Research Articles
Analytical Method Robustness Testing: A 2025 Guide for Reliable and Compliant Results
This article provides a comprehensive guide to analytical method robustness testing for researchers, scientists, and drug development professionals.
Essential Career Skills for Analytical Chemistry Researchers: A 2025 Guide to Mastery from Foundations to AI
This guide provides a comprehensive roadmap for analytical chemistry researchers and drug development professionals to master the essential skills demanded by the modern laboratory.
How to List Chromatography Skills on Your Resume: A Complete Guide for Scientists
This guide provides researchers, scientists, and drug development professionals with a comprehensive framework for effectively showcasing chromatography skills on a resume.
Good Laboratory Practice (GLP) for Analytical Chemists: A Practical Guide to Compliance, Methods, and Data Integrity
This article provides a comprehensive guide to Good Laboratory Practice (GLP) specifically for analytical and bioanalytical chemists involved in drug development.
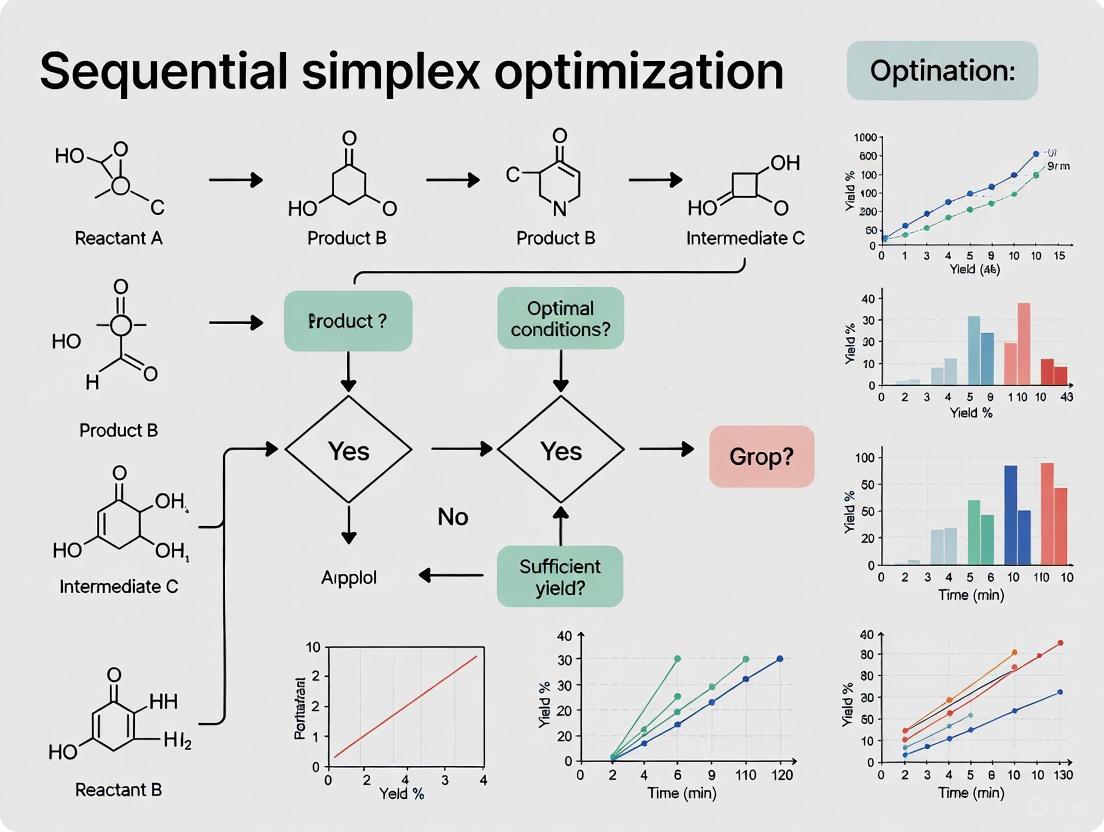
Sequential Simplex Optimization: A Practical Guide for Multi-Factor Optimization in Drug Development
This article provides a comprehensive guide to Sequential Simplex Optimization, a powerful multivariate chemometric tool for efficiently optimizing multiple factors in complex systems.
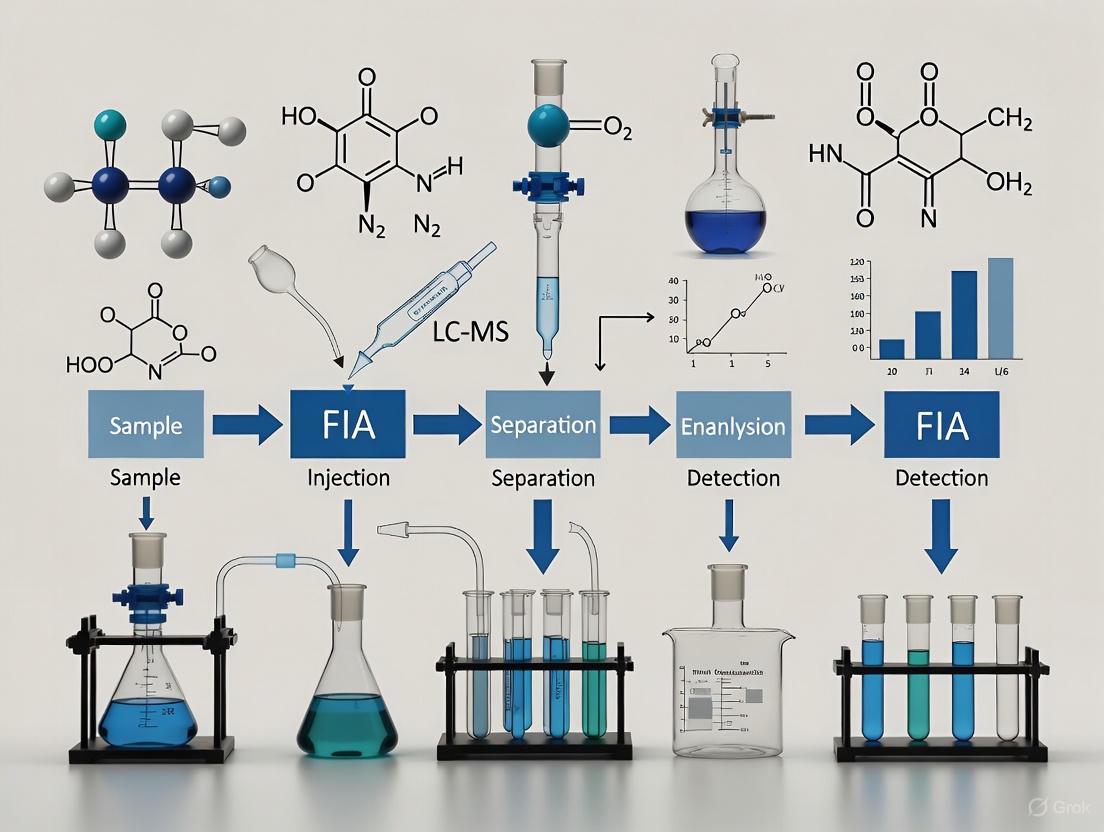
Flow Injection Analysis LC-MS Optimization: A Complete Guide for Robust Method Development
This article provides a comprehensive guide to optimizing Flow Injection Analysis (FIA) for Liquid Chromatography-Mass Spectrometry (LC-MS).
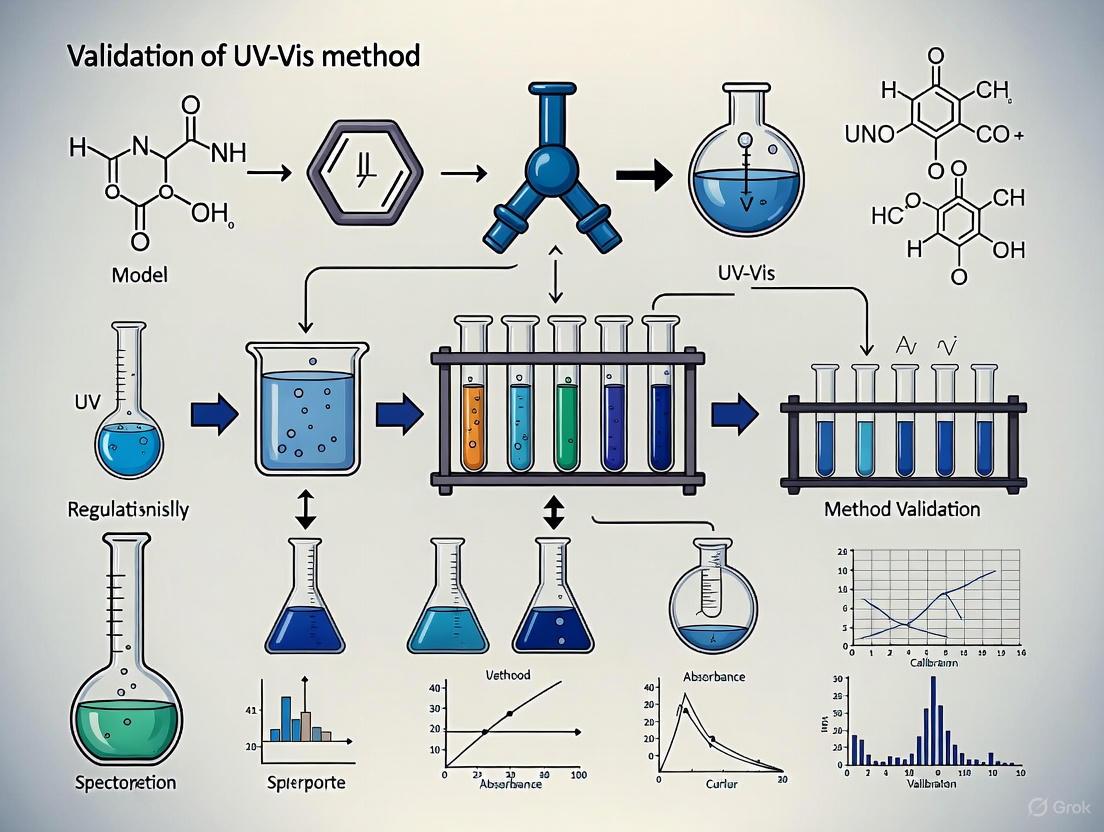
UV-Vis Method Validation for Regulatory Compliance: A Practical Guide for Pharmaceutical QC
This article provides a comprehensive framework for developing and validating UV-Vis spectrophotometric methods to meet stringent regulatory standards in pharmaceutical quality control.
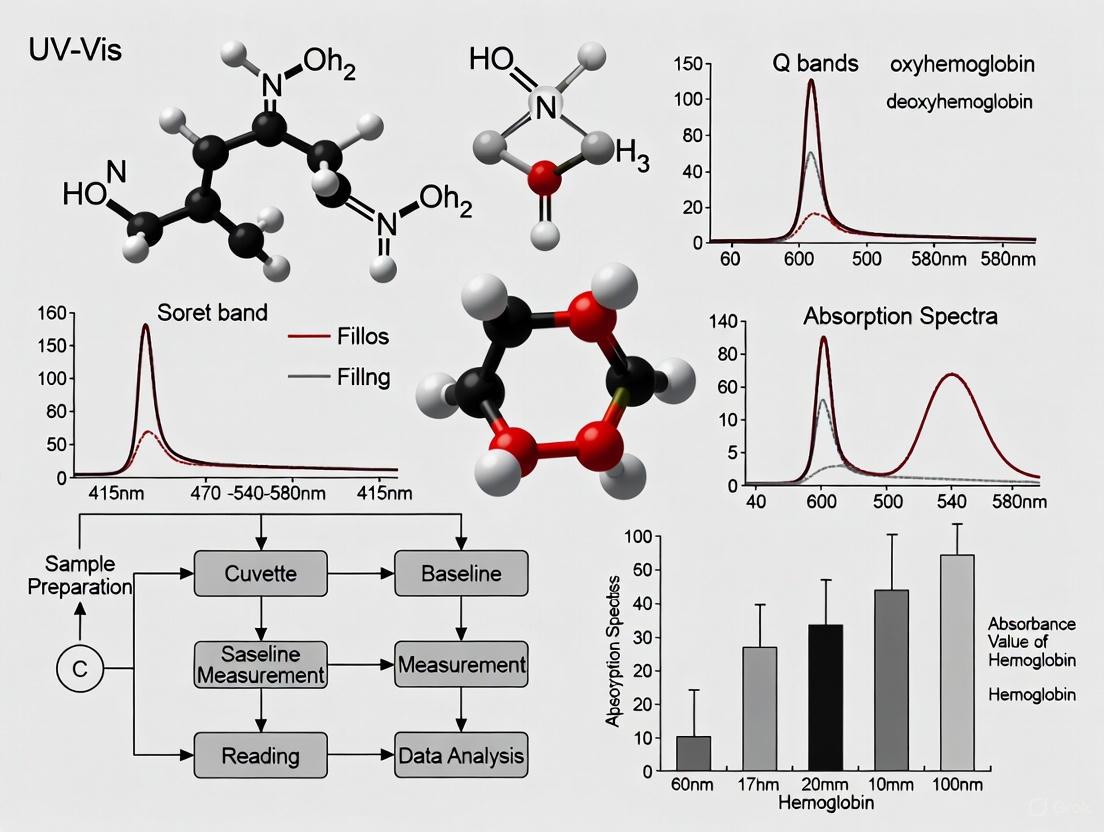
A Comprehensive Guide to UV-Vis Spectroscopy for Accurate Hemoglobin Quantification
This article provides a complete protocol for researchers and drug development professionals seeking to accurately quantify hemoglobin using UV-Vis spectroscopy.
UV-Vis Spectroscopy vs. HPLC: A Strategic Guide for Quantitative Analysis of Active Components
This article provides a comprehensive comparison of Ultraviolet-Visible (UV-Vis) spectroscopy and High-Performance Liquid Chromatography (HPLC) for the quantification of active components in pharmaceuticals and cosmetics.
Cuvette vs. Cuvette-Free UV-Vis Systems: A Complete Guide for Small-Volume Sample Analysis
This article provides researchers, scientists, and drug development professionals with a comprehensive analysis of cuvette-based and cuvette-free UV-Vis systems for small sample volumes.