Research Articles
Strategies to Lower LOD and LOQ: A Comprehensive Guide for Enhanced Trace Analysis in Biomedical Research
This article provides a comprehensive guide for researchers and drug development professionals seeking to improve the sensitivity and reliability of their analytical methods.
Assay vs. Impurity Method Validation: A Guide to ICH Q2(R2) Compliance and Lifecycle Management
This article provides a comprehensive guide for researchers and drug development professionals on the distinct validation requirements for assay and impurity methods.
Establishing Robust System Suitability Criteria for HPLC Methods: A Lifecycle Guide for Regulatory Compliance and Data Integrity
This article provides a comprehensive framework for researchers, scientists, and drug development professionals to establish, implement, and troubleshoot system suitability criteria for HPLC methods.
UV-Vis vs. UFLC-DAD: A Strategic Guide to Speed, Sensitivity, and Application in Pharmaceutical Analysis
This article provides a comprehensive comparison of UV-Vis spectroscopy and UFLC-DAD for pharmaceutical analysis, directly addressing the critical factor of analysis time.
UV-Vis vs. UFLC-DAD: A Strategic Cost-Effectiveness Analysis for Modern Laboratories
This article provides a comprehensive cost-effectiveness analysis of UV-Vis spectrophotometry and UFLC-DAD for researchers, scientists, and drug development professionals.
UV-Vis Spectrophotometry in Dissolution Testing: A Modern Guide for Robust and Sustainable Pharmaceutical Analysis
This article provides a comprehensive resource for researchers and drug development professionals on the application of UV-Vis spectrophotometry in dissolution testing.
UFLC-DAD-ESI-MS Fundamentals: A Comprehensive Guide for Pharmaceutical and Biomedical Analysis
This article provides a comprehensive exploration of Ultra-Fast Liquid Chromatography coupled with Diode Array and Electrospray Ionization Mass Spectrometry (UFLC-DAD-ESI-MS), a powerful hyphenated technique pivotal in modern analytical laboratories.
Mastering Thermal Stability: A Comprehensive Guide to Minimizing Temperature Variations in Spectrophotometric Measurements for Biomedical Research
This article provides a systematic framework for researchers, scientists, and drug development professionals to understand, mitigate, and correct for the effects of temperature variation in spectrophotometric analysis.
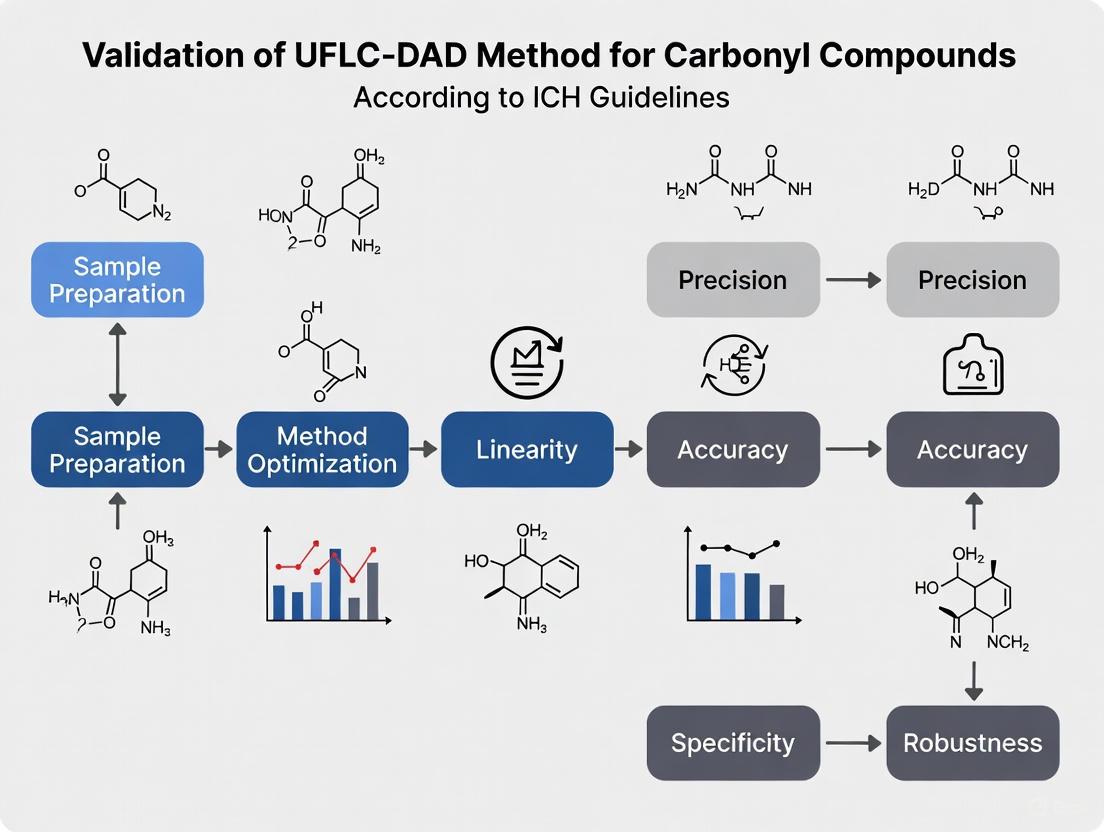
Complete Validation of a UFLC-DAD Method for Carbonyl Compounds Analysis: An ICH Q2(R2) Compliant Framework
This article provides a comprehensive guide for researchers and pharmaceutical professionals on validating a UFLC-DAD method for quantifying carbonyl compounds, following the latest ICH Q2(R2) guidelines effective June 2024.
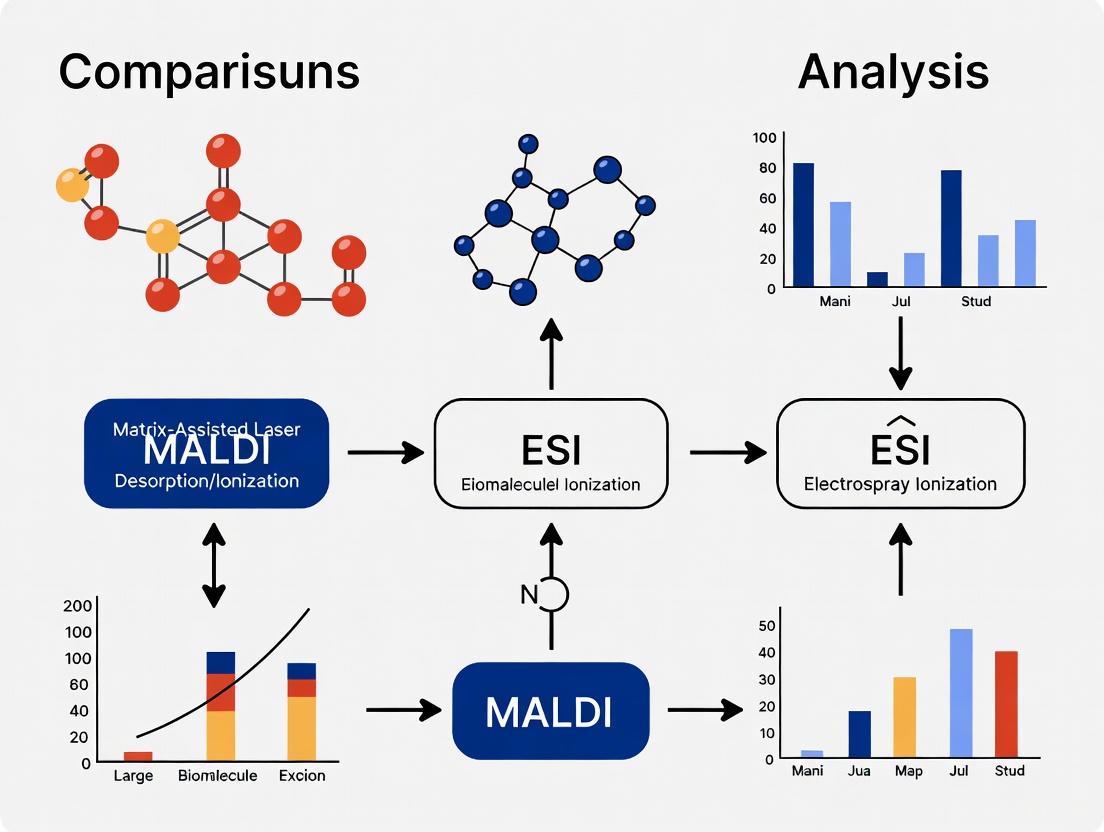
MALDI vs ESI: Choosing the Right Ionization Technique for Large Biomolecule Analysis
This article provides a comprehensive comparison of Matrix-Assisted Laser Desorption/Ionization (MALDI) and Electrospray Ionization (ESI) for the analysis of large biomolecules such as proteins and peptides.