Research Articles
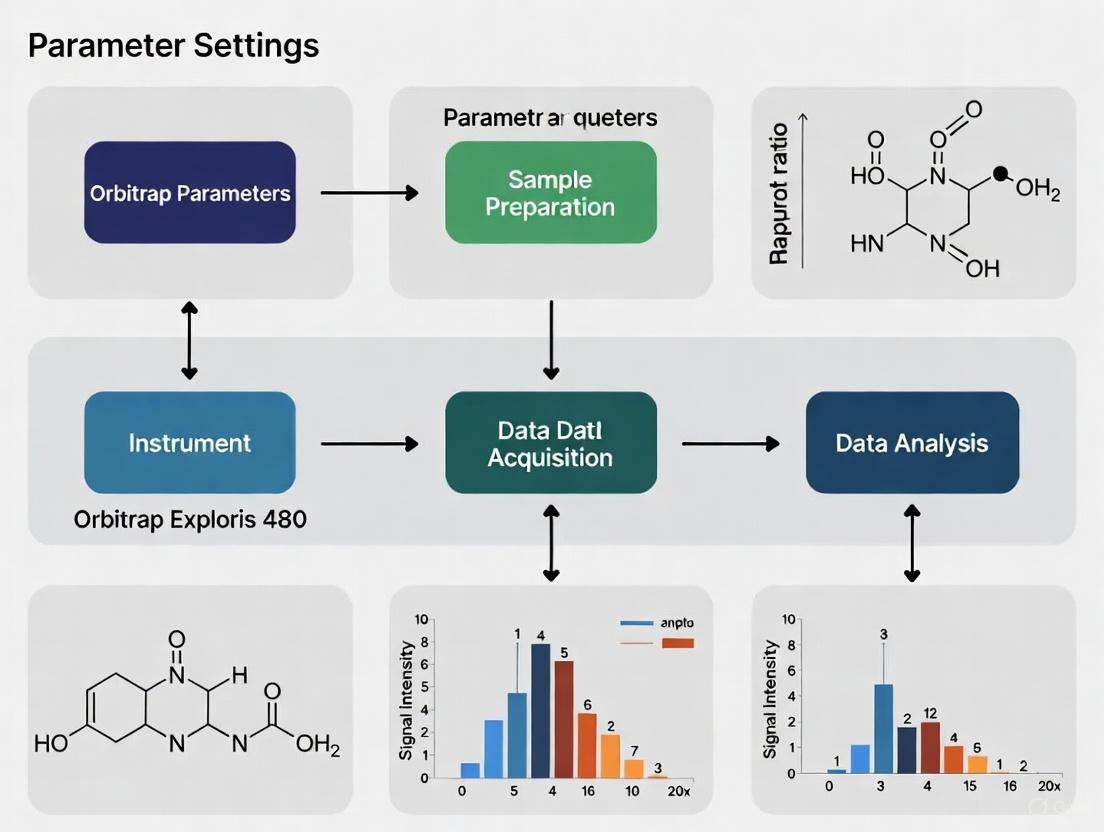
Optimizing Orbitrap Exploris 480 Parameter Settings for Robust Metabolomics: A Foundational Guide from Setup to Advanced Applications
This article provides a comprehensive guide for researchers and drug development professionals on configuring and optimizing the Thermo Scientific Orbitrap Exploris 480 mass spectrometer for metabolomics studies.
Preserving the Past: Advanced Strategies for Minimizing Sample Destruction in Cultural Heritage Analysis
This article provides a comprehensive guide for researchers and scientists on implementing non-destructive and minimally invasive analytical techniques in cultural heritage studies.
From Pixels to Chemistry: A Comprehensive Guide to Hyperspectral Imaging for Material Mapping
This article provides a comprehensive exploration of hyperspectral imaging (HSI) as a powerful, non-destructive tool for chemical mapping of materials.
The Evolution of Solid-Phase Extraction: From Historical Foundations to Modern Biomedical Applications
This article provides a comprehensive exploration of the early development and evolution of solid-phase extraction (SPE) techniques, tailored for researchers, scientists, and drug development professionals.
Portable XRF for In-Situ Environmental Monitoring: A Comprehensive Guide for Researchers and Scientists
This article provides a thorough examination of portable X-ray fluorescence (XRF) technology for in-situ environmental assessment.
From the Lab to the Artifact: The Pioneering Role of Analytical Chemistry in Cultural Heritage Science
This article explores the foundational and evolving role of analytical chemistry in the study and preservation of cultural heritage.
Limit of Detection (LOD) Determination: A Comprehensive Guide for Researchers and Scientists
This article provides a complete guide to Limit of Detection (LOD) determination for researchers, scientists, and drug development professionals.
Multi-Objective Optimization in Analytical Chemistry: Accelerating Drug Discovery and Materials Development
This article provides a comprehensive overview of multi-objective optimization (MOO) methodologies and their transformative impact on analytical chemistry, with a focus on drug discovery and materials development.
2025 Roadmap to Superior Analysis: Boosting Precision and Accuracy in Biomedical Research
This article provides a comprehensive guide for researchers and drug development professionals seeking to enhance analytical precision and accuracy in 2025.
Expert HPLC Troubleshooting Guide: From Diagnosis to Validation for Scientists
This comprehensive guide provides researchers and drug development professionals with a systematic approach to diagnosing, solving, and preventing High-Performance Liquid Chromatography (HPLC) issues.