechanisms, typically combining reversed-phase and ion-exchange properties [71]. This enables selective retention of both neutral and charged compounds within a single cartridge [71]. Experimentally, multimodal extraction can be achieved through serial connection of different cartridges or using a single cartridge containing multiple functional groups or blended sorbents [71].
Automation and High-Throughput Formats
The development of 96-well SPE systems revolutionized high-throughput bioanalysis, particularly in pharmaceutical applications [97]. This format enabled simultaneous processing of numerous samples, dramatically improving laboratory productivity [97]. The 96-well format has since been extended to include disk-phase extraction, supported liquid-liquid extraction, and 384-well systems [97].
Automated SPE approaches, whether using robotic liquid handlers or on-line column switching systems, provide significant advantages including reduced labor, improved reproducibility, and operation in closed systems that minimize sample oxidation or solvent evaporation [71].
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key SPE Research Reagents and Materials
| Reagent/Material | Function/Application | Technical Considerations |
|---|---|---|
| C18 Bonded Silica | Reversed-phase extraction of non-polar compounds | Pore size (60Å), surface area, and endcapping affect performance [2] [110] |
| Strong Cation Exchange (SCX) Resin | Extraction of basic compounds | Capacity depends on surface charge density; pH control critical [71] [110] |
| Hydrophilic-Lipophilic Balanced (HLB) Polymer | Mixed-mode extraction for broad analyte spectrum | Stable with dry conditions; suitable for pH variations [110] |
| Molecularly Imprinted Polymers (MIP) | Highly selective extraction of target compounds | Template-specific; excellent for complex matrices [71] |
| Restricted Access Media (RAM) | Direct injection of biological fluids | Excludes macromolecules while retaining small analytes [71] |
| Disk Format Membranes | Processing large volume samples | Higher flow rates; reduced clogging [2] |
| 96-Well SPE Plates | High-throughput processing | Compatible with automation; various sorbent chemistries available [97] |
Solid-Phase Extraction has evolved from its early experimental beginnings into a sophisticated, standardized technology that underpins modern analytical compliance across multiple sectors. The continued development of selective sorbents, automated formats, and miniaturized systems ensures that SPE will remain integral to analytical chemistry. Future advancements will likely focus on increasing selectivity through novel materials like molecularly imprinted polymers, enhancing automation capabilities, and further reducing solvent consumption in alignment with green chemistry principles [2] [71].
The historical development of SPE demonstrates how a fundamental sample preparation technique, when standardized and validated through methods like EPA 3535A, becomes indispensable for regulatory compliance, method validation, and reliable data generation across environmental, pharmaceutical, and food safety applications. As analytical challenges continue to evolve with emerging contaminants and increasingly complex matrices, SPE technologies will similarly advance to meet these demands while maintaining the rigorous standards required for regulatory applications.
Solid Phase Extraction (SPE) has established itself as a cornerstone technique in modern analytical laboratories, particularly within pharmaceutical research and environmental monitoring. Its advantages over traditional extraction methods are driving significant market growth, with the global SPE market projected to expand at a Compound Annual Growth Rate (CAGR) of 6.5% to 7.2%, potentially reaching values between $846 million and over $900 million by 2032-2033 [112] [113] [114]. This growth is fueled by SPE's core benefits: high selectivity for targeted analyte isolation, superior ease of automation for high-throughput workflows, and significantly lower solvent consumption compared to methods like Liquid-Liquid Extraction (LLE), supporting greener laboratory practices [96] [115] [116]. This whitepaper delves into the technical underpinnings of these advantages, providing a detailed comparative analysis and practical experimental protocols to guide researchers in the early development of SPE methodologies.
Quantitative Comparison: SPE vs. Alternative Techniques
The operational superiority of SPE becomes evident when its key performance indicators are directly compared with those of traditional Liquid-Liquid Extraction (LLE). The following table synthesizes data from recent market analyses and comparative studies to highlight these distinctions.
Table 1: Performance Comparison of SPE vs. LLE
| Factor | Solid Phase Extraction (SPE) | Liquid-Liquid Extraction (LLE) |
|---|---|---|
| Solvent Consumption | Low [96] | High (often 10x or more than SPE) [96] |
| Automation Compatibility | Excellent (96-well formats, robots) [96] | Poor (manual shaking and separation) [96] |
| Reproducibility | High [96] | Variable (due to risk of emulsions and phase separation issues) [96] |
| Labor Time | Shorter, especially when automated [96] | Labor-intensive and manual [96] |
| Selectivity | High (based on selective adsorption/desorption) [96] [116] | Moderate (based on partition equilibrium and solubility) [96] |
| Environmental Impact | Lower solvent waste, greener [96] | Higher solvent disposal burden [96] |
| Data Quality | Cleaner extracts, better signal-to-noise ratio [96] | Higher risk of contamination and analyte loss [96] |
A specific comparative study analyzing opioids and benzodiazepines in urine samples further quantified the performance of an SPE method against Dispersive Liquid-Liquid Microextraction (DLLME). The SPE method demonstrated superior recovery rates for the target analytes, underscoring its effectiveness for complex matrices [117].
Table 2: Experimental Recovery Data from a Comparative Method Study [117]
| Extraction Method | Recovery Range | Key Solvents/Consumables Used |
|---|---|---|
| SPE | 9% to 107% | ExtraBond SCX (mixed-mode sorbent), 7 mL of 5% NH₄OH in Methanol |
| DLLME | 14% to 86% | 200 µL Chloroform (extractant), 500 µL Ethyl Acetate (dispersant) |
While the DLLME method was noted for its lower absolute solvent volume and faster extraction time, the SPE method provided more consistent and higher recoveries for a broader range of analytes within the tested panel, which is critical for reliable quantitative analysis [117].
Core Technical Advantages of Solid Phase Extraction
High Selectivity
The high selectivity of SPE is derived from the sophisticated interaction between the analyte and the solid sorbent. Unlike LLE, which primarily separates compounds based on solubility in two immiscible liquids, SPE utilizes mechanisms such as reverse-phase, ion-exchange, and normal-phase interactions on a solid stationary phase [96] [116]. This allows researchers to selectively retain target analytes while washing away interfering compounds from the sample matrix.
The selectivity can be finely tuned by selecting an appropriate sorbent chemistry. For instance:
- Mixed-mode sorbents combine reverse-phase and ion-exchange properties, enabling the selective extraction of acidic or basic compounds based on both hydrophobicity and ionic state [117].
- Immunoaffinity sorbents use antibody-antigen interactions for exceptionally high specificity towards a single compound or class [114].
This targeted approach results in cleaner extracts, which significantly reduces matrix effects during subsequent analysis by LC-MS/MS and improves the signal-to-noise ratio, ultimately leading to more accurate and reliable data [96].
Ease of Automation
SPE is inherently more adaptable to automation than LLE, which relies on manual steps like shaking and phase separation that are difficult to standardize. Automated SPE systems can process samples in 96-well plates or higher densities, dramatically increasing laboratory throughput [96] [114].
The shift towards automation is a dominant trend in the SPE market, driven by the need for:
- Increased Efficiency: Unattended operation frees up skilled personnel for other tasks [114].
- Enhanced Reproducibility: Automated systems minimize human error and variability inherent in manual techniques like LLE, leading to more robust and reliable results [96].
- High-Throughput Processing: Essential for modern applications in drug discovery, clinical toxicology, and environmental monitoring, where hundreds of samples must be processed daily [112] [113].
Further integration of SPE with analytical instruments, such as online SPE-LC-MS systems, creates seamless, fully automated workflows from sample preparation to data acquisition, reducing analysis time and the potential for contamination [118] [114].
Low Solvent Consumption
SPE is recognized as a more environmentally friendly technique due to its significantly lower consumption of organic solvents. As shown in Table 1, LLE can use ten times the solvent volume compared to SPE [96]. A typical SPE protocol requires only a few milliliters of solvent for conditioning, washing, and elution.
The move towards green chemistry is a powerful driver in the sample preparation market [115] [116]. The reduced solvent usage in SPE directly translates to:
- Lower operational costs for solvent purchase and hazardous waste disposal [96] [114].
- A safer working environment for laboratory personnel.
- A reduced environmental footprint, aligning with global sustainability initiatives.
Innovations continue to push this advantage further, with developments in miniaturized SPE formats (e.g., µSPE, tip-based SPE) and novel sorbents designed to work with smaller solvent volumes or greener solvent alternatives [116] [114].
Experimental Protocol: SPE Method for Complex Matrices
The following detailed protocol, adapted from a published comparative study, outlines the steps for extracting analytes from a complex biological matrix, such as urine, using a mixed-mode sorbent [117]. This serves as a practical template for method development.
Diagram 1: SPE Experimental Workflow
Materials and Reagents (Research Toolkit)
Table 3: Essential Reagents and Materials for SPE
| Item | Function/Description | Example (From Protocol) |
|---|---|---|
| SPE Cartridge/Plate | Solid support containing the selective sorbent. | ExtraBond SCX mixed-mode (cation-exchange) cartridges [117]. |
| Conditioning Solvent | Activates the sorbent and prepares it for sample interaction. | Methanol [117]. |
| Equilibration Solvent | Creates the optimal chemical environment for analyte retention. | Water or a buffer compatible with the sorbent chemistry [117]. |
| Elution Solvent | Disrupts analyte-sorbent interaction to recover the target compound. | 5% NH₄OH in Methanol for eluting basic compounds from a mixed-mode sorbent [117]. |
| Vacuum Manifold or Automated System | Provides positive or negative pressure to drive liquids through the sorbent bed. | Automated SPE workstations or 24-port manual manifolds [116]. |
Detailed Step-by-Step Procedure
- Sample Preparation: Adjust the pH of the sample (e.g., urine) to ensure the target analytes (e.g., opioids, benzodiazepines) are in a non-protonated state for optimal interaction with a reverse-phase sorbent, or in a charged state for ion-exchange. Centrifuge to remove any particulate matter [117].
- Sorbent Conditioning: Pass 1-2 mL of methanol through the sorbent bed under gentle vacuum. This solvates the functional groups and prepares the surface for interaction. Follow immediately with 1-2 mL of water or a weak buffer (e.g., pH 7.0 phosphate buffer) to create an environment compatible with the aqueous sample. Do not allow the sorbent to run dry after conditioning [117] [116].
- Sample Loading: Slowly load the prepared sample onto the conditioned SPE cartridge. A slow, drop-wise flow rate is critical to maximize contact time and ensure quantitative retention of the analytes on the sorbent [117].
- Washing: Remove undesired matrix components by passing through one or two wash solutions. A typical sequence might be 1-2 mL of water (to remove salts and polar impurities) followed by 1-2 mL of a methanol-water mixture (e.g., 20:80 v/v) to remove less polar interferences without eluting the targets. This step is key to achieving high selectivity [117].
- Elution: Apply 1-7 mL of a strong elution solvent to disrupt the analyte-sorbent bonds and recover the purified analytes. For the mixed-mode SCX sorbent, 7 mL of 5% NH₄OH in methanol was used. The basic ammonia solution neutralizes the cationic analytes, while the methanol elutes them from the reverse-phase part of the sorbent. Collect the eluate in a clean vial [117].
- Post-Processing and Analysis: The eluate is often evaporated to dryness under a gentle stream of nitrogen. The residue is then reconstituted in a solvent compatible with the subsequent analytical instrument (e.g., a mobile phase for LC-MS/MS) prior to injection and analysis [117].
The advantages of Solid Phase Extraction—high selectivity, ease of automation, and low solvent consumption—are not merely theoretical but are driving its widespread adoption as a superior sample preparation technology. The quantitative data and experimental evidence demonstrate that SPE effectively addresses critical challenges in modern laboratories, including the need for reproducible, high-throughput, and environmentally sustainable methods. As research continues, trends such as the development of novel sorbent materials, deeper integration with analytical instrumentation, and a focus on miniaturization will further entrench SPE's role as an indispensable tool in scientific early development and quality control across the pharmaceutical, environmental, and life sciences sectors.
Solid-Phase Extraction (SPE) represents a pivotal sample preparation technology that has shaped analytical chemistry since its mid-20th century origins. As a technique designed to isolate, purify, and concentrate analytes from complex matrices, SPE filled a critical need for more efficient alternatives to liquid-liquid extraction (LLE). The development of SPE was driven by the necessity to overcome several disadvantages inherent in LLE, including extensive organic solvent consumption, lengthy operation times, numerous procedural steps, potential emulsion formation, and high costs associated with solvent disposal [2].
The foundational principle of SPE involves the distribution of analytes between a liquid sample and a solid sorbent phase. As the liquid sample passes through adsorbent particles, analytes with greater affinity for the sorbent than the bulk liquid are retained. These captured analytes are subsequently released through elution with an appropriate solvent [2]. This straightforward mechanism belies the sophisticated development that has transformed SPE from its crude beginnings using animal charcoal for pigment removal to today's advanced sorbent technologies [2].
This technical guide examines the limitations and scope of SPE techniques, particularly within the context of early development research where understanding methodological constraints is essential for generating reliable analytical data. By critically evaluating both the historical constraints and ongoing challenges, we provide researchers with a framework for selecting appropriate sample preparation methodologies and recognizing situations where alternative techniques may offer superior solutions.
Historical Development and Technical Evolution
The genesis of SPE dates to the 1940s, with significant maturation occurring during the 1970s as the technique gained recognition within the scientific community [2]. The historical progression of SPE technology can be divided into three distinct developmental phases:
- 1968–1977: The initial phase characterized by early sorbent development and application exploration
- 1977–1989: Commercialization accelerated with the introduction of pre-filled cartridges/columns containing silica sorbents in October 1977
- 1989–present: Ongoing innovation including disk formats, monolithic polymers, and high-throughput systems [2]
A transformative advancement occurred in 1978 with the introduction of the Sep Pak C18 cartridge for cleaning histamine from wine, marking the first published application of silica-based SPE [2]. This innovation demonstrated the potential of stable, covalently bonded chromatographic adsorbents, particularly reversed-phase materials, which subsequently unlocked applications across environmental, clinical, and pharmaceutical markets.
The late 1980s witnessed another leap forward with the introduction of SPE disks or membranes, where absorbent material was embedded between Teflon or fiberglass pads or within a matrix structure [2]. This design created extremely short yet precise SPE cartridges with greater cross-sectional areas that enabled faster flow rates while ignoring extract filtration needs. The 1990s saw the incorporation of monolithic polymer stationary phases into SPE devices, beginning with poly(styrene-co-divinylbenzene) (PS-DVB) [2]. These monolithic sorbents delivered superior mass transfer characteristics through highly interconnected pores and excellent permeability, making them ideal for high-throughput SPE experiments.
A critical innovation in SPE automation emerged in the early 1990s with the development of the first 96-well SPE system at Pfizer's Sandwich UK laboratories [119]. This advancement was driven by the need to increase throughput for bioanalysis, particularly for HIV-AIDS drug development programs. The transition from individual 1- or 2-mL columns to 96-well plates fundamentally addressed sample preparation bottlenecks that left expensive mass spectrometry instrumentation idle while awaiting sample processing [119]. This format evolution enabled the analysis of entire clinical studies within a week rather than months, establishing 96-well SPE as the standard workhorse for pharmaceutical bioanalysis worldwide.
Table 1: Historical Evolution of SPE Formats and Their Impact
| Time Period | Primary SPE Format | Key Innovation | Impact on Analytical Science |
|---|---|---|---|
| Pre-1977 | Laboratory-packed columns | Basic sorbent materials | Established alternative to LLE |
| 1977-1989 | Pre-filled cartridges | Commercial silica-based sorbents | Standardized SPE procedures |
| 1989-1990s | Disks/membranes | Increased surface area | Higher flow rates, larger sample volumes |
| 1990s-present | 96-well plates | High-throughput automation | Dramatically increased sample processing capacity |
| 2000s-present | On-line SPE | Direct instrument integration | Reduced manual handling, improved reproducibility |
Fundamental Limitations of Solid-Phase Extraction
Despite its widespread adoption and continuous technological improvements, SPE exhibits several inherent limitations that researchers must consider during method development. These constraints become particularly relevant when working with novel compounds or complex matrices in early research phases.
Throughput and Automation Constraints
While 96-well formats addressed major throughput limitations, fundamental constraints persist. Traditional cartridge-based SPE exhibits partially small cross-sections with sluggish flow rates and significant void volumes [2]. These factors contribute to lengthy processing times, particularly when dealing with complex samples that may cause cartridge clogging. The plugging and channeling phenomena in conventional SPE cartridges remain problematic, especially with samples containing particulate matter or complex matrices [2].
The disposable nature of most commercial SPE systems creates ongoing consumable costs that become significant with large sample volumes [2]. While restricted access materials (RAM) and molecularly imprinted polymers (MIP) have been fabricated as reusable extraction cartridges, most mainstream applications continue to employ disposable formats due to concerns about carryover and performance consistency [2].
Technical and Operational Challenges
Several technical challenges persist across SPE platforms, including potential breakthrough volume issues, particularly with smaller sample formats [2]. The limited sorbent mass in miniaturized formats constrains the binding capacity, potentially leading to analyte loss during loading phases. This limitation becomes critical when working with samples containing high concentrations of target analytes or interfering compounds that compete for binding sites.
The requirement for careful conditioning steps in many SPE protocols introduces additional complexity and potential error sources [2]. Improper conditioning can lead to reduced extraction efficiency, variable recovery, and compromised data quality. While techniques like pipette-tip SPE (PT-SPME) eliminate conditioning requirements and operate with smaller elution volumes, they exhibit decreased breakthrough volumes and limited effectiveness for certain applications [2].
Economic and Environmental Considerations
The cost structure of SPE presents significant considerations for research planning. While the technique reduces solvent consumption compared to traditional LLE, the ongoing expense of cartridges, plates, or disks constitutes an appreciable operational cost [2]. This economic factor becomes particularly relevant in high-volume laboratories or extended research programs.
Environmental considerations have gained prominence in analytical chemistry, leading to critical evaluation of SPE's ecological footprint. Although SPE represents an improvement over many traditional extraction methods, it still generates significant plastic waste [2]. The predominantly disposable nature of SPE consumables creates sustainability concerns that researchers must balance against performance requirements.
Comparative Analysis of SPE Techniques and Configurations
SPE technology has evolved into multiple configurations, each with distinct advantages, limitations, and optimal application domains. Understanding these differences is crucial for selecting the most appropriate format for specific research needs.
Table 2: Performance Comparison of SPE Techniques and Configurations
| Parameter | SPE Cartridge | Pipette-Tip SPE | Disk Format | 96-Well SPE | Magnetic SPE |
|---|---|---|---|---|---|
| Sorbent Weight | 4–30 mg | 4–400 µg | 4–200 mg | 3–200 mg | Variable |
| Sample Volume | 500 µL–50 mL | 0.5–1 mL | 0.5–1 L | 0.65–2 mL | 1–100 mL |
| Flow Characteristics | Sluggish flow rates | Controlled by pipette action | Fast flow rates | Variable, amenable to automation | Mixing-dependent |
| Primary Applications | Wide variety of sample matrices | Biological samples | Substantial volume samples | High-throughput bioanalysis | Environmental, biological samples |
| Key Advantages | Easy lab assembly, wide applicability | Simplicity, no conditioning, small elution volume | Greater cross-sectional area, faster processing | High throughput, automation compatible | No centrifugation/filtration needed |
| Major Limitations | Plugging, channeling, costly at scale | Decreased breakthrough volume | Costly with many samples | Significant plastic waste | Limited commercial availability |
The selection of appropriate SPE format must consider specific application requirements. Cartridge-based SPE remains the most versatile format for diverse sample matrices, while pipette-tip SPE offers advantages for small-volume biological samples where conditioning steps are undesirable [2]. Disk formats excel with substantial sample volumes, providing greater cross-sectional areas that support faster flow rates [2]. The 96-well SPE format has become the industry standard for high-throughput bioanalysis, particularly within pharmaceutical development where rapid processing of clinical samples is essential [119].
Emerging techniques like magnetic SPE (MSPE) represent promising alternatives that eliminate the need for centrifugation or filtration steps [2]. In MSPE, magnetic sorbents are added to the sample solution, allowing target analytes to be extracted through application of an external magnetic field rather than passing the sample through a stationary phase. This approach circumvents many clogging and channeling issues associated with conventional SPE formats.
Scope and Application-Specific Limitations
The applicability of SPE varies significantly across different analytical domains, with technique-specific limitations emerging in particular research contexts.
Pharmaceutical and Bioanalysis Applications
SPE has become indispensable in pharmaceutical analysis and bioanalysis, particularly for sample clean-up and concentration prior to LC-MS analysis. The 96-well SPE format specifically addresses the throughput requirements of modern drug development, where rapid bioanalysis is essential for pharmacokinetic studies [119]. However, limitations persist for certain pharmaceutical compounds, particularly those with extreme physicochemical properties.
Highly polar pharmaceutical compounds may demonstrate poor retention on conventional reversed-phase sorbents, necessitating alternative retention mechanisms such as ion-exchange or mixed-mode phases [2]. Similarly, compounds with specific structural features may exhibit irreversible binding to certain sorbent materials, resulting in low recovery and challenging method development.
Environmental Analysis
Environmental applications frequently employ SPE for monitoring organic pollutants, pesticides, and pharmaceuticals in water samples [2]. Disk-based formats particularly excel in these applications where large sample volumes (up to 1 liter) require processing to achieve adequate detection limits for trace contaminants [2]. The recent European Urban Wastewater Treatment Directive (2024/3019), which mandates advanced "quaternary" treatment to remove pharmaceutical residues, underscores the importance of reliable SPE methods for environmental monitoring [107].
Despite its widespread use, SPE faces challenges with certain environmental contaminants. Matrix effects from complex environmental samples can significantly impact extraction efficiency and method accuracy [120]. The presence of humic acids, particulate matter, and competing contaminants can reduce analyte recovery and necessitate additional clean-up steps or method optimization.
Food, Beverage, and Natural Product Analysis
The analysis of natural compounds, foods, and beverages represents another significant application area for SPE [2]. These matrices often present unique challenges due to their complexity and the presence of interfering compounds such as pigments, sugars, and lipids. While SPE effectively addresses many of these challenges, limitations emerge when dealing with certain natural product classes.
Current research on plant secondary metabolites faces constraints including low detection sensitivity, incomplete spectral databases, and challenges in separating complex mixtures [121]. These limitations can be exacerbated by SPE techniques that may not provide sufficient selectivity or recovery for certain phytochemical classes. Structural elucidation remains labor-intensive, and the poor standardization of natural product extracts further complicates method development [121].
When to Consider Alternative Techniques
Despite its versatility, SPE is not universally applicable. Recognizing situations where alternative techniques may be preferable is essential for developing robust analytical methods. The following decision framework guides researchers in evaluating alternative sample preparation approaches.
Diagram 1: Technique Selection Decision Framework. This flowchart guides researchers in selecting appropriate sample preparation methods based on specific sample characteristics and analytical requirements.
Scenarios Favoring Alternative Approaches
Several specific scenarios warrant consideration of alternatives to SPE:
Highly volatile analytes: For volatile organic compounds, especially in headspace analysis, Solid-Phase Microextraction (SPME) often provides superior performance compared to SPE [2]. The open-bed configuration of many SPE formats makes them unsuitable for retaining volatile analytes, leading to potential losses during sample loading or processing.
Limited sample volumes: When sample volume is severely constrained (e.g., pediatric or neonatal clinical samples, rare biological specimens), pipette-tip SPE or SPME may offer advantages due to their minimal sample volume requirements [2].
Extremely polar compounds: While mixed-mode SPE sorbents address some polar compound challenges, certain highly polar analytes may still demonstrate poor retention. In such cases, techniques like HILIC (Hydrophilic Interaction Liquid Chromatography) with minimal sample preparation or traditional LLE with appropriate solvent systems may be preferable.
High-throughput demands for simple matrices: For less complex matrices where extensive clean-up is unnecessary, protein precipitation (PPT) in 96-well format may provide adequate sample preparation with significantly higher throughput than SPE [119].
Green chemistry priorities: When environmental impact minimization is a primary concern, techniques like QuEChERS (Quick, Easy, Cheap, Effective, Rugged, Safe) may offer reduced solvent consumption and plastic waste compared to conventional SPE [107].
Emerging Alternatives and Complementary Techniques
Recent technological advances have yielded several promising alternatives that address specific SPE limitations:
Magnetic Solid-Phase Extraction (MSPE): This technique utilizes magnetic nanoparticles as sorbents, which can be easily separated from the sample matrix by applying an external magnetic field [107]. MSPE eliminates the need for centrifugation or filtration steps, reducing processing time and potential analyte loss.
Molecularly Imprinted Polymers (MIPs): These synthetic materials offer highly selective recognition sites complementary to target molecules [107]. MIPs provide superior selectivity for challenging separations, particularly when dealing with structurally similar compounds or complex matrices.
On-line SPE-LC/MS systems: These integrated approaches automate the transfer of extracted analytes from SPE to analytical instrumentation, reducing manual handling and improving reproducibility [107]. On-line systems offer particular advantages for high-throughput laboratories processing large sample batches.
Microextraction techniques: Methods such as SPME and in-tube SPME (IT-SPME) represent miniaturized alternatives that significantly reduce solvent consumption while maintaining excellent sensitivity [2]. These techniques are particularly valuable when sample volume is limited or when green chemistry principles are prioritized.
Experimental Protocols and Method Development Considerations
Successful implementation of SPE methods requires careful protocol development and optimization. The following section outlines key methodological considerations and provides representative experimental frameworks.
Representative SPE Protocol for Pharmaceutical Analysis
This standardized protocol for pharmaceutical compounds in biological matrices illustrates key steps in SPE method execution:
Sorbent Selection: Choose appropriate sorbent chemistry based on analyte properties. Reverse-phase C18 for non-polar compounds; mixed-mode for compounds with ionizable groups; HILIC for polar compounds.
Conditioning: Sequentially pass 2 column volumes of methanol and 2 column volumes of water or buffer through the sorbent bed. Do not allow the bed to dry completely after conditioning.
Sample Loading: Adjust sample pH to suppress ionization for target analytes (typically pH 2 units away from pKa for acidic/basic compounds). Load sample at controlled flow rate (1-5 mL/min for cartridges; 0.1-0.5 mL/min for 96-well plates).
Washing: Pass 2-3 column volumes of wash solution (typically 5-20% organic in water or buffer) to remove interfering compounds while retaining analytes.
Elution: Apply 1-2 column volumes of elution solvent (typically high organic content with modifiers if needed). Collect eluate for analysis.
Reconstitution: If necessary, evaporate and reconstitute eluate in mobile phase compatible solvent.
Table 3: Research Reagent Solutions for SPE Method Development
| Reagent Category | Specific Examples | Primary Function | Application Notes |
|---|---|---|---|
| Reverse Phase Sorbents | C18, C8, C4, phenyl | Retention of non-polar compounds | Work best for compounds with log P > 0 |
| Normal Phase Sorbents | Silica, alumina, florisil | Retention of polar compounds | Suitable for hydrophilic compounds |
| Ion Exchange Sorbents | SCX, SAX, WCX, WAX | Retention of ionizable compounds | pH control critical for performance |
| Mixed-Mode Sorbents | C18/SCX, C8/SAX | Combined hydrophobic/ion-exchange retention | Ideal for basic/acidic drugs |
| Polymeric Sorbents | PS-DVB, Oasis HLB | Hydrophilic-lipophilic balance | Broad-spectrum retention |
| Specialty Sorbents | RAM, MIP, immunosorbents | Selective recognition | Improved selectivity for complex matrices |
Troubleshooting Common SPE Method Issues
Method development frequently encounters challenges that require systematic troubleshooting:
Low Recovery: Potential causes include insufficient sorbent capacity, incomplete elution, analyte breakthrough, or strong secondary interactions. Solutions involve increasing sorbent mass, optimizing elution solvent strength/composition, reducing sample load, or adding modifiers to disrupt secondary interactions.
Poor Reproducibility: Often results from inconsistent flow rates, variable conditioning, or sorbent lot variations. Implementing controlled flow rates, standardized conditioning protocols, and testing new sorbent lots can improve consistency.
Matrix Effects: Caused by co-eluting compounds that interfere with ionization or detection. Solutions include optimizing wash steps, changing sorbent chemistry, or implementing additional clean-up procedures.
Clogging: Common with dirty samples. Remedies include sample filtration, centrifugation, dilution, or switching to disk formats with larger surface areas.
Solid-Phase Extraction remains a cornerstone technique in modern analytical chemistry, offering significant advantages over traditional extraction methods. Its continuous evolution—from basic cartridges to high-throughput 96-well plates and selective MIPs—demonstrates the technique's adaptability to changing analytical demands. Nevertheless, understanding SPE's limitations and scope is essential for appropriate technique selection and robust method development.
The future trajectory of SPE technology points toward increased miniaturization, automation, and selectivity. Emerging trends include the development of eco-friendly workflows, automated time-efficient techniques, increasingly selective sorbents, and robust analytical methods [107]. The integration of SPE with analytical instrumentation through on-line systems will continue to reduce manual intervention and improve reproducibility.
For researchers in early development stages, a critical assessment of SPE applicability should consider the specific analytical challenges presented by their unique samples and analytes. While SPE provides an excellent solution for many sample preparation needs, recognizing scenarios where alternative techniques offer superior performance is fundamental to generating reliable, reproducible analytical data. Through thoughtful technique selection and method optimization that acknowledges both capabilities and constraints, researchers can leverage SPE most effectively within the broader analytical toolkit.
Conclusion
The historical development of solid-phase extraction represents a pivotal shift in analytical science, evolving from a simple concept using active carbon to a sophisticated, foundational tool in modern laboratories. The key takeaways from its journey include the critical importance of sorbent chemistry, the value of standardized formats for reproducibility, and the necessity of optimized protocols for challenging matrices. The foundational work established the principles that continue to drive methodological innovations today. For the future of biomedical and clinical research, the implications are profound. The ongoing development of highly selective sorbents, automation-friendly configurations, and miniaturized formats like pipette-tip SPE will be crucial for advancing high-throughput proteomics, metabolomics, and therapeutic drug monitoring. As research demands greater sensitivity and faster analysis of complex biological samples, the principles of SPE will continue to underpin the sample preparation strategies that enable groundbreaking discoveries.
References
- 1. Historic journey of activated carbon and breakthroughs [https://www.camfil.com/en-us/insights/innovation-technology-and-research/journey-of-activated-carbon?srsltid=AfmBOopktgv11Kc_6JuR2hFVNlvgVat_7-IOpD2tVZ_F-BvoLzhMZfQI]
- 2. A review of the modern principles and applications of solid ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9659506/]
- 3. Solid-Phase Extraction [https://chem.libretexts.org/Bookshelves/Analytical_Chemistry/Supplemental_Modules_(Analytical_Chemistry)/Analytical_Sciences_Digital_Library/Contextual_Modules/Sample_Preparation/03_Solid-Phase_Extraction]
- 4. Activated Carbon [https://www.calgoncarbon.com/activated-carbon/]
- 5. Vehicle Emissions and Air Quality: The Early Years (1940s ... [https://www.mdpi.com/2073-4433/12/10/1354]
- 6. Solid Phase Extraction Guide [https://www.thermofisher.com/us/en/home/industrial/chromatography/chromatography-sample-preparation/sample-preparation-consumables/solid-phase-extraction-consumables/spe-guide.html]
- 7. History of chromatography [https://en.wikipedia.org/wiki/History_of_chromatography]
- 8. The People Who Shaped Chromatography – Mikhail Tsvet [https://phenomenex.blog/2020/09/29/mikhail-tsvet/]
- 9. What is Chromatography? [https://concentratingonchromatography.com/what-is-chromatography]
- 10. The Evolution of Gas Chromatography and later GC-MS ... [https://emerypharma.com/blog/the-evolution-of-gas-chromatography-and-later-gc-ms-and-lc-ms-one-mans-journey-from-student-to-ceo/]
- 11. R.L.M. Synge | Biochemist, Nobel Prize, Chromatography [https://www.britannica.com/biography/R-L-M-Synge]
- 12. Technique in Chromatography - an overview [https://www.sciencedirect.com/topics/biochemistry-genetics-and-molecular-biology/technique-in-chromatography]
- 13. What is Solid Phase Extraction (SPE)? [https://www.organomation.com/what-is-solid-phase-extraction-spe]
- 14. Introduction to Solid Phase Extraction Technology - Hawach [https://www.specartridge.com/introduction-to-solid-phase-extraction-technology/]
- 15. Origins of SPE Technology [https://poseidon-scientific.com/blog/spe/origins-of-spe-technology/]
- 16. Solid-Phase Extraction - an overview | ScienceDirect Topics [https://www.sciencedirect.com/topics/chemistry/solid-phase-extraction]
- 17. Development of a Solid-Phase Extraction (SPE) Cartridge ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6189673/]
- 18. Review Fifty years of solid-phase extraction in water analysis [https://www.sciencedirect.com/science/article/abs/pii/S0021967399011449]
- 19. The Origins of Sample Preparation Technologies [https://www.chromatographyonline.com/view/the-origins-of-sample-preparation-technologies]
- 20. Validation of new solid-phase extraction materials for the ... [https://www.sciencedirect.com/science/article/abs/pii/S0165993698001083]
- 21. The Fundamentals of Solid Phase Extraction (SPE) [https://www.restek.com/articles/the-fundamentals-of-solid-phase-extraction-SPE?srsltid=AfmBOoq8Dw-_FE3enIWk7b48kVQuNkUfdzI10kxZhu_O8VKUiLxBPQRr]
- 22. Solid phase microextraction for quantitative analysis [https://www.sciencedirect.com/science/article/pii/S2772577422000465]
- 23. Comparison of Two Solid-Phase Extraction (SPE) Methods ... [https://pubmed.ncbi.nlm.nih.gov/30513899/]
- 24. Advantages of Solid Phase Extraction - Hawach - SPE Cartridge [https://www.specartridge.com/advantages-of-solid-phase-extraction/]
- 25. Solid Phase Extraction vs Liquid-Liquid ... [https://www.labmanager.com/solid-phase-extraction-vs-liquid-liquid-extraction-vs-filtration-which-extraction-method-is-more-efficient-33687]
- 26. Solid Phase Extraction Guide [https://www.thermofisher.com/tr/en/home/industrial/chromatography/chromatography-sample-preparation/sample-preparation-consumables/solid-phase-extraction-consumables/spe-guide.html]
- 27. Liquid-Liquid Extraction vs. Solid-Phase Extraction Methods [https://kjhil.com/liquid-liquid-extraction-vs-solid-phase-extraction/]
- 28. Current state and future perspectives of supercritical fluid ... [https://www.sciencedirect.com/science/article/abs/pii/S0165993622000334]
- 29. Supercritical Fluids Extraction: Benefits and Applications [https://extraktlab.com/supercritical-fluids-extraction/]
- 30. New Trends in Supercritical Fluid Technology and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10254675/]
- 31. Advances and trends in compressed fluid extraction to ... [https://www.sciencedirect.com/science/article/abs/pii/S0896844624003401]
- 32. When should I choose a mixed-mode SPE? [https://www.biotage.com/blog/when-should-i-choose-a-mixed-mode-spe]
- 33. Chemometric Optimization of SPE for the Determination of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10924705/]
- 34. Application of a homemade silica-based mixed-mode ion ... [https://www.sciencedirect.com/science/article/pii/S2772582023000244]
- 35. The Technology And Application Of SPE - Hawach [https://www.specartridge.com/the-technology-and-application-of-spe/]
- 36. Evaluating the reliability of solid phase extraction ... [https://www.sciencedirect.com/science/article/abs/pii/S0021967324008094]
- 37. Advanced Topics in Solid-Phase Extraction: Chemistries [https://www.chromatographyonline.com/view/advanced-topics-solid-phase-extraction-chemistries-0]
- 38. Solid-Phase Extraction: From Sample Prep Fundamentals ... [https://www.waters.com/blog/solid-phase-extraction-from-sample-prep-fundamentals-to-best-practices/?srsltid=AfmBOoqeWtkjDDxBcBTTZZeWrbt9XonDztxb7Q6RqqdyY__07O5pCc_P]
- 39. Solid- phase extraction - Wikipedia [https://en.wikipedia.org/wiki/Solid-phase_extraction]
- 40. Do You Really Know About SPE - Hawach Normal Phase [https://www.specartridge.com/do-you-really-know-about-spe-normal-phase-and-reverse-phase-extraction/]
- 41. Understanding SPE Retention Mechanisms [https://www.biotage.com/blog/understanding-spe-retention-mechanisms]
- 42. Solid Phase Extraction: Ion Exchange Methodology [https://www.merckmillipore.com/GA/en/technical-documents/technical-article/analytical-chemistry/solid-phase-extraction/ion-exchange-methodology-solid-phase-extraction]
- 43. How It Works: Ion-Exchange SPE [https://www.chromatographyonline.com/view/how-it-works-ion-exchange-spe]
- 44. Reshaping of pipette tip: A facile and practical strategy for ... [https://www.sciencedirect.com/science/article/abs/pii/S0003267019314230]
- 45. Solid Phase Extraction Cartridges and Disks Selection Guide: Types, Features, Applications | GlobalSpec [https://www.globalspec.com/learnmore/labware_scientific_instruments/sample_preparation_wet_chemical_analysis/solid_phase_extraction_cartridges_discs]
- 46. Recent advances in accelerating solid-phase extraction [https://www.sciencedirect.com/science/article/pii/S2772391724000434]
- 47. A Self‐Assembly Pipette Tip Restricted Access ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12039168/]
- 48. Comparison of Two Solid-Phase Extraction (SPE) Methods for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6321002/]
- 49. Fifty years of solid - phase in water analysis-- extraction ... historical [https://pubmed.ncbi.nlm.nih.gov/10941664/]
- 50. Application of Solid in Environmental Sample... Phase Extraction [https://www.raykolgroup.com/application-of-solid-phase-extraction-in-environmental-sample-analysis.html]
- 51. High Flow-Rate Sample Loading in Large Volume Whole ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6479934/]
- 52. HPLC 2025 Preview: Functionalized Monoliths as ... [https://www.chromatographyonline.com/view/functionalized-monoliths-as-selective-sample-preparation-materials]
- 53. Monolithic vs. particle-based solid-phase extraction for ... [https://pubs.rsc.org/en/content/articlehtml/2025/va/d5va00057b]
- 54. A perspective on developing solid-phase extraction ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8979348/]
- 55. Functionalized monolithic columns - PubMed Central - NIH [https://pmc.ncbi.nlm.nih.gov/articles/PMC9388943/]
- 56. Versatile sorbent materials in sample preparation [https://www.sciencedirect.com/science/article/pii/S2772582025000634]
- 57. Analytical Methods for the Determination of ... [https://www.mdpi.com/1420-3049/29/16/3900]
- 58. Quantitative mass spectrometry methods for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5031633/]
- 59. Comparative Study of (Q)SAR Models for Predicting the ... [https://www.sciencedirect.com/science/article/pii/S3050620425000491]
- 60. Three Most Common Problems Regard to Solid Phase ... [https://www.hawach.com/news/three-most-common-problems-regard-to-spe.html]
- 61. Solid Phase Extraction (SPE) Method Development Guide [https://www.jk-sci.com/blogs/lab-tips/solid-phase-extraction-spe-method-development-guide?srsltid=AfmBOopoI_n-Lndf8BNFtdpBz2Qma9f0pMytbzbX-lyaaMNfToifp5dm]
- 62. Understanding and Improving Solid-Phase Extraction [https://www.chromatographyonline.com/view/understanding-and-improving-solid-phase-extraction-1]
- 63. Comparison of Solid-Phase Extraction Sorbents for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9858392/]
- 64. HPLC 2025 Preview: The Road To Sustainable Analytical ... [https://www.chromatographyonline.com/view/hplc-2025-preview-the-road-to-sustainable-analytical-chemistry]
- 65. Miniaturized solid-phase extraction techniques in sample ... [https://www.sciencedirect.com/science/article/pii/S0026265X25011488]
- 66. Guide To Solid Phase Extraction (SPE): Step-by- ... [https://www.alwsci.com/news/guide-to-solid-phase-extraction-spe-step-by-85168348.html]
- 67. Highly Reproducible Solid Phase Extraction [https://www.labmanager.com/highly-reproducible-solid-phase-extraction-1-23066]
- 68. Early Development GMPs for Small-Molecule Specifications [https://www.pharmtech.com/view/early-development-gmps-small-molecule-specifications-industry-perspective-part-v]
- 69. Compensate for or Minimize Matrix Effects? Strategies ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7412464/]
- 70. Optimizing LC‑MS/MS Sensitivity: Strategies to Overcome ... [https://biotech-spain.com/en/articles/optimizing-lc-ms-ms-sensitivity-strategies-to-overcome-ion-suppression-and-boost-robustness-in-bioanalysis/]
- 71. Advanced Topics in Solid-Phase Extraction: Chemistries [https://www.chromatographyonline.com/view/advanced-topics-solid-phase-extraction-chemistries-1]
- 72. Critical Assessment of Clean-Up Techniques Employed in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8781265/]
- 73. Comparison of efficiency of different sorbents used during ... [https://www.sciencedirect.com/science/article/abs/pii/S0963996909003603]
- 74. Tip on recoveries greater than 100% [https://discover.phenomenex.com/greater-recoveries-spe-technical-tip-en-0325]
- 75. The Fundamentals of Solid Phase Extraction (SPE) [https://www.restek.com/articles/the-fundamentals-of-solid-phase-extraction-SPE?srsltid=AfmBOoq6p2-cmBOPrEiSM2tRxATPwmWpKgsWSkiggQTnxDQTFa5aYHqb]
- 76. Cross-Signal Contribution as a Challenge in LC-MS ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12268833/]
- 77. Chemometric cleanup to eliminate ionic liquid ... [https://www.sciencedirect.com/science/article/abs/pii/S0167732221003524]
- 78. SPE - Optimize Your Solid Phase Extraction-Method - Wiki [https://community.agilent.com/technical/consumables/w/wiki/21631/spe---optimize-your-solid-phase-extraction-method]
- 79. The Fundamentals of Solid Phase Extraction (SPE) [https://www.restek.com/articles/the-fundamentals-of-solid-phase-extraction-SPE?srsltid=AfmBOorCAem5_QAKtGNttKHV5T_r9Wp9-ng4fUPZ-17S9kSD4d0ESOg8]
- 80. Use of response surface methodology to optimize triclosan ... [https://www.sciencedirect.com/science/article/pii/S0169131723000662]
- 81. A Simplified Approach to Optimize SPE Method ... [https://www.spectroscopyonline.com/view/simplified-approach-optimize-spe-method-development-downstream-lc-ms-analysis-allowing-100-organic-0]
- 82. Optimising factors affecting solid phase extraction performances of molecular imprinted polymer as recent sample preparation technique - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC7848654/]
- 83. Development and optimization of SPE-HPLC-UV/ELSD for ... [https://pubmed.ncbi.nlm.nih.gov/26825340/]
- 84. Solid Phase Extraction (SPE) Market Competitive Analysis ... [https://introspectivemarketresearch.com/reports/solid-phase-extraction-spe-market/]
- 85. The Fundamentals of Solid Phase Extraction (SPE) [https://www.restek.com/articles/the-fundamentals-of-solid-phase-extraction-SPE?srsltid=AfmBOopmljjHomy4NaJ1cxRAGoF8igCqzCoEj80dhKZiNGbGpUcaHUNm]
- 86. Optimization of solid phase extraction for simultaneous ... [https://www.frontiersin.org/journals/environmental-science/articles/10.3389/fenvs.2025.1539932/full]
- 87. Solid-Phase Extraction: From Sample Prep Fundamentals ... [https://www.waters.com/blog/solid-phase-extraction-from-sample-prep-fundamentals-to-best-practices/?srsltid=AfmBOoqHlqBdOq5UWCxNyP4DCvi_0fl2ISAabPof3sHeBpfKFSz8bsLo]
- 88. The solvent-assisted dispersive solid-phase extraction ... [https://www.sciencedirect.com/science/article/pii/S2772582025000154]
- 89. The Ultimate SPE Guide [https://www.numberanalytics.com/blog/ultimate-guide-to-solid-phase-extraction]
- 90. Integrated Solid - Phase , Ultra- Extraction - High ... Performance Liquid [https://pmc.ncbi.nlm.nih.gov/articles/PMC9696973/]
- 91. Sample Preparation of Drug Substances and Products in ... [https://www.chromatographyonline.com/view/sample-preparation-of-drug-substances-and-products-in-regulated-testing-a-primer]
- 92. PMC - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC12475962/]
- 93. An improved and validated solid-phase extraction ... [https://www.sciencedirect.com/science/article/pii/S0026265X25028103]
- 94. (PDF) Solid - Phase Combined with Extraction ... High Performance [https://scispace.com/papers/solid-phase-extraction-combined-with-high-performance-liquid-432d83hl6e]
- 95. The application of solid - phase and reversed extraction ... phase [https://link.springer.com/article/10.1007/BF02492087]
- 96. SPE Vs. LLE: Choosing The Right Extraction Technique ... [https://www.alwsci.com/news/spe-vs-lle-choosing-the-right-extraction-tec-85163402.html]
- 97. 96-Well solid-phase extraction: a brief history of its ... [https://pubmed.ncbi.nlm.nih.gov/15680790/]
- 98. SPE, LLE, and SLE - SCIENCE UNFILTERED [https://phenomenex.blog/2022/02/22/spe-vs-lle-vs-sle/]
- 99. A quantitative study for liquid-liquid extraction [https://www.sciencedirect.com/science/article/abs/pii/S0043135424012697]
- 100. Green Solvents for Liquid–Liquid Extraction [https://www.mdpi.com/2673-4591/56/1/174]
- 101. Comparison of Two Solid-Phase Extraction (SPE) Methods ... [https://www.mdpi.com/1422-0067/19/12/3847]
- 102. 2025 Proteomics Breakthroughs [https://www.genengnews.com/topics/drug-discovery/2025-proteomics-breakthroughs/]
- 103. Protocol for dual metabolomics and proteomics using ... [https://www.sciencedirect.com/science/article/pii/S2666166725001510]
- 104. The Fundamentals of Solid Phase Extraction (SPE) [https://www.restek.com/articles/the-fundamentals-of-solid-phase-extraction-SPE?srsltid=AfmBOoqL_GGId97eeipdx-8T80W6bs38SlbPAqAd2LxS29B_SbXnq0Di]
- 105. Recovery Rate, Extraction Speed of SPE Cartridge - Hawach [https://www.specartridge.com/recovery-rate-extraction-speed-and-sample-capacity-of-spe-cartridge/]
- 106. Solid-Phase Extraction: From Sample Prep Fundamentals ... [https://www.waters.com/blog/solid-phase-extraction-from-sample-prep-fundamentals-to-best-practices/?srsltid=AfmBOoocCWcZTey8KarYTb9CCKSaLQSdgM3VaP9wfj1usDF8p1_UdL_2]
- 107. Recent Trends in Solid-Phase Microextraction for the ... [https://www.mdpi.com/2297-8739/12/9/256]
- 108. EPA Method 3535A (SW-846): Solid-Phase Extraction (SPE) [https://www.epa.gov/esam/epa-method-3535a-sw-846-solid-phase-extraction-spe]
- 109. SW-846 Test Method 3535A: Solid-Phase Extraction (SPE) [https://www.epa.gov/hw-sw846/sw-846-test-method-3535a-solid-phase-extraction-spe]
- 110. An Overview of Solid-Phase Extraction [https://welchlab.com/blogs/news/an-overview-of-solid-phase-extraction?srsltid=AfmBOooj4qXTZlvZhca_TVnhUbh4l59RTPsyn6ja1s9WffxSOLj5Fz2T]
- 111. Comparison of conventional and dispersive solid phase ... [https://www.sciencedirect.com/science/article/pii/S0039914022009882]
- 112. Solid Phase Extraction (SPE) System Analysis Report 2025 [https://www.archivemarketresearch.com/reports/solid-phase-extraction-spe-system-220248]
- 113. Solid Phase Extraction SPE Market Report [https://dataintelo.com/report/global-solid-phase-extraction-spe-market]
- 114. Solid Phase Extraction (SPE) System Market 2025-2032 [https://marketresearchcommunity.com/europe-solid-phase-extraction-spe-system-market/]
- 115. Solid Phase Extraction (SPE) System Market [https://www.linkedin.com/pulse/solid-phase-extraction-spe-system-market-trends-outlook-cydkf/]
- 116. Solvent Extraction Techniques [https://www.organomation.com/solvent-extraction-techniques]
- 117. A comparative study of SPE- and DLLME-based methods ... [https://www.sciencedirect.com/science/article/pii/S0026265X24024676]
- 118. New HPLC, MS, and CDS Products from 2024–2025 [https://www.chromatographyonline.com/view/new-hplc-ms-and-cds-products-from-2024-2025-a-brief-review]
- 119. Review 96-Well solid-phase extraction: a brief history of its ... [https://www.sciencedirect.com/science/article/abs/pii/S1570023204006737]
- 120. Limitations and disadvantages of GC-MS [https://labioscientific.com/limitations-and-disadvantages-of-gc-ms/]
- 121. A comprehensive review-current development in ... [https://www.sciencedirect.com/science/article/pii/S2211715625003248]